Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
Objective UTK5.5: Dysproteinemic Nephropathies: Describe the pathogenesis of the nephropathies associated with dysproteinemia.
Competency 2: Organ System Pathology; Topic UTK: Kidney; Learning Goal 5: Renal Syndromes
Patient Presentation
A 68-year-old man presents to the nephrology clinic with swelling of both lower extremities that has gradually worsened over the past 2 months, fatigue, and easy bruising. He has had hypertension for 15 years, which has been well controlled on enalapril for the last 5 years, and he is being evaluated for a restrictive cardiomyopathy and worsening cardiac function. Review of systems shows no history of recent injury, recent travel, cough, or chest pain. He reports that he has never smoked or used smokeless tobacco and that he does not drink alcohol or use drugs. He is allergic to ciprofloxacin. Family history is significant for hypertension and type II diabetes mellitus in his father. There is no family history of renal disease or cancer.
Diagnostic Findings, Part 1
Physical Examination
On physical examination, his temperature is 98.2°F (36.8°C); pulse rate, 86/min; blood pressure, 150/96 mm Hg; respiratory rate, 18/min; and body mass index, 30.42 kg/m2. Physical examination reveals a well-built, alert, and oriented man in no acute distress. Examination of head, eyes, ears, nose, and throat is remarkable for mild dermal thickening and purple discoloration around the eyes. The lungs reveal faint bilateral rales on auscultation. Cardiac examination reveals mild jugular venous distension with a normal S1 and S2, a low-pitched S3, and a loud S4 gallop with neither rubs nor pericardial knock. Neither palpable hepatosplenomegaly nor abdominal fluid wave is noted. Bilateral pitting edema of the lower extremities, 3+, to the level of the knee is noted. The neurological examination is unremarkable.
Questions/Discussion Points, Part 1
What Is the Differential Diagnosis Based Upon History and Physical Findings?
This patient’s main presenting complaint is gradually worsening edema over the past 2 months, for which he is undergoing cardiac evaluation. The presence of hypertension, bilateral rales, jugular venous distension, and bilateral lower extremity edema is supportive of edema of cardiac origin. However, patients can have more than one etiology for edema, so causes referable to the liver and kidneys should also be considered.
Broadly, edema can occur due to increased hydrostatic pressure, as in congestive heart failure or constrictive pericarditis. Impaired venous return due to deep venous thrombosis could lead to increased hydrostatic pressure and edema in a local region. The absence of recent travel and the bilateral nature of his lower extremity edema make a deep venous thrombosis less likely. Any etiology that lowers plasma albumin levels, be it albuminuria in nephrotic syndrome or reduced albumin synthesis in decompensated chronic liver disease, can cause generalized edema due to decreased plasma oncotic pressure and hypoproteinemia.
What Are the Next Best Steps in the Diagnostic Evaluation of This Patient? Why?
In a patient with reduced cardiac output (cardiac failure), edema due to decreased renal perfusion or reduced glomerular filtration rate should be considered, so a serum blood urea nitrogen or serum creatinine would be helpful. These indices serve as indirect measures of glomerular filtration rate. In cardiac failure, a decrease in renal perfusion triggers the juxtaglomerular cells to convert prorenin in the blood to renin. Renin converts angiotensinogen to angiotensin I, which is then converted to the active form, angiotensin II, by angiotensin-converting enzyme, a potent arteriolar vasoconstrictor. This conversion causes the release of aldosterone, which inappropriately prompts the kidney to reabsorb more sodium and water, increasing intravascular volume and blood pressure and potentially creating peripheral edema and pulmonary edema.
A comprehensive metabolic profile, including liver function tests and serum electrolytes, would help assess for the presence of liver or renal functional impairment. In reduced cardiac output, low serum potassium would suggest activation of the renin–angiotensin–aldosterone axis. Serum albumin levels would detect hypoalbuminemia, which, if present, would point to reduced plasma oncotic pressure as a cause for the patient’s edema. If not included in the comprehensive panel, a lipid profile might be helpful, particularly if the presence of nephrotic syndrome due to renal disease is suspected.
A screening urinalysis to include gross, chemical, and microscopic evaluation would determine significant protein loss in the urine. Associated urinary findings, such as the presence of oval fat bodies or fatty casts, would be consistent with significant urine protein loss, as in nephrotic syndrome. He has no urinary complaints, so other significant urine findings might further elucidate an asymptomatic cause. If protein is detected in the urine, a 24-hour urine collection may provide clues to the etiology of his edema by quantitation or characterization of protein excretion. In particular, such characterization would demonstrate whether he has one of the 3 described general patterns of urine protein loss: Glomerular—with primarily albumin loss (selective proteinuria, as in minimal change disease) or multiple plasma protein loss (nonselective proteinuria, as in membranous nephropathy), Tubular—with primarily loss of small-molecular-weight proteins (as in hypertensive nephrosclerosis), or Overproduction—as in plasma cell dyscrasias like multiple myeloma, with loss of monoclonal light chain immunoglobulin (Ig) fragments in urine.
Given the patient’s faint rales, a chest X-ray would help determine the presence and extent of pulmonary edema.
An echocardiogram would be of use in determining the presence and extent of cardiac insufficiency. Because the patient is already undergoing cardiac evaluation, reviewing his chart might reveal that these studies have been performed. Comparison to current studies might be informative.
Diagnostic Findings, Part 2
Laboratory Studies
The patient’s serum creatinine is 3.4 mg/dL today, up from 1.8 mg/dL 6 months ago.
Results of serum electrolytes are within reference ranges. There is hypoalbuminemia with inversion of the serum albumin:globulin ratio. The liver function tests show total serum protein is 9.5 g/dL (normal range: 6.0-8.0 g/dL); albumin, 3.0 g/dL (normal range: 3.2-4.6 g/dL); and globulin, 6.5 g/dL (normal range: 2.0-3.5 g/dL). The remainder of the comprehensive metabolic profile, including liver transaminase tests, is unremarkable. The fasting lipid profile shows an elevated total cholesterol of 280 mg/dL (desirable level <200 mg/dL); elevated triglycerides, 340 mg/dL (desirable level <150mg/dL); estimated low-density lipoprotein, 200 mg/dL (optimal level <100 mg/dL); and decreased high-density lipoprotein, 26 mg/dL (normal range: 40-60 mg/dL).
Urinalysis reveals specific gravity, 1.015; pH, 7; protein, 4+; and negative glucose, ketones, blood, bilirubin, nitrite, and leukocyte esterase. On microscopic evaluation, rare red blood cells (0-2/high-power field [HPF]), rare white blood cells (0-2/HPF), and a few fatty (1-2/low-power field [LPF]) and rare (0-1/LPF) granular casts are noted.
A 24-hour urine collection reveals significant proteinuria: 31 g protein/24 hours (nephrotic range). Characterization of the protein by urine protein electrophoresis reveals overproduction-type proteinuria with increased globulins.
Imaging
A chest X-ray reveals a slightly enlarged cardiac silhouette and slightly prominent vascular markings, compared with studies done 6 months ago.
Echocardiogram performed as part of the evaluation for restrictive cardiomyopathy a week ago reveals moderate concentric left ventricular hypertrophy with a “speckled” pattern, a left ventricular ejection fraction of 55%, and diastolic dysfunction.
Questions/Discussion Points, Part 2
Do the Findings in This Patient Support the Presence of a Specific Syndrome? If So, What Is the Differential Diagnosis in General and As It Applies to This Patient?
This patient has massive proteinuria in the nephrotic range, which, in association with the presence of hypoalbuminemia, edema, and hyperlipidemia, represents nephrotic syndrome. Hyperlipidemia in nephrotic syndrome occurs due to increased hepatic lipogenesis, which is a nonspecific reaction to falling oncotic pressure secondary to hypoalbuminemia. The presence of fatty casts is indicative of lipiduria.
In adults, the differential diagnosis of nephrotic syndrome 2 includes focal and segmental glomerulosclerosis, membranous nephropathy, membranoproliferative glomerulonephritis, minimal change disease, and glomerular damage due to systemic diseases like diabetes mellitus, amyloidosis, systemic lupus erythematosus, certain infections (hepatitis B and C viruses), and drug ingestion.
There is evidence of cardiac dysfunction in this patient with signs of right and left heart failure, as evidenced by jugular venous distension and bilateral lower extremity edema on physical examination, as well as changes suggestive of pulmonary edema on chest X-ray. The presence of additional heart sounds, S3 and S4, combined with the left ventricular hypertrophy noted on echocardiography, is suggestive of a restrictive cardiomyopathy, for which the patient is being evaluated. The patient has a history of essential hypertension that could also lead to concentric left ventricular hypertrophy.
In a patient with nephrotic syndrome and a restrictive cardiomyopathy, one might consider that these 2 entities are related and suggest multi-organ involvement by a systemic disease. The increase in total serum protein, including an elevation in serum globulins and an inverted albumin:globulin ratio, is indicative of excess globulin production that is known to occur in clonal plasma cell dyscrasias and B-cell lymphoproliferative disorders. Both can lead to the deposition of excess Igs or light chains as amyloid. Amyloidosis can involve multiple organ systems and should be considered in the differential diagnosis of patients presenting with nephrotic syndrome. Coexistence of restrictive cardiomyopathy makes amyloid a likely cause in this patient. Renal damage due to diabetes mellitus, lupus nephritis, and hepatitis could also have a similar presentation and should be excluded.
What Additional Diagnostic Tests May Be Helpful in This Patient?
In this patient with nephrotic syndrome, massive proteinuria, and cardiac concerns (with suspected multi-organ involvement), a serum protein electrophoresis and serum immunofixation study would detect monoclonal Igs in serum, if present, and help elucidate the type of globulin fraction being produced in excess (α1, α2, β1, β2, or γ). Intact monoclonal Ig proteins with or without free light chains (“M” spike/paraprotein) are found in the serum electrophoresis of patients with certain B-cell lymphoproliferative disorders and plasma cell dyscrasias like multiple myeloma.
In some cases of B-cell lymphoproliferative disorders and plasma cell dyscrasias, only light chains are produced without intact Igs. In these instances, the monoclonal light chains can be detected only by urine electrophoresis, as they are rapidly cleared from the serum due to their small size. Hence, urine protein electrophoresis and urine immunofixation are also important to perform because they will detect intact Igs or free light chains in the urine, if present. Serum and urine electrophoresis are quantitative tests and can determine the amount, but not the type, of abnormal protein present. Serum and urine immunofixation define the type of monoclonal protein present but do not quantitate it. Therefore, these studies are both helpful and complementary.
Because this patient’s initial laboratory studies did not reveal an abnormal random serum glucose, fasting and postprandial serum glucose measurements to rule out diabetes mellitus are likely not indicated. Serologic tests for serum antinuclear antibodies (ANA) and screening studies to detect hepatitis B and C virus infections should be performed.
A renal biopsy is indicated to determine the cause of nephrotic syndrome. If abnormal proteins are detected in the above serum and urine tests, a skeletal survey and a bone marrow evaluation are indicated to further evaluate for the presence of a B-cell lymphoproliferative disorder or plasma cell dyscrasia. If amyloidosis is strongly suspected, an endomyocardial biopsy could be performed to evaluate for the presence of amyloid in cardiac tissue.
Diagnostic Findings, Part 3
Additional Laboratory Studies
Both serum and urine electrophoresis in this patient show an “M” spike (Figure 1) in the γ region, which is noted to be a monoclonal IgG-λ band by serum immunofixation. Urine immunofixation reveals a monoclonal IgG-λ band as well.
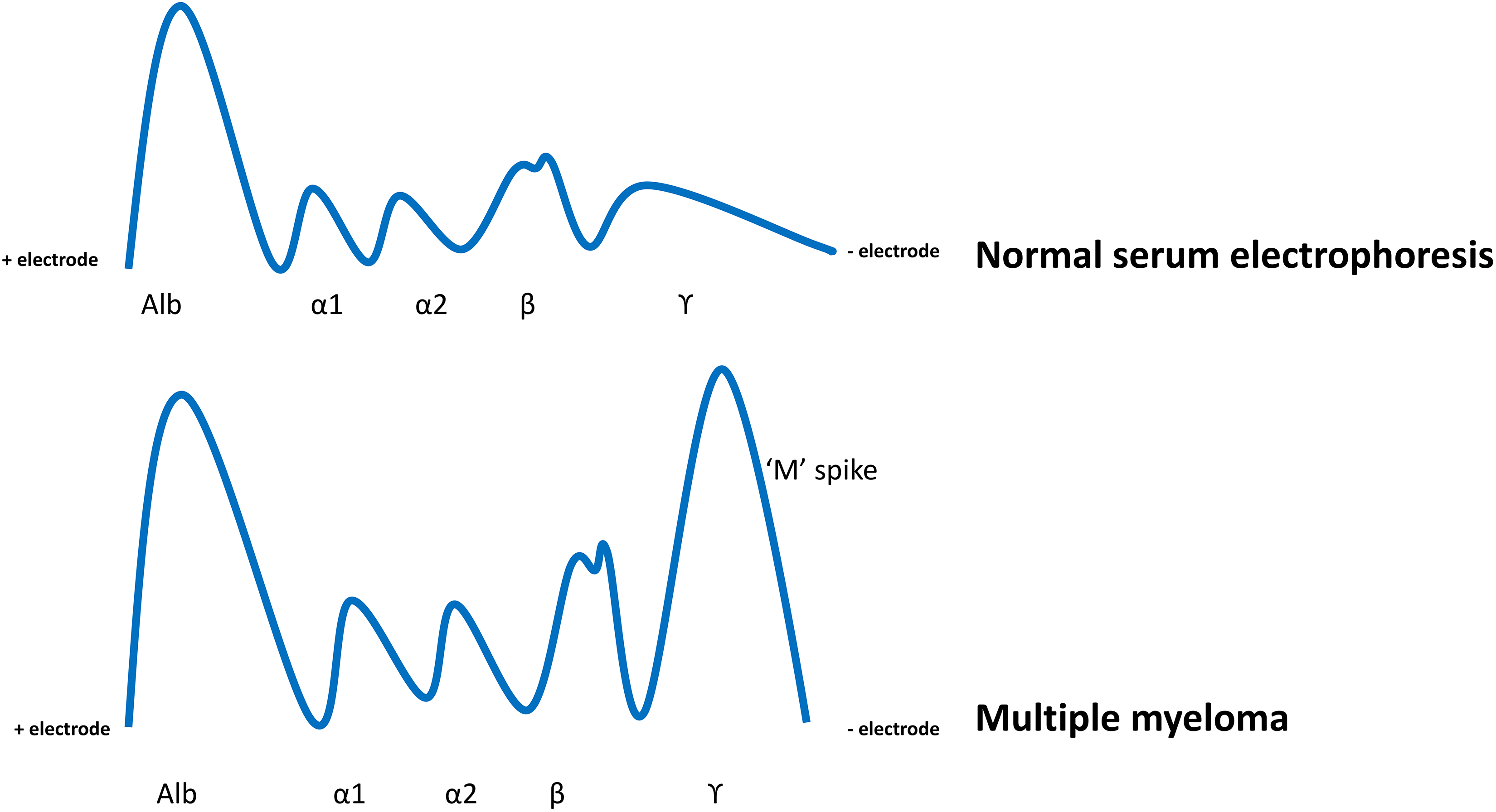
Serum electrophoresis patterns. Alb indicates albumin, α1-α1 protein fraction (α1-antitrypsin, thyroid-binding globulin, and transcortin), α2-α2 protein fraction (acute phase reactant), β-β fraction (has 2 peaks β1-transferrin and β2-β lipoprotein), ϒ-γ protein fraction.
His screening serum ANA test is negative. Serologic studies to detect hepatitis B and C virus infections are negative.
Renal Biopsy Findings
A renal biopsy is performed, and tissue is collected for light, direct immunofluorescence, and electron microscopy. An adequate sample of renal tissue is available for evaluation.
Light microscopy shows 21 glomeruli with vague nodularity and an increase in mesangial matrix material with compression of the capillary lumens (Figures 2 and 3). The glomeruli are not hypercellular. No significant inflammation or crescent formation is identified. The expanded mesangial areas and the walls of a few blood vessels show pale, homogeneous, amorphous, eosinophilic material that is weakly positive on periodic acid–Schiff (PAS) stain (Figure 4) and nonargyrophilic (silver negative) on Jones methenamine silver-PAS stain (Figure 5). A Congo red stain is strongly positive in the glomeruli (Figure 6) and in the blood vessel walls, which on polarization reveals classic apple green birefringence (Figure 7).
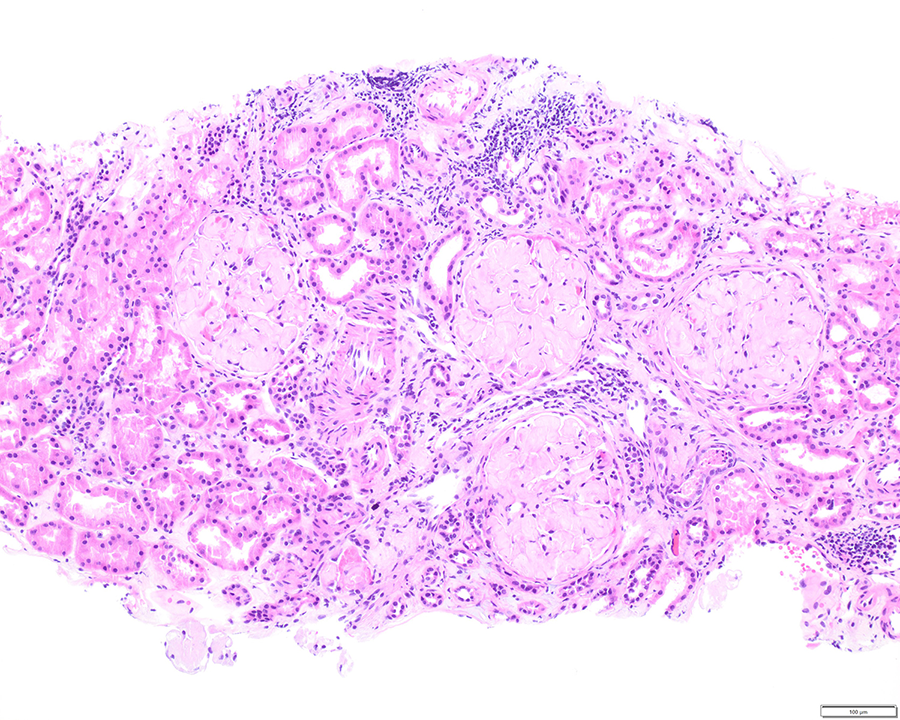
Glomeruli with prominent mesangial expansion and vague nodularity (hematoxylin-eosin, original magnification ×100).
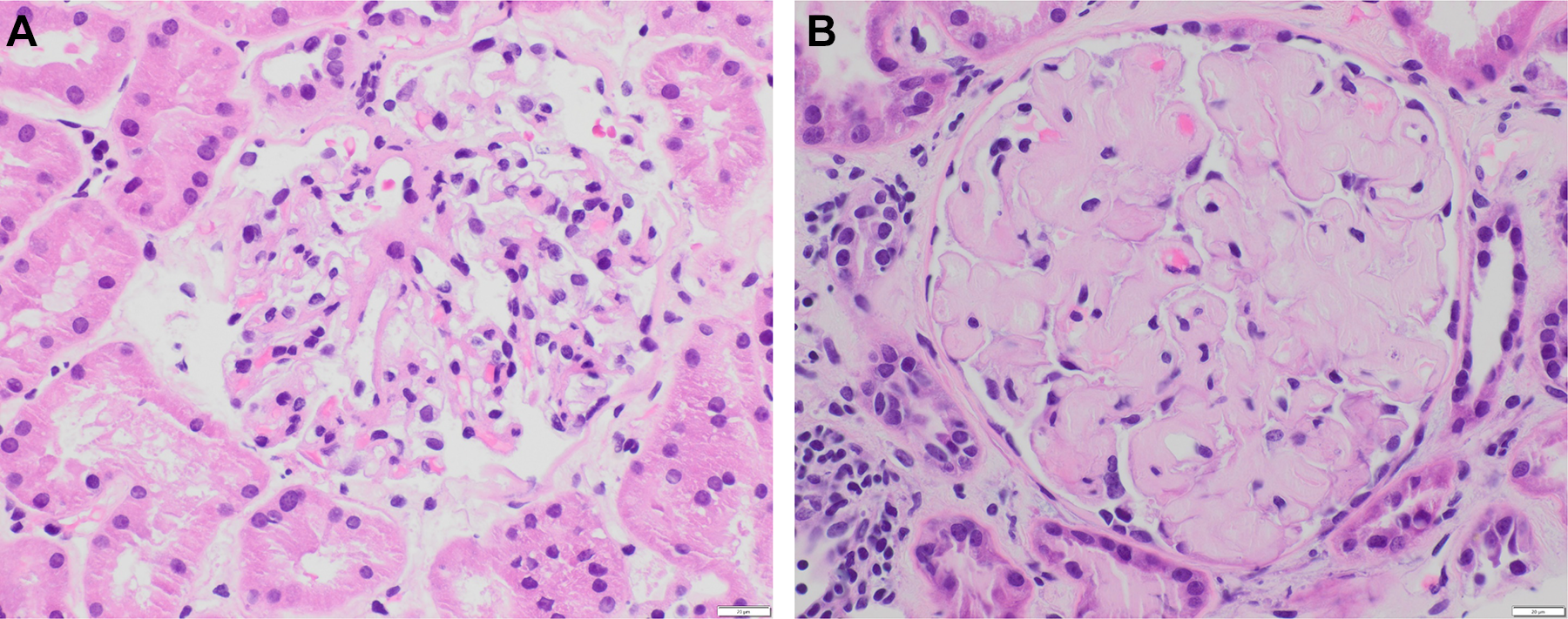
Normal glomerulus with patent capillary lumens (A) contrasts with a glomerulus (B) that demonstrates nodularity and prominent mesangial expansion by amorphous, eosinophilic, acellular material. Note the impingement on the capillary lumens by the amorphous material (hematoxylin-eosin, original magnification ×400).
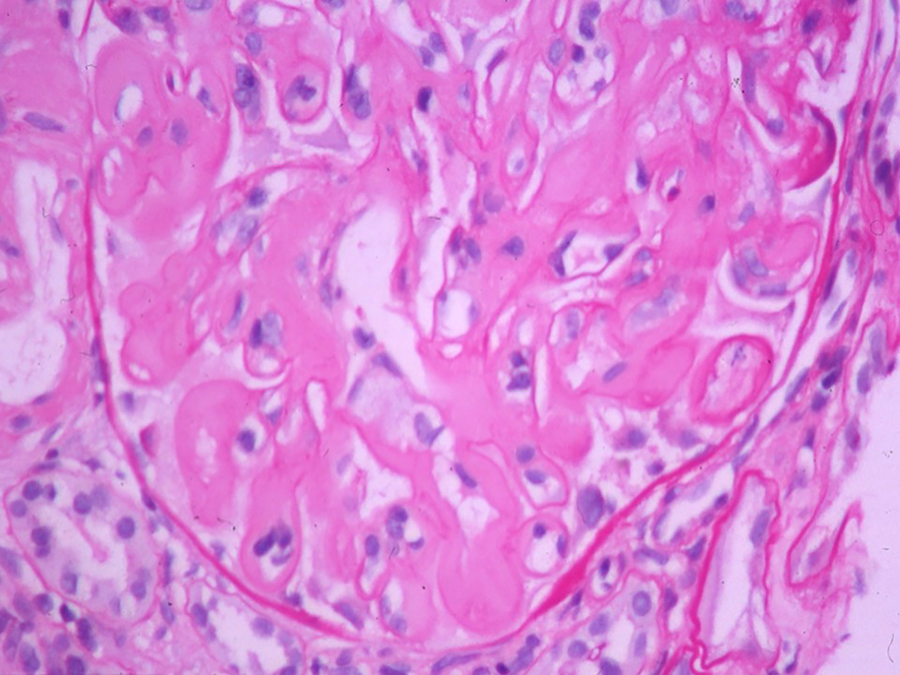
Glomerulus with mesangial areas expanded by weakly PAS-positive acellular deposits (original magnification ×400). PAS indicates periodic acid–Schiff.
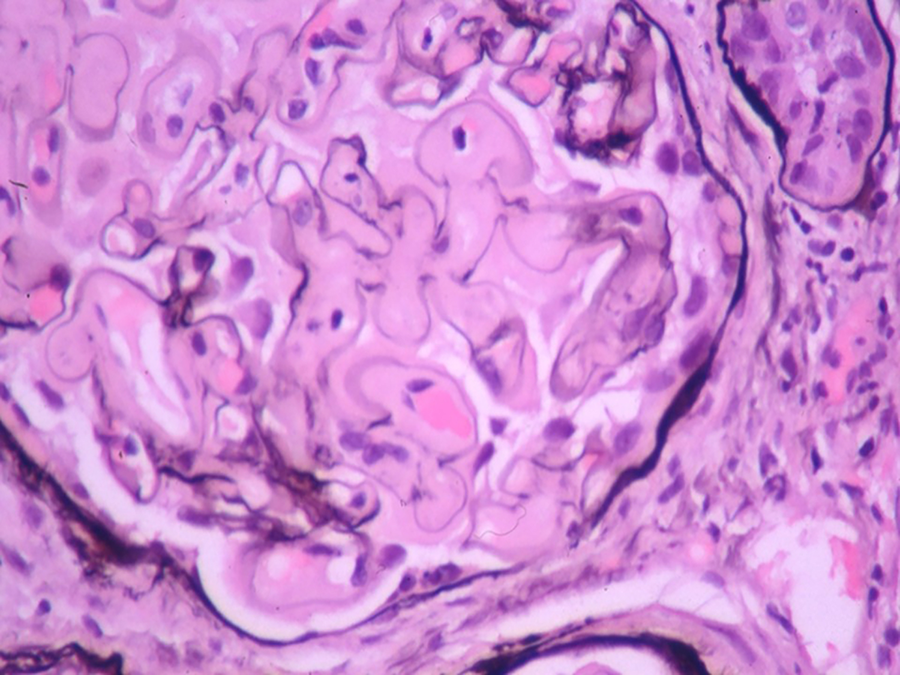
Glomerular mesangial deposits are nonargyrophilic (silver negative) (Jones methenamine silver periodic acid–Schiff, original magnification ×400).
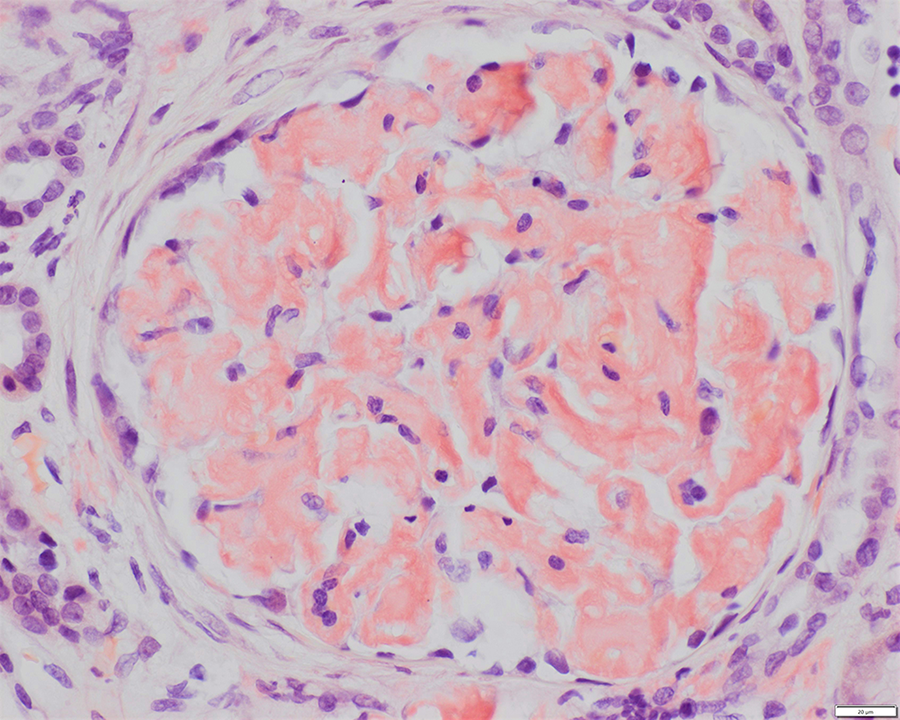
Glomerulus with Congo red–positive mesangial deposits (amyloid) with orange to salmon pink staining (Congo red, original magnification ×400).
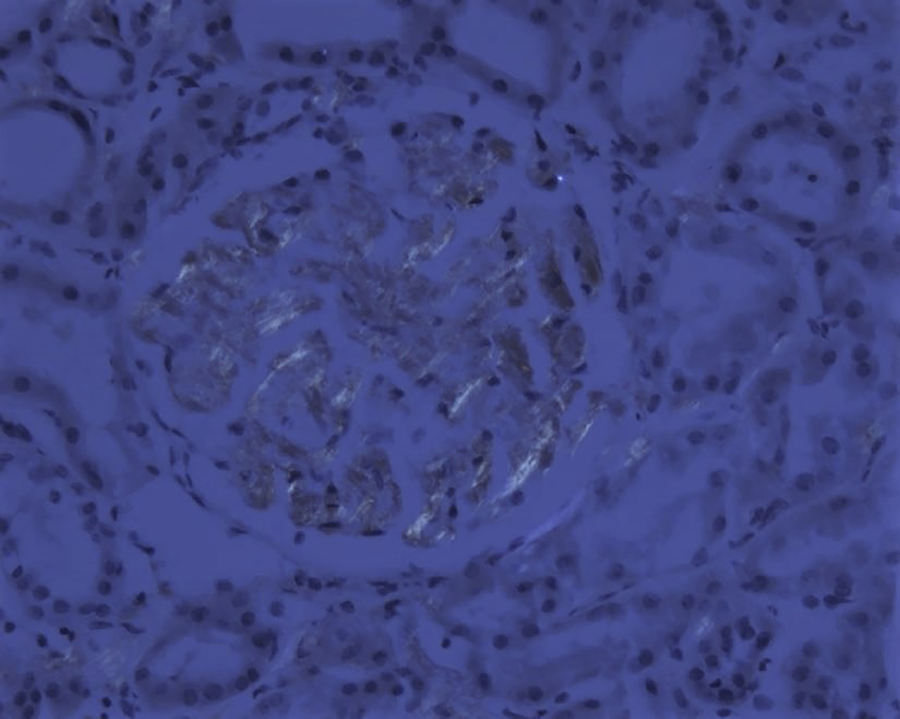
Glomerular amyloid deposits with apple green birefringence when viewed under polarized light (Congo red, original magnification ×200).
On direct immunofluorescence microscopy, 10 glomeruli are available for evaluation. The glomerular deposits show positive staining with IgG (3+) and dominant positivity for λ (3+). No staining is detected for κ, IgM, IgA, c1q, c3, c4, albumin, or fibrinogen (negative). A thioflavin-T stain shows strong positive staining in the mesangium and along some capillary loops by immunofluorescence (Figure 8).
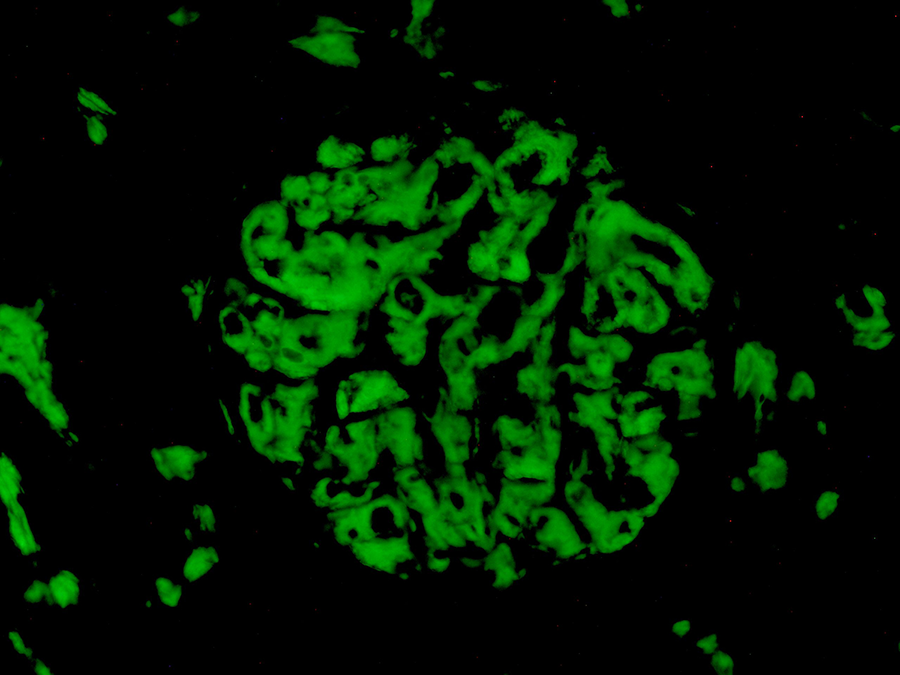
Glomerular mesangial amyloid deposits with intense thioflavin-T positive immunofluorescent staining (thioflavin-T direct immunofluorescence, original magnification ×400).
Electron microscopy shows randomly dispersed, nonbranching fibrils within the expanded mesangial matrix (Figures 9 –11), along glomerular capillaries, and within blood vessel walls. These fibrils range from 8.2 to 13.5 nm in diameter, with a mean of 10.8 nm, based upon 30 measurements.
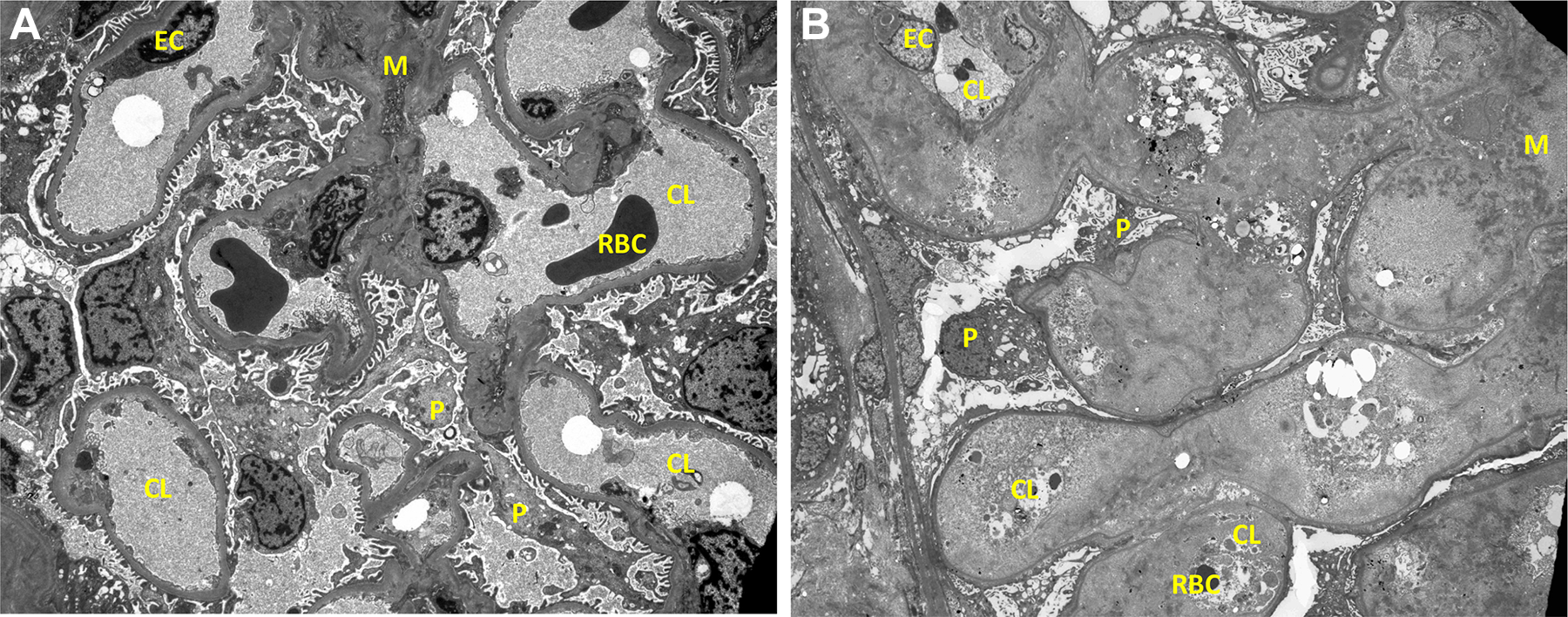
Normal glomerulus (A) with patent capillary lumens and normal ultrastructure contrasts to a glomerulus (B) with narrowed glomerular capillary lumens due to deposits of amorphous material (transmission electron microscopy, original magnification ×700). CL indicates capillary lumen; EC, endothelial cell; M, mesangium; P, podocyte; RBC, red blood cell.
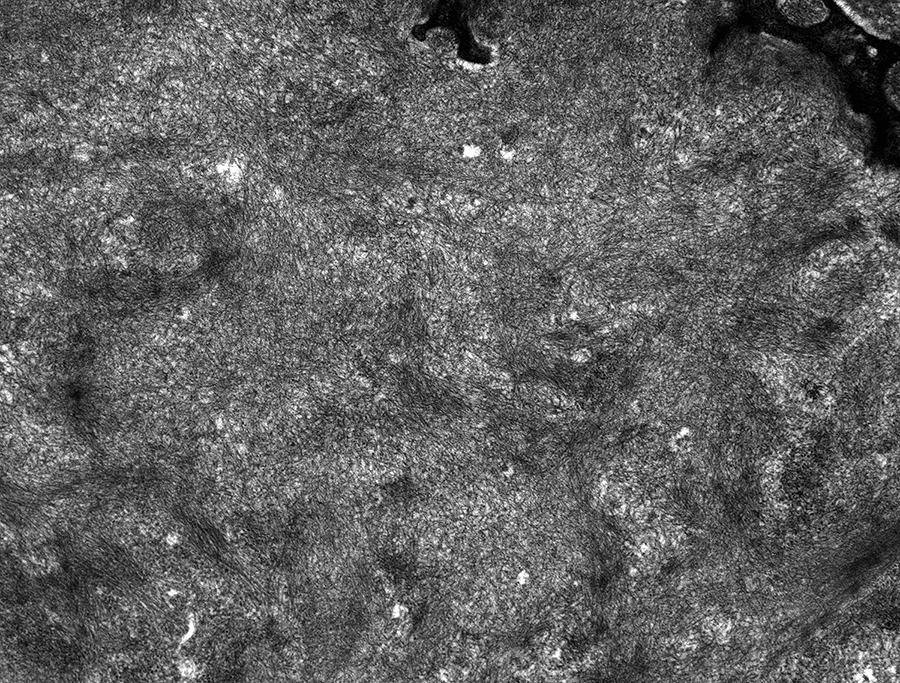
Fibrillary deposits present in mesangial matrix (transmission electron microscopy, original magnification ×6000).
Based on the light, direct immunofluorescence, and electron microscopic findings, a diagnosis of renal amyloidosis is rendered.
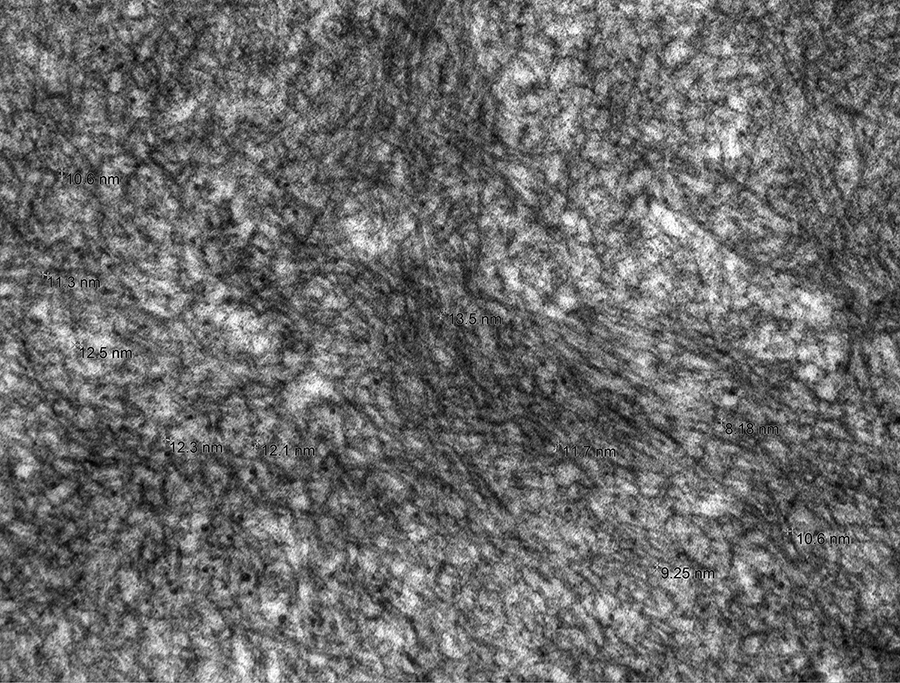
Nonbranching, small-diameter fibrils present in mesangial matrix (transmission electron microscopy, original magnification ×25 000).
Additional Imaging and Pathologic Studies
In light of the renal biopsy findings, additional imaging and pathologic studies are performed. A skeletal survey shows multiple sharp, “punched out” lytic lesions in the skull and in the left femoral head. Bone marrow aspirate and biopsy reveal a plasma cell dyscrasia in the spectrum of multiple myeloma, with the presence of a clonal population of λ-restricted plasma cells, estimated to comprise approximately 45% of the bone marrow cell population. An endomyocardial biopsy reveals the presence of focal subendocardial amyloid deposits with staining characteristics similar to those described in the patient’s renal biopsy specimen.
Questions/Discussion Points, Part 3
What Is the Diagnosis for This Patient?
Systemically, this patient meets criteria for a diagnosis of multiple myeloma. He has greater than 10% of a clonal bone marrow plasma cell population in addition to end-organ damage attributable to the plasma cell proliferative disorder as noted clinically (elevated serum creatinine greater than 2 mg/dL) and on renal biopsy. He also has osteolytic lesions on skeletal radiography.
More specifically, in this patient with multiple myeloma, a diagnosis of primary AL (λ light chain) amyloidosis can be rendered. The presence of dominant λ light chain staining on direct immunofluorescence of the renal biopsy specimen is concordant with the findings noted on serum and urine immunofixation as well as the bone marrow aspirate and biopsy findings. The patient’s reported history of easy bruising and dermal thickening with purple discoloration around his eyes (noted on physical examination) could possibly represent dermal involvement by amyloid deposits.
What Does Amyloidosis Represent? Describe Some Clinical Presentations and Patterns of Organ Involvement in Amyloidosis
Amyloidosis refers to a heterogeneous group of disorders characterized by extracellular deposition of randomly oriented insoluble fibrils which form β-pleated sheets. These represent misfolded proteins that are prone to self-aggregation, which leads to tissue damage. Currently, nearly 30 different forms of amyloid have been described, including hereditary forms of amyloidosis. 3 -5 Amyloid can be composed of various types of polypeptides and precursor proteins, including monoclonal Ig light chains (AL amyloid), monoclonal Ig heavy chains (AH amyloid), serum amyloid A protein (AA amyloid), leukocyte chemotactic factor type 2 (ALECT2 amyloid), β2-microglobulin, procalcitonin, apolipoprotein AI/AII, gelsolin, and cystatin C.
Amyloid deposition is interestingly diverse: It can be systemic or localized to 1 site, acquired (secondary to long-term dialysis in end-stage renal disease) or hereditary, present with life-threatening symptoms, or found incidentally. Amyloid of any type is slightly more common in men than women and usually presents in the sixth decade of life.
Localized amyloidosis involves a particular organ and can be clinically silent or life-threatening, depending upon the organ affected. For example, cardiac amyloidosis can be insidious or present with restrictive cardiomyopathy and fatal arrhythmias. Amyloid β-amyloidosis is localized to the central nervous system and presents with Alzheimer’s symptoms of dementia.
In contrast, amyloidosis can be systemic, and the clinical features vary depending on the type of amyloid fibrils and the level of organ dysfunction caused by the deposition. For example, as in this patient, primary AL amyloidosis can be systemic. In some cases, it can lead to heart failure with left ventricular hypertrophy, hepatomegaly, renal involvement with nephrotic syndrome, macroglossia, articular deposits, skin/soft tissue deposition, and neuropathy.
Describe the Pathogenesis of Amyloidosis
In amyloidosis, misfolding or abnormal folding of the precursor proteins is a key feature. In their normal state, the fibrils are soluble. But they turn insoluble and aggregate in extracellular spaces within organs to cause destruction of tissue, pressure atrophy, and often compromised organ function.
Broadly, some circumstances that lead to amyloid deposition are
6
: An overabundance of a certain protein that is normally present at a low level—this is seen in AA type amyloidosis where excess serum AA amyloid is produced by the liver in response to chronic inflammatory stimuli, as seen in conditions like rheumatoid arthritis and ankylosing spondylitis. Exposure to a normal level of a potentially amyloidogenic protein over a long period of time—such as transthyretin in senile amyloidosis and amyloid-β protein in Alzheimer disease. An acquired or an inherited mutation leading to the formation of an abnormal amyloidogenic protein—such as AL amyloid in AL amyloidosis.
Regardless of their composition, amyloid fibrils have a propensity to self-aggregate and take on a β-pleated structure. As more such molecules assemble, they align and interdigitate and form sheets that are resistant to degradation. The amyloidogenic precursor protein and intermediate and proto filaments each have toxicities independent of the amyloid deposits. The unique Congo red staining and birefringent nature of amyloid are due to the accumulation of the fibril monomers as β-pleated sheets, which is evident on X-ray crystallography.
Briefly Describe Features of AL Amyloidosis
Amyloid light chain protein is composed of monoclonal light chain fragments that can be either κ or λ and are produced by monoclonal B-lymphocytic or plasma cell proliferations, such as multiple myeloma in this patient. When these proteins are deposited as amyloid, the disorder is termed AL amyloidosis. Typically, there is alteration in the normal protein sequence by replacement of a single amino acid that determines the amyloidogenic potential of the protein; AL amyloidosis is the most common type of systemic amyloidosis. The excess intact Ig with or without free light chains produces an M spike on serum or urine electrophoresis. Monoclonal Igs or free κ or λ light chains can also be detected in the urine, as seen in this patient.
In the kidney, the glomeruli are most commonly involved (greater than 95% of cases), followed by the vascular compartment (about 80%), and, lastly, the interstitium (about 60%). Kidney involvement is most frequent and is seen in up to two-thirds of AL amyloidosis cases at the time of diagnosis. 7,8 Although AL is the most common type, other types of amyloidosis that involve the kidney include ALECT2 (commonly involves interstitium), Afib (massive glomerular obliteration), AApo AI, AApo AIV (involves predominantly the medullary interstitium), and AGel (mostly glomerular).
What Methods Are Used to Detect Amyloid?
Histologic demonstration of amyloid on clinical specimens from sites such as the kidney, tongue, abdominal fat pad, and rectum is considered diagnostic. Histologically, amyloid in the kidney appears as homogenous, amorphous, pale eosinophilic material on hematoxylin and eosin stain. In addition to being found in glomeruli, amyloid deposits may involve the peritubular interstitium and the walls of blood vessels to varying degrees in some types of amyloidosis. Hence, each compartment of the biopsy should be screened carefully for the presence of amyloid deposits. Special stains, as discussed under the renal biopsy findings in the case, are useful in the tissue diagnosis of amyloid.
Direct immunofluorescence staining to various Ig and light chain components is useful in detecting clonality of the light chains in AL amyloidosis. Moreover, these stains are helpful in characterizing other disorders in the spectrum of dysproteinemic nephropathies.
At an electron microscopic level, demonstration of glomerular, interstitial, and vascular involvement by amyloid fibrils is possible. The various types of amyloid all appear as randomly dispersed, rigid, nonbranching fibrils ranging from 7 to 14 nm in diameter. 4,5 They tend to form compact arrays close to cell membranes but show loose arrays when away from cell surfaces. The fibrils in the glomeruli typically first appear in the mesangium. In more advanced cases, the fibrils encroach into the peripheral capillary wall with significant narrowing of the glomerular capillary lumina. Although histologic stains are often employed when amyloidosis is suspected, demonstration of the characteristic nonbranching fibrils by electron microscopy remains a mainstay of diagnosis.
Immunohistochemistry for AA, AL, and other amyloidogenic proteins can be performed on histologic specimens to distinguish between the various types of amyloid.
Direct amyloid typing by analysis of the peptide spectra with the use of laser microdissection/mass spectrometry has the highest sensitivity and specificity for the detection of amyloid and overcomes many of the cross-reactivity issues that are seen with immunohistochemical and immunofluorescent staining. This detection method is not routinely used for diagnosis, due to its limited availability in only select centers. 5
Amyloidosis: Treatment and Prognosis
A detailed discussion on the treatment of amyloid is beyond the scope of this case study. In general, the prognosis depends on the type of amyloid and the organ systems involved. The prognosis is poor, if the disease is left untreated. Cardiac involvement has been noted to be an independent negative prognostic factor.
In broad terms, treatment in AL amyloidosis is chemotherapy based and directed at eliminating the monoclonal protein and the plasma cell clone that is producing the monoclonal protein. In contrast, for example, in AA amyloidosis, efforts are directed toward treatment and control of the underlying inflammatory condition.
Kidney transplantation is offered in selected cases of AL and hereditary amyloidosis. Recurrent amyloid deposition in the renal transplant occurs in about 20% to 30% of those transplanted patients, due to active underlying disease.
What Are Dysproteinemic Nephropathies?
Overproduction of monoclonal Igs results in dysproteinemia. Dysproteinemic nephropathies refer to a group of disorders that causes renal impairment attributable to deposition, precipitation, or crystallization of abnormal Ig molecules or proteins. These abnormal molecules or proteins are produced during clonal proliferation of plasma cells in plasma cell dyscrasias or by atypical lymphocytes in B-cell lymphoproliferative disorders. 2,3,9 In 2012, the International Kidney Monoclonal Gammopathy Research Group introduced the term monoclonal gammopathy of renal significance to refer to monoclonal gammopathies that result only in kidney dysfunction due to monoclonal Ig deposition. 2 Although there are many ways that monoclonal Igs can injure the kidney, one of the more common and clinically significant manifestations is the deposition as amyloid within the kidney. The kidney is commonly affected by abnormal Igs because it is exposed to nearly a quarter of the cardiac output every minute.
Disorders included under the umbrella of dysproteinemic nephropathies can be categorized based upon whether they have organized or nonorganized monoclonal Ig deposits. Examples 10 -12 of organized deposit disorders include Ig-related amyloidosis (AL, AHL, AH), fibrillary glomerulonephritis, immunotactoid glomerulonephritis, cryoglobulinemic glomerulonephritis type 1, and light-chain proximal tubulopathy.
As noted previously, abnormal monoclonal Igs serve as precursors for some types of amyloid deposition. The most common type of renal amyloidosis is due to Igs associated with plasma cell dyscrasias that produce monoclonal light chains (AL). 7,13,14 Rarely, the amyloid is composed of both Ig heavy and light chains (AHL) or can be composed of Ig heavy chains only (AH).
Although the main focus of this case discussion is renal amyloidosis, Table 1 elaborates on the clinical presentation, serum Ig class, and serum free light chains that are seen in the major entities considered in the morphologic differential for renal amyloidosis. 2,4,7,10,12,15 -17
Dysproteinemic Nephropathies as a Histologic Differential Diagnosis for Renal Amyloidosis.*
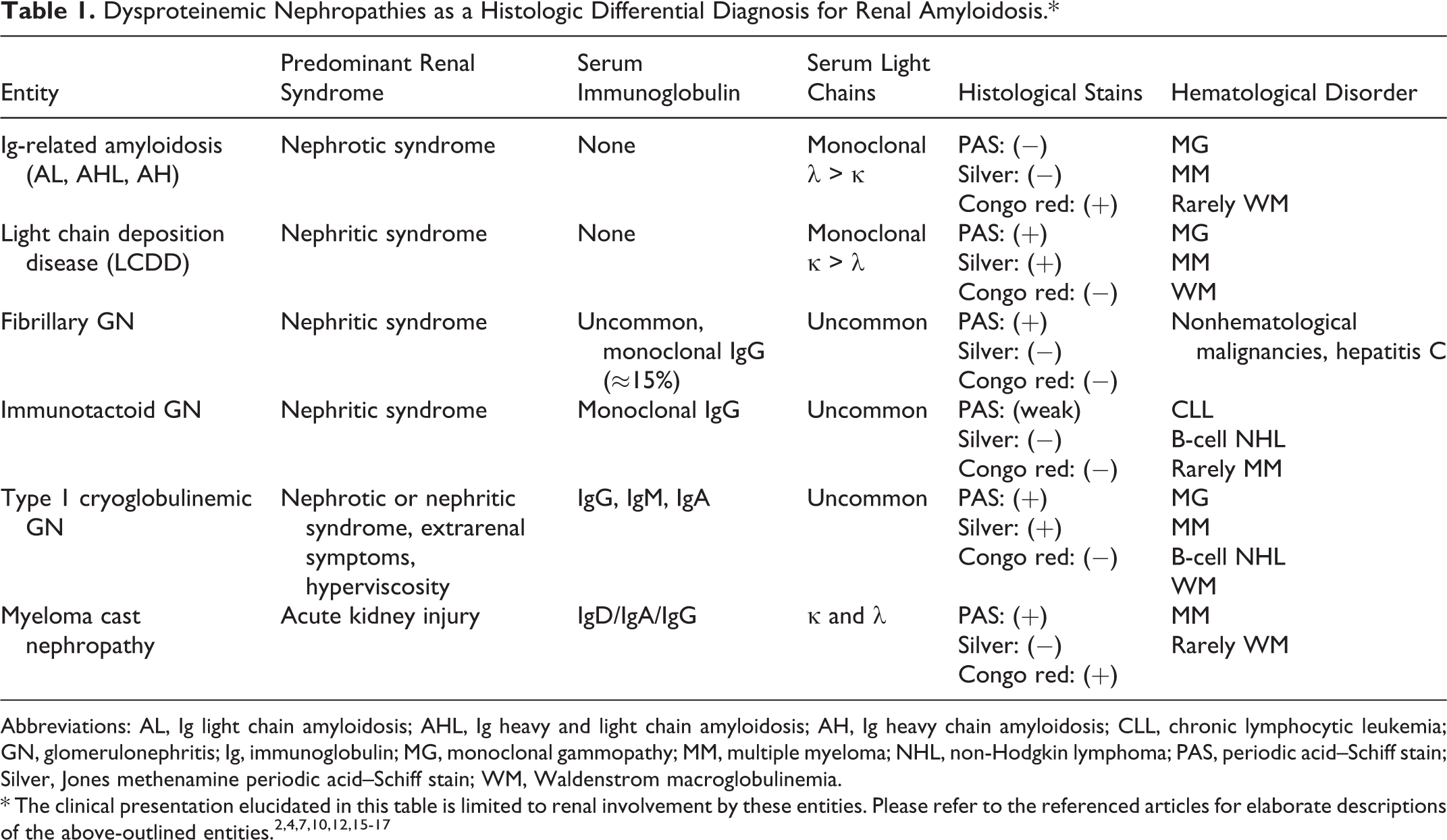
Abbreviations: AL, Ig light chain amyloidosis; AHL, Ig heavy and light chain amyloidosis; AH, Ig heavy chain amyloidosis; CLL, chronic lymphocytic leukemia; GN, glomerulonephritis; Ig, immunoglobulin; MG, monoclonal gammopathy; MM, multiple myeloma; NHL, non-Hodgkin lymphoma; PAS, periodic acid–Schiff stain; Silver, Jones methenamine periodic acid–Schiff stain; WM, Waldenstrom macroglobulinemia.
Teaching Points
Dysproteinemia is a state of excess production of monoclonal proteins (such as the entire Ig, light chain only, or, rarely, heavy chain only) by neoplastic B-lymphocytes or plasma cells. Dysproteinemic disorders frequently affect the kidney, leading to renal dysfunction due to monoclonal Ig deposition.
The differential diagnosis of edema is broad and includes cardiac, renal, and liver origins.
Evaluation of patients with edema should include renal (serum creatinine and blood urea nitrogen), liver (total protein and serum albumin levels), and cardiac (electrocardiogram and echocardiogram) function tests.
Nephrotic syndrome is defined by the presence of proteinuria (more than 3.5 g/24 hours) with hypoalbuminemia, edema, hyperlipidemia, and lipiduria.
The differential diagnosis of nephrotic syndrome in adults includes minimal change disease, focal and segmental glomerulosclerosis, membranous nephropathy, and glomerular damage due to systemic diseases like diabetes mellitus, amyloidosis, and systemic lupus erythematosus.
Intact Igs, with or without free light chains, form the “M” spike on serum or urine electrophoresis in patients with certain B-cell lymphoproliferative disorders and plasma cell dyscrasias.
Serum and urine immunofixation studies define the type of monoclonal protein present but do not quantitate it. These studies are complementary to serum and urine electrophoresis studies, which are quantitative.
Monoclonal Igs may injure the kidney. Clinically significant manifestations of such injury may include the extracellular deposition as amorphous amyloid deposits within the glomeruli, peritubular interstitium, and blood vessels.
Amyloidosis is a heterogeneous group of disorders characterized by extracellular accumulation of insoluble misfolded proteins acquiring a β-pleated structure, giving rise to characteristic histologic features.
In the kidney, on light microscopy, amyloid appears as pale, homogenous, light pink, amorphous material that is PAS negative, silver negative, and Congo red positive (salmon pink or orange by light microscopy with apple green birefringence when viewed under polarized light).
On direct immunofluorescence microscopy, dominant staining of either κ or λ light chain immunoreactants may be seen in certain types of amyloid, such as primary AL amyloidosis. Thioflavin-T stain is positive in most cases of amyloidosis.
By electron microscopy, amyloid fibrils range from 7 up to 14 nm in diameter and are randomly dispersed, rigid, and nonbranching.
AL (light chain) amyloidosis should be suspected in patients with nephrotic syndrome, renal insufficiency, congestive heart failure, peripheral neuropathy, skin/soft tissue abnormalities (easy bruising), and obscure hemorrhage.
Treatment options in AL amyloidosis are chemotherapy based and are directed at prevention of end-organ damage, elimination of the monoclonal protein, and elimination of the neoplastic clonal B-cell or plasma cell population that produces the protein.
Prognosis of renal amyloidosis is based on age and extent of tissue involvement, with cardiac involvement portending a poor prognosis. Amyloidosis may recur in transplanted organs.
Other monoclonal gammopathies can involve the kidney and can appear similar to amyloid histologically. However, Congo red stain, immunofluorescence, and electron microscopic findings help differentiate and distinguish these different entities from each other and from amyloid.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
