Abstract
The following fictional case is intended as a learning tool within the Pathology Competencies for Medical Education (PCME), a set of national standards for teaching pathology. These are divided into three basic competencies: Disease Mechanisms and Processes, Organ System Pathology, and Diagnostic Medicine and Therapeutic Pathology. For additional information, and a full list of learning objectives for all three competencies, see http://journals.sagepub.com/doi/10.1177/2374289517715040.1
Keywords
Primary Objective
Objective UTK 5.3: Immune-Mediated Renal Disease. Compare and contrast the mechanisms of immune complex and antibody-mediated glomerulonephritis.
Competency 2: Organ System Pathology; Topic UTK: Kidney; Learning goal 5: Renal syndromes.
Secondary Objective
Objective UTK 5.1: Nephritic Syndrome. Describe the proliferative and pro-inflammatory pathologies of conditions presenting with nephritic syndrome.
Competency 2: Organ System Pathology; Topic UTK: Kidney; Learning goal 5: Renal syndromes.
Patient Presentation
A 61-year-old man presents to the emergency department with shortness of breath and bilateral leg pain with swelling. The shortness of breath has been present for the past 3 weeks, and he is now short of breath after walking less than 1 block. Additionally, he complains of an occasional dry cough. He has also noticed increased leg pain and swelling with a burning sensation. He has tried aspirin for the pain, but it provides minimal relief. He is taking no other medications. He has no significant past medical or surgical history. No one in his family has experienced similar symptoms. His social history includes a 7-pack-year history of smoking in high school and alcohol consumption of 1 drink per week. He worked in construction for 30 years including concrete, carpentry, and ironworking until 3 years prior to presentation.
On review of systems, he reports an unintentional 20-pound weight loss, decreased appetite, early satiety, and jaw pain and fatigue while chewing, which have all developed in the past month. He does not report fevers, chest pain, or hemoptysis. He has not had any change in urinary frequency or hematuria.
Vitals signs are temperature of 37.2 °C, blood pressure of 154/101 mm Hg, pulse of 82 beats per minute, respiratory rate of 20 breaths per minute, and a body mass index of 29.2 kg/m2. On physical examination, there are bibasilar crackles but no wheezing upon auscultation of his lungs. He has a regular heart rhythm without murmurs or rubs. He has 3+ pitting edema of his legs up to his mid-thighs bilaterally. On neurologic examination, he has intact cranial nerve function, 1/5 strength on bilateral dorsal and plantar flexion of his feet, and 4/5 strength in all other major muscle groups of the arms and legs. He has decreased sensation to light touch and pain below the knees with no sensation below the ankles. Sensation is preserved in his upper extremities.
Diagnostic Findings, Part 1
In the emergency department, a complete blood count, complete metabolic panel, and urinalysis and other tests are performed. His baseline creatinine is 0.8 mg/dL. The fractional excretion of sodium (FENa) is 2.2%. The results are presented in Tables 1 to 3.
Complete Blood Count on Presentation to Emergency Department.

Abbreviations: RBC, red blood cell; WBC, white blood cell.
Complete Metabolic Panel on Presentation to Emergency Department.
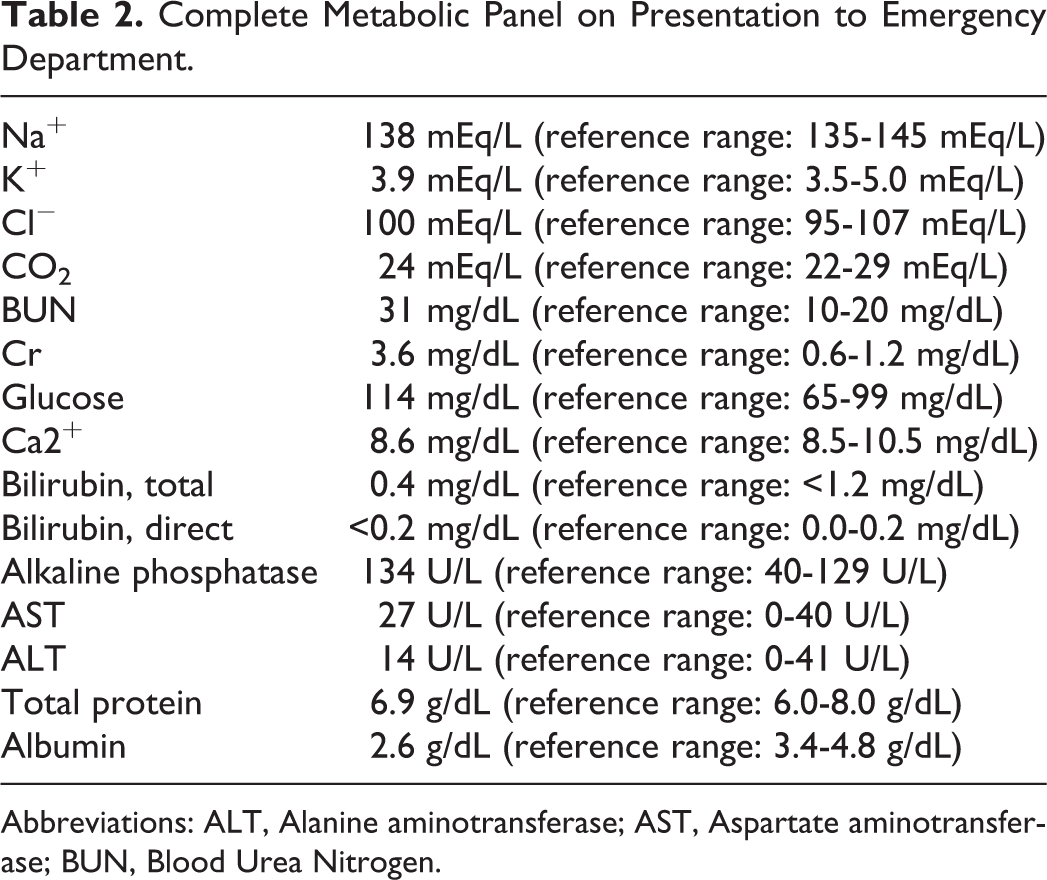
Abbreviations: ALT, Alanine aminotransferase; AST, Aspartate aminotransferase; BUN, Blood Urea Nitrogen.
Urinalysis on Presentation to Emergency Department.

Abbreviations: RBC, red blood cell; WBC, white blood cell.
Imaging studies are performed including a chest X-ray and renal ultrasound. The chest X-ray demonstrates increased interstitial prominence at the peripheral and basilar aspects of the lungs without evidence of consolidation, atelectasis, pleural effusion, or enlarged heart silhouette (Figure 1). An electrocardiogram (ECG) demonstrates normal sinus rhythm, and an echocardiogram shows mild left ventricular hypertrophy with an ejection fraction of 62% (reference range: 55%-70%). A renal ultrasound demonstrates normal-sized kidneys with mild microvascular disease. No renal stones or masses are observed. There is also no hydronephrosis to suggest a distal obstruction to the urinary tract. These imaging findings along with the urinalysis result make renal stones and malignancy less likely. Based on the lab and imaging results, the patient is admitted for further workup.
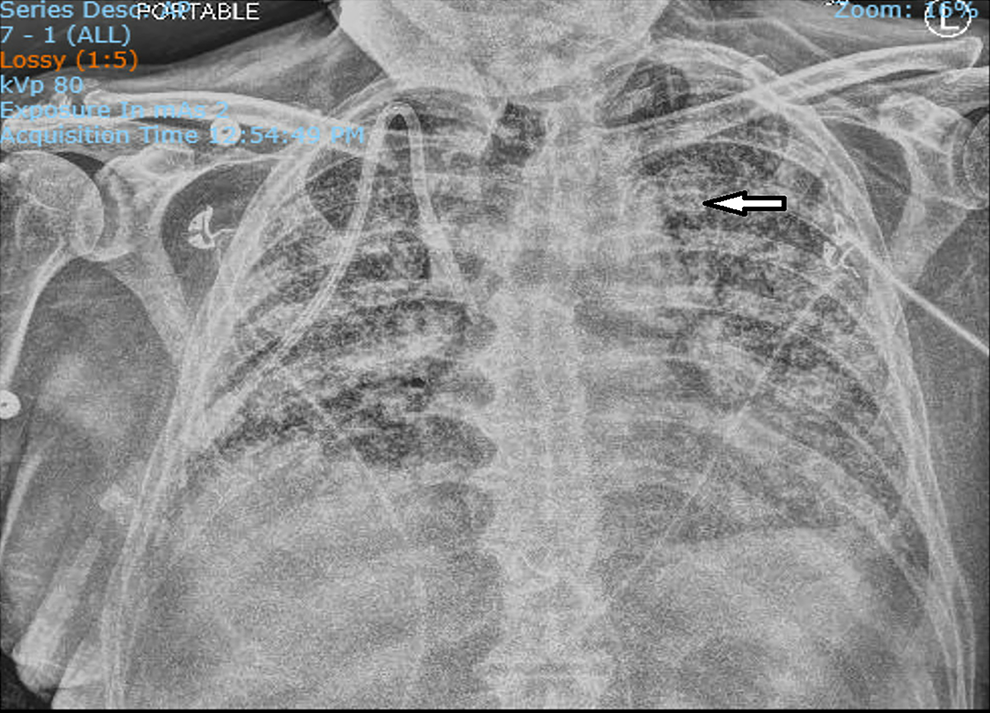
Chest X-ray showing perihilar and interstitial prominence (arrow). The dialysis catheter is inserted in the superior vena cava (the correct space), from later in the clinical course.
Questions/Discussion Points, Part 1
What Is the Significance of Red Blood Cell Casts in the Urine?
Urinalysis showed the presence of proteins and red blood cells (RBCs) and RBC casts. Red blood cell casts are seen in the urine when RBCs leak into the urinary space of the glomeruli after glomerular damage. They coalesce into RBC casts as they move through the kidney tubules and are excreted in the urine. Red blood cell casts are only observed when the kidney is directly injured. Red blood cells can also be found in the urine when there is a renal stone, renal mass, or a mass in the urinary bladder. In these scenarios, the RBCs are intact, and no casts are identified.
Based on the Initial History, Physical Examination, and Lab Data, What Is Included in the Differential Diagnosis?
The clinical differential diagnosis for a patient presenting with shortness of breath and significant edema, weight loss, joint pains, and neurologic symptoms suggests a multisystem disease. Potential etiologies would include an immune-mediated process involving cardiac, pulmonary, and renal systems. Congestive heart failure, acute kidney injury due to a variety of etiologies, and a pulmonary renal syndrome could potentially result in the presentation as described. The patient’s labs indicate he had developed acute renal failure because he had an abrupt rise in creatinine from his baseline of 0.8 to 3.6 mg/dL.
Acute renal failure can present with hypertension and lower extremity edema, as seen with this patient. It may result from a prerenal, intrarenal, or postrenal process. 2 Prerenal causes include states of true hypovolemia, as seen in dehydration or acute blood loss, or perceived hypovolemia by the kidneys, as seen in systolic heart failure or severe liver disease. The most common cause of postrenal acute renal failure is obstruction of the ureters or bladder. Finally, intrarenal processes include damage to any portion of the kidney, including the glomeruli or tubules.
Additional tests performed showed his FENa is 2.2%. Fractional excretion of sodium is used to evaluate the cause of renal failure. A FENa of <1% indicates a prerenal process, and a FENa of >2% indicates an intrinsic renal process, as in this patient, suggestive of an intrarenal process. Furthermore, urinalysis demonstrated hematuria and proteinuria (quantitated to be 1.81 g/24 hours). Renal ultrasound showed the presence of microvascular disease but no evidence of postrenal obstruction. The presence of RBC casts indicates bleeding into the kidney tubules due to damage of the glomeruli.
As noted in Figure 1, a chest X-ray showed extensive coarse interstitial markings and nodular opacities in both lung fields. The differential diagnosis for the interstitial infiltrates includes pulmonary fibrosis, interstitial pneumonia, early diffuse alveolar hemorrhage, and pulmonary edema. A cardiac cause for this patient’s symptoms was less likely since his chest X-ray did not demonstrate an enlarged heart or pleural effusion, his ECG was normal, and his echocardiogram showed a normal ejection fraction without evidence of heart failure.
Describe the Differences Between a Nephritic Syndrome and a Nephrotic Syndrome. Which Process Does This Patient Most Likely Have?
Intrinsic renal processes involving the glomeruli can be described as a nephritic syndrome or nephrotic syndrome based on laboratory data. Nephritic syndrome is characterized by hematuria, variable proteinuria, azotemia, edema, and hypertension. 2 A nephrotic syndrome is characterized by >3.5 g protein in the urine per day, hypoalbuminemia, hyperlipidemia, edema, and the lack of RBCs or WBCs in the urine. 2 Based on this patient’s clinical symptoms of hypertension and edema along with his azotemia, hematuria, low-grade proteinuria, and hypoalbuminemia, he had features suggestive of a nephritic syndrome.
What Is the Differential Diagnosis for the Cause of His Nephritic Syndrome Based on His Clinical History and Other Laboratory Data?
The differential for a nephritic syndrome would include broadly: Antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis such as granulomatosis with polyangiitis (GPA), microscopic polyangiitis (MPA), and eosinophilic GPA (EGPA). Anti-glomerular basement membrane disease (Goodpasture syndrome). Immune complex-mediated glomerulonephritis (GN) including immunoglobulin A (IgA) nephropathy, lupus nephritis, postinfectious GN, types I and II membranoproliferative GN, and cryoglobulinemic GN. Drug-induced papillary necrosis from the use of aspirin.
3
What Additional Tests Should Be Ordered to Further Elucidate the Cause of His Acute Kidney Injury?
Based on the differential diagnosis, the next set of laboratory tests need to evaluate for immune-mediated processes. These tests include serologies detected by either indirect immunofluorescence or direct enzyme-linked immunoassays (ELISAs). The serology tests necessary to help narrow the differential diagnosis include antinuclear antibody (ANA), double-stranded DNA (dsDNA), ANCA including myeloperoxidase (MPO) and proteinase 3 (PR3), glomerular basement membrane antibody (anti-GBM), and plasma C3 and C4 complement.
Antinuclear antibody is an antibody commonly found in multiple autoimmune diseases with multiorgan system clinical presentations, but it is nonspecific without additional clinical and laboratory findings. The presence of dsDNA antibodies is more specific for systemic lupus erythematosus (SLE). Positive tests for both ANA and dsDNA are highly specific for SLE, and lupus nephritis is the renal finding seen with the disease. 2 Antineutrophil cytoplasmic antibody is an antibody directed toward specific antigens in the cytoplasm of neutrophils and it is associated with systemic vasculitis. Myeloperoxidase is an ANCA antibody directed at MPO and its positivity is seen in MPA and EGPA. 2 PR3 is also an ANCA antibody, but its target is proteinase 3. PR3-ANCA positivity is associated with GPA. 2 Both diseases can present with nephritic syndrome.
Diagnostic Findings, Part 2
Results of additional laboratory tests are in Table 4.
Serology Results.

Abbreviations: ANCA, antineutrophil cytoplasmic antibody; ANA, antinuclear antibody; dsDNA, double-stranded DNA; GBM, glomerular basement membrane; MPO, myeloperoxidase.
A renal biopsy is also indicated because of the unexplained acute change in renal function. 4 Representative images are seen in Figures 2 and 3.
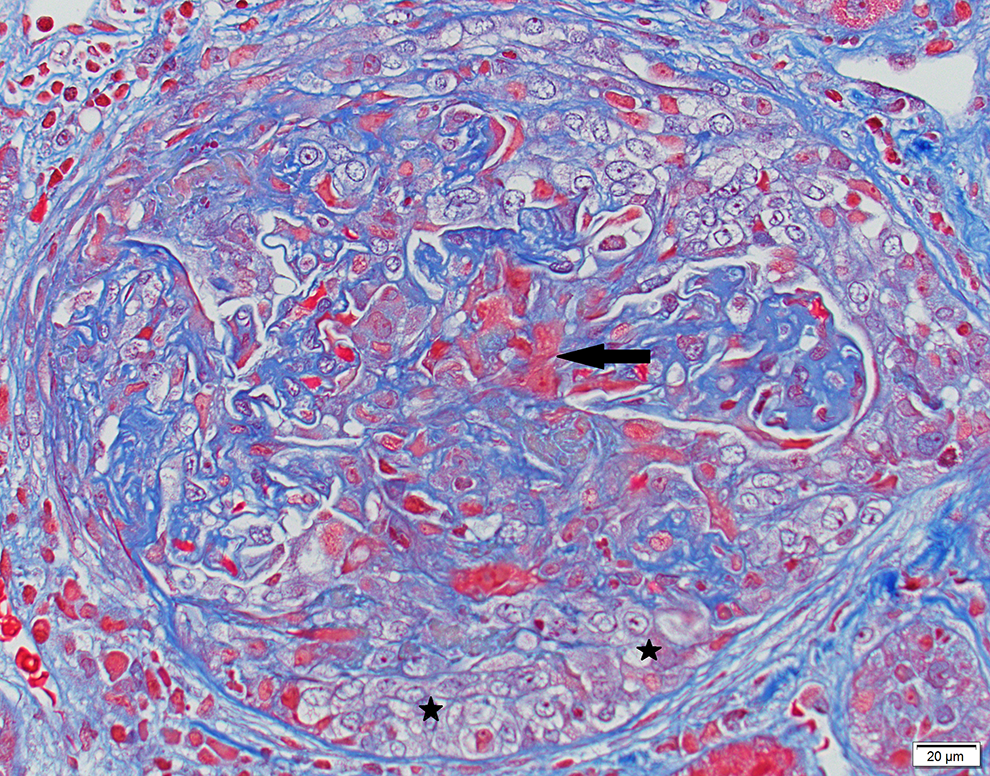
Trichrome stain shows foci of fibrinoid necrosis (arrow) and crescent (Star).
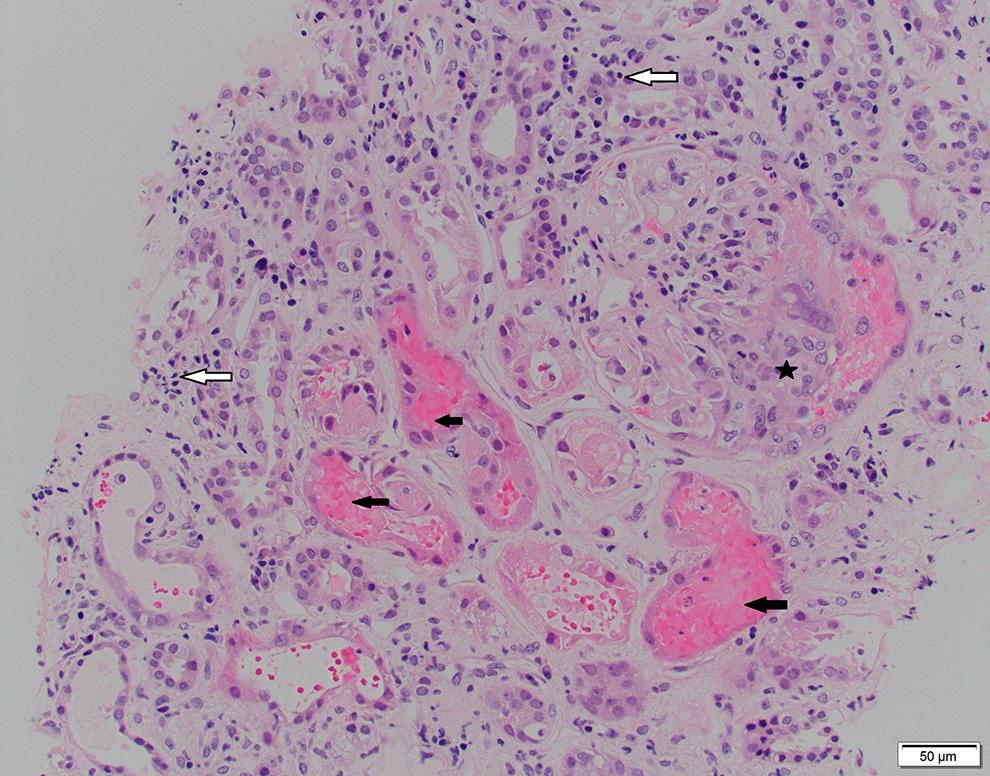
Hematoxylin and eosin stain showing glomerular crescent (star), tubules with injury and red cell casts (black arrows), and interstitial inflammation (white arrows).
Questions/Discussion Points, Part 2
Describe the Significance of the Antibody and Renal Biopsy Results
Based on the laboratory tests, lupus nephritis (negative ANA and dsDNA), C3 glomerulonephritis (normal complement levels), and ANCA-mediated GN can be ruled out as well (negative ANCA and ELISA for MPO and PR3). However, anti-GBM is positive (>8 units). Light microscopy revealed almost all the glomeruli to have cellular or fibrocellular crescents with foci of fibrinoid necrosis (Figure 2). There was also evidence of acute tubular injury with loss of the brush border, luminal cell exfoliation, and rare granular casts (Figure 3). There was no coagulative necrosis in the biopsy to suggest analgesic-induced papillary necrosis.
Describe the Progression of Glomerular Injury That Leads to Crescent Formation
A crescent is described as 2 or more layers of cells between the visceral and parietal epithelium that partially or completely fill the Bowman’s space. Discontinuation or breakdown of the capillary wall leads to extravasation of plasma proteins and fibrin into the Bowman’s space, allowing for an influx of macrophages and T lymphocytes. The pro-inflammatory cascade is then activated, with interplay of interleukin 1 and tumor necrosis factor α, additionally recruiting more inflammatory cells. This leads to complement activation, downstream neutrophil infiltration within the capillary loops, and proliferation of visceral and parietal epithelial cells. 2,5 Taken together, these processes lead to crescent formation. 3 Early in the disease process, the crescents have increased cellularity, and once the active inflammation ceases, fibrocellular and fibrous crescents develop due to collagen deposition and fibroblast proliferation. 2,3 Inflammation and antibodies directed at the vessels also leads to vascular destruction with necrosis and fibrin deposition. 2 Any inflammatory glomerular disease can result in the formation of crescents.
What Is the Differential Diagnosis for Crescentic Glomerulonephritis and the Pathogenesis of Each Disease?
Diverse disease processes may result in crescentic GN. The etiology can be narrowed down based on clinical assessment coupled with laboratory results and immunofluorescence performed on the kidney biopsy. Some major categories are discussed below:
Pauci-immune necrotizing and crescentic GN: Autoantibodies against neutrophil lysosomal components (antineutrophil cytoplasmic antibodies or ANCA) can initiate signal transduction pathways that lead to neutrophil activation, alternate complement pathway, and endothelial cell damage, eventually leading to crescent formation and necrosis. The trigger for this autoantibody generation is usually unknown and no antibody or immune complex deposition noted on immunofluorescence staining and electron microscopy, 2 hence the name Pauci-immune. Examples of ANCA-associated vasculitis include GPA, MPA, and EGPA. In all these 3 categories, the morphologic changes are very similar by renal biopsy and are further distinguished based on lab abnormalities and other organs involved. Granulomatosis with polyangiitis is associated with granulomatous inflammation of the respiratory tract and positive PR3-ANCA. 2 Microscopic polyangiitis presents with hemoptysis, hematuria, cutaneous, and neurological findings and with MPO-ANCA in at least 50% cases. 2 Eosinophilic GPA may have variable ANCA expression (MPO>PR3 and absent in some cases).
Goodpasture syndrome and anti-GBM GN are often described together in the literature. Anti-GBM GN manifests in the kidney as acute kidney injury, hematuria, and proteinuria. When there is simultaneous pulmonary involvement, which may start with dyspnea, overt hemoptysis, and chest infiltrates on imaging, it is called Goodpasture syndrome. The pathogenesis of this entity is due to an autoantibody targeting the noncollagenous 1 domain of α3 chain of collagen type IV, which is found in the glomerular basement membrane. Interaction between the autoantibody and its target in the GBM leads to injury and crescent formation. 2 It is characterized by numerous crescents, usually all at the same stage of development. Bright, linear immunofluorescent immunoglobulin G (IgG) staining of the glomerular capillary loop basement membrane is noted, 2,4,6 while other immunoglobulins and complements are negative. 2,6
Immune complex-mediated GN includes IgA nephropathy, lupus nephritis, and membranoproliferative GN (MPGN) where circulating antigen–antibody complexes deposit in the kidneys. 2
In IgA nephropathy, there is granular IgA staining in the mesangium and capillary loops on immunofluorescence, 2,4 with corresponding deposits noted on electron microscopy. It may be associated with infections, hepatic cirrhosis, inflammatory bowel disease, neoplasia, and so on. Henoch-Schönlein purpura or IgA vasculitis refers to IgA deposition in skin and gastrointestinal tract microvasculature, resulting in purpuric presentation, abdominal pain, and so on. (This entity will not be further discussed here due to the limited scope of this article.)
Lupus nephritis is the renal manifestation of SLE. The classic immunofluorescence finding is a “full house” pattern of granular staining with IgG, IgA, immunoglobulin M (IgM), C3, and C1q. 4 Patients with SLE may have manifestations in many other organ systems including the skin, mucous membranes, joints, heart, lungs, and central nervous system.
Membranoproliferative GN defines a histomorphologic pattern of glomerular injury that presents with proliferative glomeruli, IgG and C3 staining, corresponding immune complex deposits on electron microscopy, and rarely crescents. The diseases that may result in an MPGN pattern of injury include broadly infections, immunologic, dysproteinemias, malignancies, chronic liver disease, and so on (this list is by no means exhaustive, but for the purpose of this article, we will limit our description). The etiology can be traced back to the dysregulation of the complement pathway and immune complex deposition in the glomeruli. The differential diagnoses with their associated clinical and laboratory findings are summarized in Table 5.
Crescentic Glomerulonephritis Clinical and Laboratory Findings.
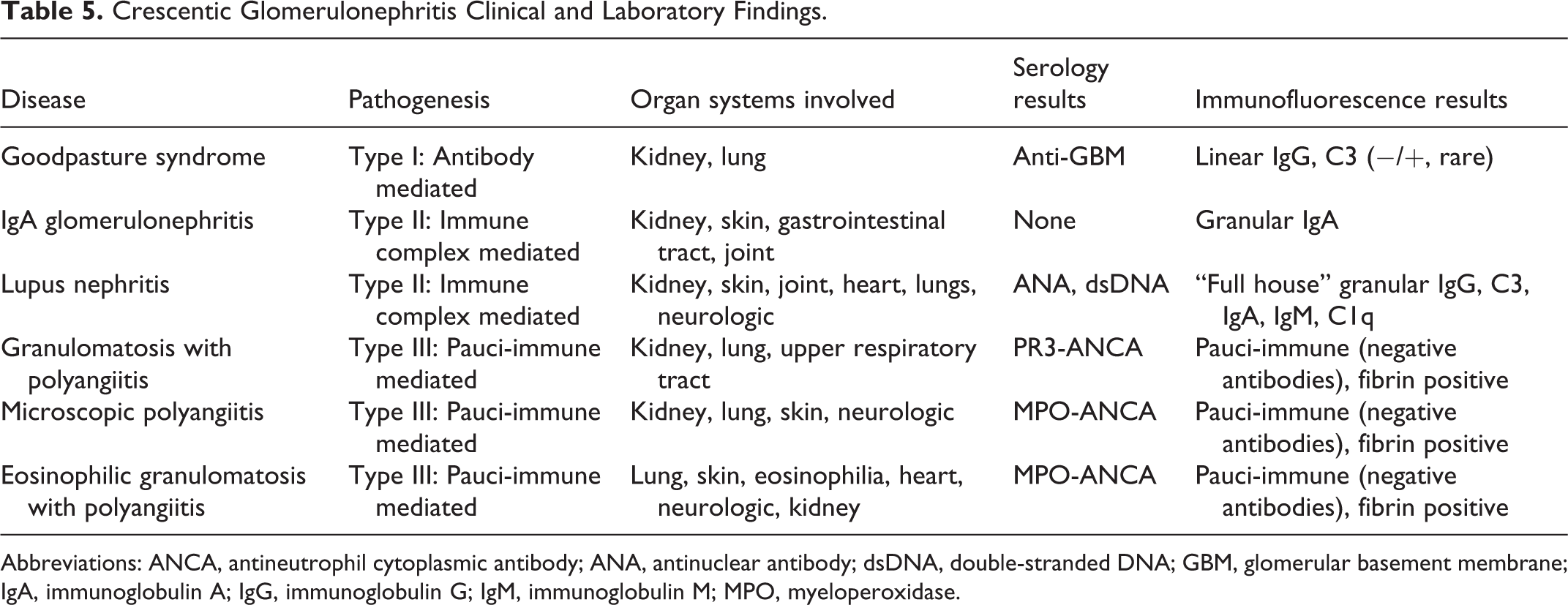
Abbreviations: ANCA, antineutrophil cytoplasmic antibody; ANA, antinuclear antibody; dsDNA, double-stranded DNA; GBM, glomerular basement membrane; IgA, immunoglobulin A; IgG, immunoglobulin G; IgM, immunoglobulin M; MPO, myeloperoxidase.
Diagnostic Findings, Part 3
Immunofluorescence stains are performed on the renal biopsy. The glomeruli show linear global capillary loop staining for IgG with minimal costaining for C3 (Figure 4). This indicates IgG antibodies are bound to the glomerular basement membrane. Almost all glomeruli also show fibrinoid necrosis, which is positive for fibrinogen (Figure 5). This may occur in severe cases of crescentic GN regardless of the underlying etiology. The glomeruli are negative for IgA, IgM, C1q, kappa light chains, and lambda light chains.
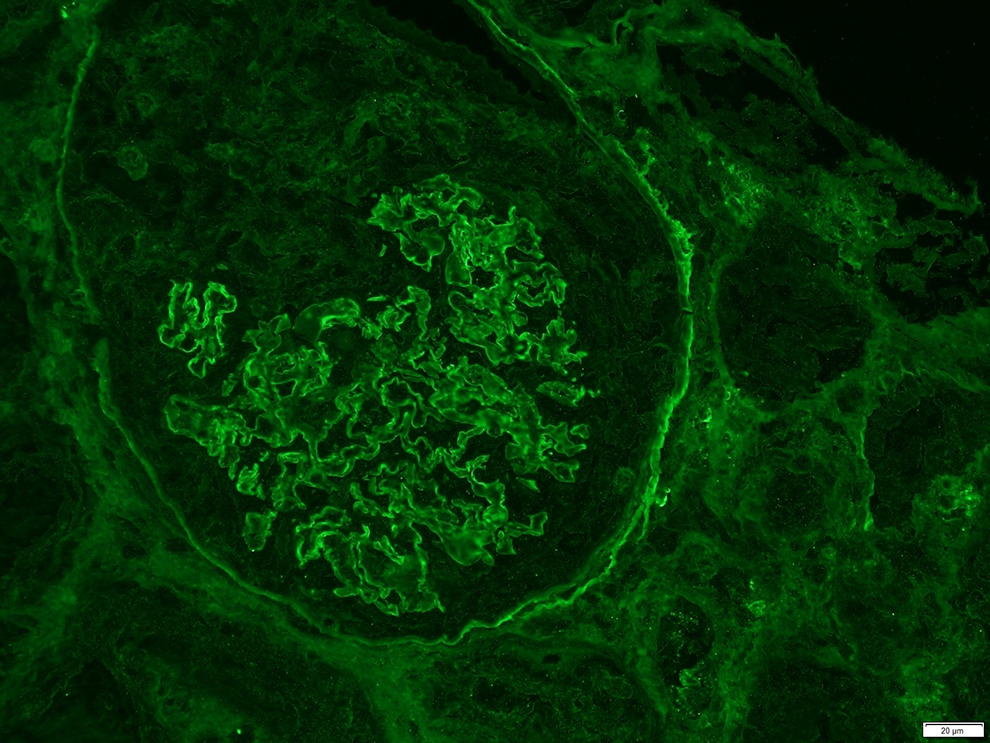
Immunofluorescence stain shows linear immunoglobulin G (IgG) staining on the glomerular capillary loops.
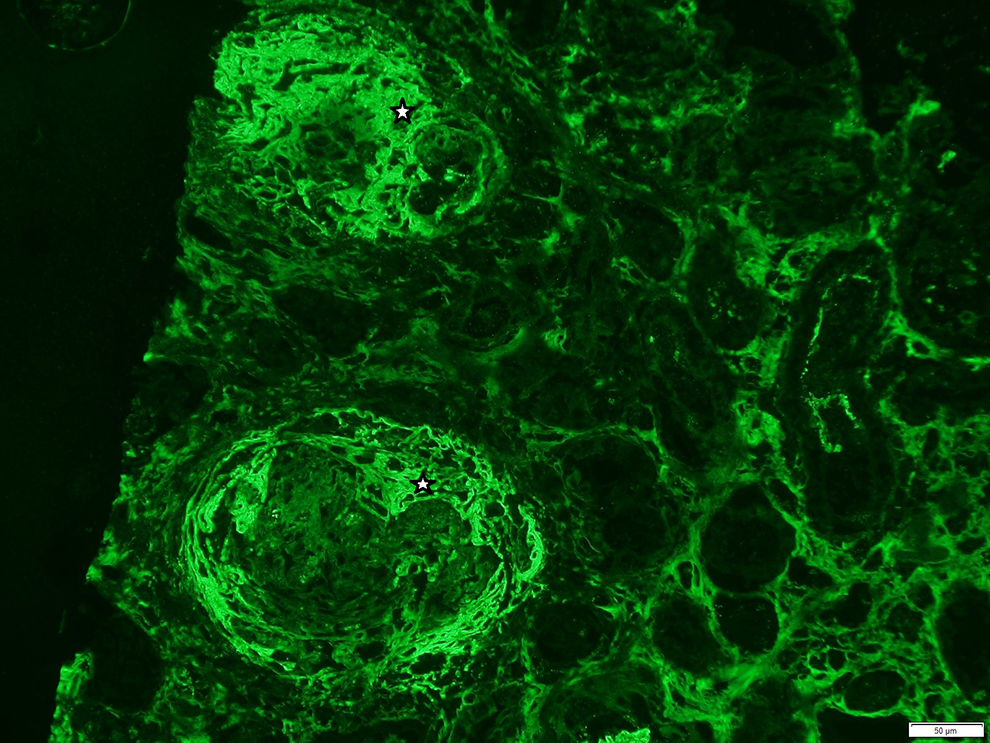
Immunofluorescence stain for fibrin highlights the crescentic and fibrinoid lesions in the 2 glomeruli (Star).
Toluidine blue-stained thick section for electron microscopy reveals all the glomeruli to have cellular crescents with fibrinoid necrosis. There is no ultrastructural evidence of immune complex-mediated electron-dense deposits. There is moderate to severe podocyte epithelial foot process effacement (Figure 6).
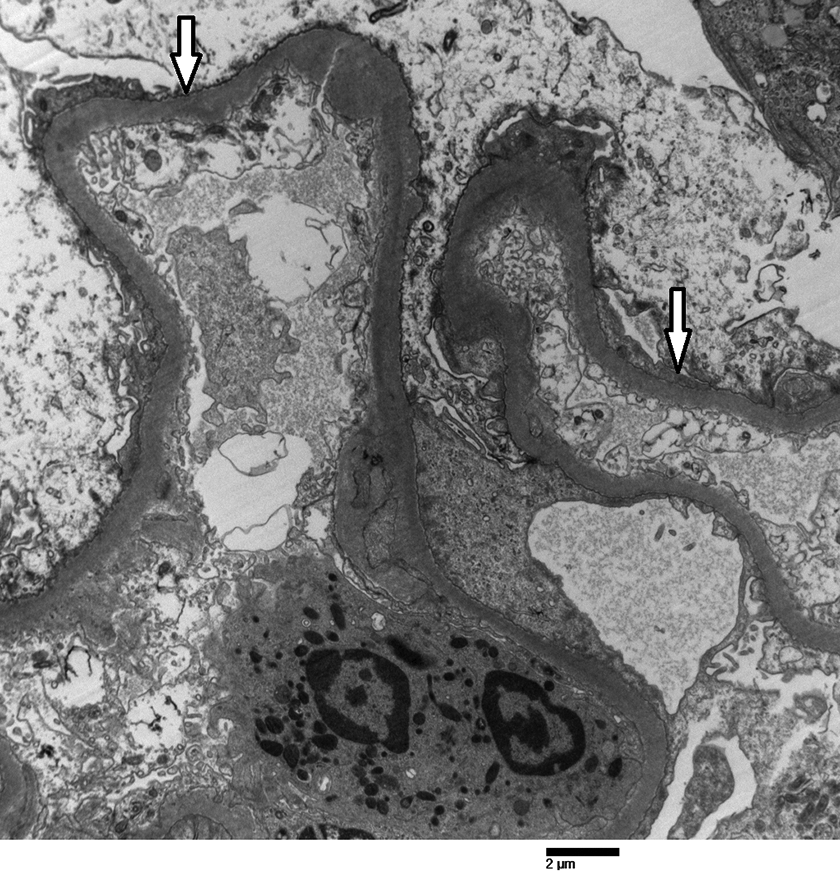
Scanning electron microscopy highlights the capillary loops with foot process effacement (arrows).
Shortly after admission, the patient’s respiratory status acutely worsens, requiring intubation. A bronchoscopy is performed to evaluate the cause of his acute respiratory decline and bronchoalveolar lavage reveals bloody fluid, indicating diffuse alveolar hemorrhage due to acute lung injury.
Questions/Discussion Points, Part 3
Based on the Light Microscopy and Additional Lab Results, What Is the Most Likely Diagnosis?
The lab results were only positive for anti-GBM. Other tests including ANA, dsDNA (excludes lupus), ANCA (excludes Pauci-immune), MPO, PR3, and plasma C3 and C4 complement (excludes C3 GN) were negative or within normal limits.
Based on the laboratory values, the glomerular crescents noted on light microscopy, linear IgG staining of glomerular capillary loops on immunofluorescence, and no immune complex deposits, the most likely diagnosis is anti-GBM disease. Since the patient also presented with pulmonary symptoms, this is best classified as Goodpasture syndrome.
What Is the Pathophysiology of This Disease?
The linear IgG staining by immunofluorescence is diagnostic of Goodpasture syndrome or anti-GBM disease. Anti-GBM disease is a systemic vasculitis that specifically impacts the glomerular and alveolar capillary basement membranes. 6 The autoantibody targets the α3 chain of type IV collagen. The α3 chain is also found in the basement membranes of the choroid plexus, retina, cochlea, and testis. 5,6 The binding of the autoantibody is an example of a type II antigen–antibody reaction where the antibody directly binds to a specific antigen on the cell surface leading to tissue destruction. 3
Goodpasture syndrome classically presents with the triad of pulmonary hemorrhage, rapidly progressive GN (RPGN), and positive anti-GBM antibodies. 5 This trio occurs in 60% to 80% of patients. 3 Renal disease alone is seen in 20% to 40% of patients, and the other 10% present with only pulmonary findings. 3,6 It is to be noted here that RPGN is a clinical term that refers to a progressive nephritic presentation that results in more than 50% loss of renal function in 3 months. Other symptoms include burning with urination, gross hematuria, foamy urine, and edema. High blood pressure is a late disease finding. 3 Lab results most often demonstrate hematuria, mild to moderate proteinuria, and RBC casts. Anti-GBM antibodies can be screened for using ELISAs and confirmed with kidney biopsy. 3,5,7 A chest X-ray may show bilateral and symmetric patchy parenchymal consolidation related to the pulmonary hemorrhage. 3 In this case, pulmonary symptoms are most likely related to the presence of circulating antibodies binding to the alveolar capillary membranes with leakage of blood into the alveolar spaces.
The incidence of Goodpasture syndrome is 0.5 to 1.0 patient per million population. 3,5,6 The age distribution of the disease is bimodal, with the first peak in the second to third decade and the second peak occurring in the sixth to seventh decade. 5,6 Overall, the disease affects men and women equally. 5 No specific trigger for the development of the autoantibody has been identified, but exposure to metallic dust, fumes, tobacco smoke, and respiratory infections has been associated with higher risk. 3
What Are the Treatment Options?
Treatment is based on the severity of symptoms at presentation and the titers of anti-GBM antibodies. Patients with pulmonary and renal symptoms are more likely to get more aggressive therapy because of the higher risk of mortality. 3,6 Titers also have prognostic associations, where higher titers of anti-GBM are associated with worse outcomes because the antibodies are considered directly pathogenic and the presence of any anti-GBM antibodies predisposes to disease development. 5,6 Since the 1970s, the mainstay of treatment has been a combination of immunosuppressive medications including methylprednisolone, cyclophosphamide, and rituximab. Plasma exchange is also beneficial because it directly removes the autoantibodies causing the disease and can rapidly slow down disease progression when compared with immunosuppressive medications alone. 3,6
Diagnostic Findings, Part 4
The patient is diagnosed with Goodpasture syndrome based on the combined renal and pulmonary findings. He is started on high-dose prednisone, cyclophosphamide, rituximab, and plasmapheresis. A total of 10 plasmapheresis procedures are planned, which is the minimum number of procedures recommended for patients with Goodpasture syndrome.
Questions/Discussion Points, Part 4
What Are the Clinical Indications for Using Plasmapheresis in the Setting of Goodpasture Syndrome?
The apheresis guidelines recommend plasmapheresis for patients with creatinine <6.6 mg/dL who are not dialysis dependent because they are the most likely to benefit from the removal of the autoantibodies by the procedure. 8 Patients who have a higher creatinine are less likely to recover renal function in the long term and do not receive as much benefit. The exception is in patients with diffuse alveolar hemorrhage because of the higher mortality risk associated with the severe presentation. 8 Despite this, data suggest that the procedure will only prevent 40% to 45% of patients from either progressing to end-stage renal disease or dying from Goodpasture syndrome. 8 Treatment with plasmapheresis is done everyday or every other day for at least 2 to 3 weeks and may be continued longer depending on the patient’s clinical course. 3,8
What Are the Major Complications and Outcomes Associated With Goodpasture Syndrome?
Recurrence is not usually seen with anti-GBM antibodies. 6 In patients with anti-GBM antibodies alone, the 1-year patient overall survival ranges from 75% to 90% unless they require persistent dialysis and then the survival is lower at 65%. 6 Despite a good overall survival at 1 year, the survival of the kidney is much lower at 20% to 40%. 7 Most patients progress to chronic renal failure requiring dialysis and transplant. Predicting which patients will not recover renal function depends on creatinine at presentation. 6 As well, the kidney biopsy helps predict the recovery of renal function. Patients with a higher percentage of crescents in the glomeruli or who need dialysis at presentation rarely recover renal function and will require dialysis in the long term. 3,6,7
The median survival for patients with Goodpasture syndrome is around 6 years. 3 The risk factors that portend earlier mortality are older age and history of pulmonary hemorrhage. 3
Teaching Points
Acute renal failure is defined by an abrupt rise in creatinine and can be due to prerenal, intrarenal, or postrenal processes.
Nephritic syndrome is characterized by hematuria, variable proteinuria, azotemia, edema, hypertension, and a decline in glomerular filtration rate (GFR).
Nephrotic syndrome is a renal syndrome characterized by >3.5 g proteinuria, hypoalbuminemia, and hyperlipidemia.
A renal biopsy is indicated in the setting of an acute change in GFR for which an underlying etiology has not be elicited.
Crescentic GN is a class of diagnoses that cause nephritic syndrome and are characterized by the formation of crescents in the urinary space of the glomerulus in response to various insults.
There are 3 types of crescentic GN: direct antibody mediated, immune complex mediated, and Pauci-immune mediated.
Common differential diagnosis for crescentic GN includes anti-GBM disease, ANCA-associated vasculitis, and SLE.
Anti-GBM or Goodpasture syndrome is an example of direct antibody-mediated crescentic GN, where antibodies develop and bind to the alpha 3.
Immune complex-mediated crescentic GN is caused by circulating antigen–antibody complexes that deposit in the glomerulus leading to granular immunofluorescence staining.
Immunoglobulin A and lupus nephritis are examples of immune complex-mediated GN.
Pauci-immune-mediated crescentic GN is glomerular injury due to circulating antibodies that do not deposit in the glomerulus and leads to negative immunofluorescence staining by IgG, IgA, IgM, C3, and C1q.
Antineutrophil cytoplasmic antibody–associated vasculitis including GPA and MPA are examples of Pauci-immune-mediated crescentic GN.
Necrotizing GN with fibrin deposition can be highlighted by fibrinogen immunofluorescent staining.
Pulmonary renal syndromes are multisystem diseases where antibodies impact both organ systems. Examples include anti-GBM disease, ANCA-associated vasculitis, and SLE.
Anti-GBM titers correlate with disease severity and response to treatment.
The mainstay of treatment for anti-GBM disease is plasmapheresis in combination with immunosuppressive medications.
Median survival for Goodpasture Syndrome is around 6 years, but renal survival is usually less than 1 year.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
