Abstract
Background:
Malignant pleural mesothelioma (MPM) is a rapidly fatal tumor of increasing worldwide incidence. Diagnosis of MPM may be difficult and is an area of academic and clinical interest. Diagnosis by effusion cytology is controversial, and the clinical utility of soluble biomarkers, particularly mesothelin, remains unexplored in the difficult to diagnose patient.
Objectives:
We studied the diagnostic value of measuring mesothelin in patients with cytology-negative pleural effusion undergoing medical thoracoscopy.
Methods:
Patients were enrolled prospectively and underwent medical thoracoscopy. Biopsy samples were reviewed following standard care guidelines. Soluble mesothelin was quantitated by commercial enzyme-linked immunosorbent assay in effusion and serum samples collected at the time of thoracoscopy. Patients were followed up until death or for a median of 28 (interquartile range 18-43) months.
Results:
Biopsy for thoracoscopy was definitive for malignancy in 17 of the 36 cases (16 MPM and 1 lung adenocarcinoma) and suspicious of malignancy in 6 cases. A final diagnosis of malignancy was made for 27 cases (24 MPM, 2 lung adenocarcinoma, and 1 primary pleural T-cell lymphoma). At thoracoscopy, soluble mesothelin was elevated in the effusion of 13 cases all of which were MPM. Serum mesothelin was elevated in 6 cases with MPM. One case with elevated mesothelin on whom thoracoscopic biopsy demonstrated a pleural plaque was diagnosed with metastatic MPM 9 months later.
Conclusion:
Elevated effusion mesothelin had a positive predictive value of 100% for a final diagnosis of MPM. Elevated mesothelin, in effusion or serum, should raise the index of suspicion of malignancy and warrants continued clinical follow-up.
Introduction
Malignant pleural mesothelioma (MPM) is an incurable cancer with a median survival of less than 12 months. 1 Malignant pleural mesothelioma is a significant concern in Europe and Australasia, and its incidence continues to grow worldwide. The unregulated use of asbestos in developing countries predicts a global rise in MPM in the coming decades. 2
Establishing the diagnosis of MPM is notoriously difficult and often requires multiple pleural procedures. Cytology analyses of cells from pleural effusion have been reported to have a relative low yield at distinguishing mesothelioma from benign mesothelial cells. 3 No pathognomonic radiological features exist for mesothelioma, and image-guided biopsies can be hampered by difficulties in distinguishing mesothelioma changes from benign pleural thickening. Even at thoracoscopy or thoracostomy when pleural tissue is sampled under direct visual guidance, false-negative biopsies are well recognized to occur because of patchy tumor development in the pleura. 4 –6 There is thus an eager search for a biomarker that can aid the diagnosis of MPM. Several molecules have shown promise. Of these, mesothelin is the most studied and is approved by the US Food and Drug Administration for use in MPM. 7
A large number of studies have focussed on the use of mesothelin as a sole diagnostic tool and found that it lacks the sensitivity and specificity to be a stand-alone diagnostic test. 8 Both serum and pleural fluid mesothelin levels are elevated in up to 67% of cases with MPM 9 but can occasionally be positive in other cancers, including ovarian and pancreatic. Mesothelin is not elevated in 50% of patients at presentation and is seldom raised in the sarcomatoid subtype of MPM. 10
The role of mesothelin as an adjunct biomarker to add to existing radiological, cytological, and histological modalities has seldom been explored. Davies et al investigated the additional value of measuring mesothelin levels in pleural fluid when the latter is sampled for cytological examination. The study found that in patients with MPM who were cytology positive, a raised mesothelin level could serve to confirm the cytologic diagnosis. In cases where cytology was negative but the mesothelin level was raised, most turned out to have a pleural malignancy (most commonly MPM). In those patients whose fluid was suspicious but not diagnostic of MPM, a raised mesothelin level was 100% correct in defining an MPM. All 5 patients in this category were eventually diagnosed to have MPM. 11 Subsequent international guidelines have highlighted the role of mesothelin measurements in patients suspicious of mesothelioma, particularly if the cytology was equivocal or suspicious (though not definitive) of MPM. 12
When a patient presents with an undiagnosed pleural effusion, thoracentesis is generally the first intervention performed. If the fluid sample failed to capture any cancer cells and no alternative etiology for the effusion is found, thoracoscopy (medical or surgical) with biopsy is recommended. In 3 series of medical thoracoscopy, false-negative rates in defining a malignant cause ranged between 5% and 15%, and the majority of those cases turned out to be MPM. 4 –6 No quality data exist to guide care for these patients: The need for further biopsy, thoracotomy, or observation only (if so, for how long) remains unclear. A test to help identify high-risk patients for further investigation/biopsy will be valuable. We hypothesized that the measurement of mesothelin in these patients, cytology negative for malignancy undergoing medical thoracoscopy, may help to guide clinical care.
Methods
Patients who underwent medical thoracoscopy for evaluation of an undiagnosed pleural effusion were prospectively included. Patients who had an established cancer diagnosis from pleural fluid cytologic examination or other tests were excluded. The study was approved by the Sir Charles Gairdner Human Research Ethics Committee, and all patients provided informed written consent. Patient demographics, asbestos exposure history, and clinical data were collected.
Pleural biopsy tissue and pleural fluid were collected at medical thoracoscopy and processed, per standard care, at the PathWest Laboratory (Queen Elizabeth II Medical Centre, Perth, Western Australia, Australia) and reviewed by experienced pleural pathologists. In addition, mesothelin was quantified in the sera and pleural fluid. Briefly, pleural fluids were collected at medical thoracoscopy in 500-mL Techono-Plas sterile containers (St Marys, South Australia, Australia) and then centrifuged for 10 minutes at 400
Soluble mesothelin concentrations were determined following the manufacturer’s instructions using a double determinant enzyme-linked immunosorbent assay (Mesomark; Fujirebio Diagnostics, Malvern, Pennsylvania). Mesothelin concentrations were determined from a standard curve performed on each plate and expressed as nmol/L. As previously established, a threshold value of 20 nmol/L for pleural effusion 9 and >2.5 nmol/L for serum mesothelin 13 were used. Samples below the limit of detection were assigned a value of 0.3 nmol/L for statistical purposes.
Descriptive statistical analyses were performed using GraphPad Prism (San Diego, California) and expressed as median (interquartile range [IQR]) or mean (±standard deviation [SD]) unless otherwise stated. Patient survival was calculated from the time of thoracoscopy and analyzed using the Kaplan-Meier product limit procedure with patients dichotomized into groups based on whether effusion or serum mesothelin levels were above the predefined cutoff thresholds and median survival compared using the log-rank test.
Results
Patient Characteristics
Of the 37 patients prospectively enrolled in the study over a 4-year period, the majority were men (n = 33), and the mean age was 70 years (SD 9). Detailed asbestos exposure history was available in 32 patients, and 81% reported previous asbestos exposure. One participant withdrew from the study following negative biopsy and was excluded from further analysis.
Most patients (n = 33) had at least 1 pleural fluid cytologic analysis performed at a median of 15 (IQR 7-24) days before medical thoracoscopy. Most (n = 23, 70%) were reported as negative for malignant cells, though 10 (30%) were reported to have atypical cells or cells suspicious (but not definitive) of malignancy. In the remaining 3 patients, no prior thoracenteses were performed.
A final diagnosis of malignancy was made in 27 cases (24 MPM, 2 lung adenocarcinoma, and 1 primary pleural T-cell lymphoma). Diagnosis was made based on biopsies performed at thoracoscopy, subsequent biopsy, or a combination of biopsy result together with clinicoradiological impression. The remaining 9 patients had final diagnoses considered benign (see subsequently).
Effusion Mesothelin at Thoracoscopy
Pleural effusion mesothelin was above 20 nmol/L in 13 patients (range 22-140), all of whom were diagnosed with MPM on histology, providing a positive predictive value of 100% (Figure 1). Of the 13 patients, 11 had mesothelioma on thoracoscopy tissue biopsy (8 had epitheloid, 1 sarcomatoid, and 2 biphasic MPM). In the remaining 2 cases, 1 had changes “suspicious” (but not definitive) of MPM. Computed tomography-guided biopsy 20 days postthoracoscopy confirmed desmoplastic MPM. In the second case, thoracoscopic biopsies showed pleural fibrosis/plaque only; subsequently, a diagnosis of MPM was established on a Positron Emission Tomography (PET)-avid lymph node biopsy after 9 months of follow-up. Pleural effusion mesothelin was below 20 nmol/L in 23 cases (Figure 1): 18 were true negatives (17 benign and 1 adenocarcinoma) but 5 had MPM (3 epitheloid, 1 sarcomatoid, and 1 biphasic) at thoracoscopy.

Clinical outcome of patients with a high preprocedural index of suspicion of malignancy and nondiagnostic effusion cytology undergoing medical thoracoscopy dichotomized by effusion mesothelin result.
Serum Mesothelin at Thoracoscopy
Serum mesothelin was elevated above 2.5 nmol/L in 7 of the 33 patients who provided serum samples (range 2.5-7.6 nmol/L): all were diagnosed with MPM. In 6 of these 7 patients, both the serum and the pleural fluid mesothelin levels were raised. In the remaining patient, the pleural fluid mesothelin concentration (17.9 nmol/L) was marginally below the cutoff of 20 nmol/L (Figure 2).

Clinical outcome of patients with a high preprocedural index of suspicion of malignancy and nondiagnostic effusion cytology undergoing medical thoracoscopy dichotomized by serum mesothelin result.
Cases With MPM
A final diagnosis of MPM was made in 24 patients (14 epitheloid, 3 sarcomatoid, 3 biphasic, 1 desmoplastic, and 3 not specified). Their median pleural fluid mesothelin concentration was 23.7 nmol/L (IQR 11.7–52.4). In all, 13 (54%) patients had a mesothelin concentration above the 20 nmol/L threshold; in 11 of them, the thoracoscopic biopsy was diagnostic for MPM (Figure 4). Serum mesothelin was measured in 23 of the 24 patients, all of whom had normal creatinine levels reported. The median mesothelin concentration was 1.2 nmol/L (IQR 0.3-2.8), with 7 of these 23 cases with levels above the preset threshold of 2.5 nmol/L (Figure 5).
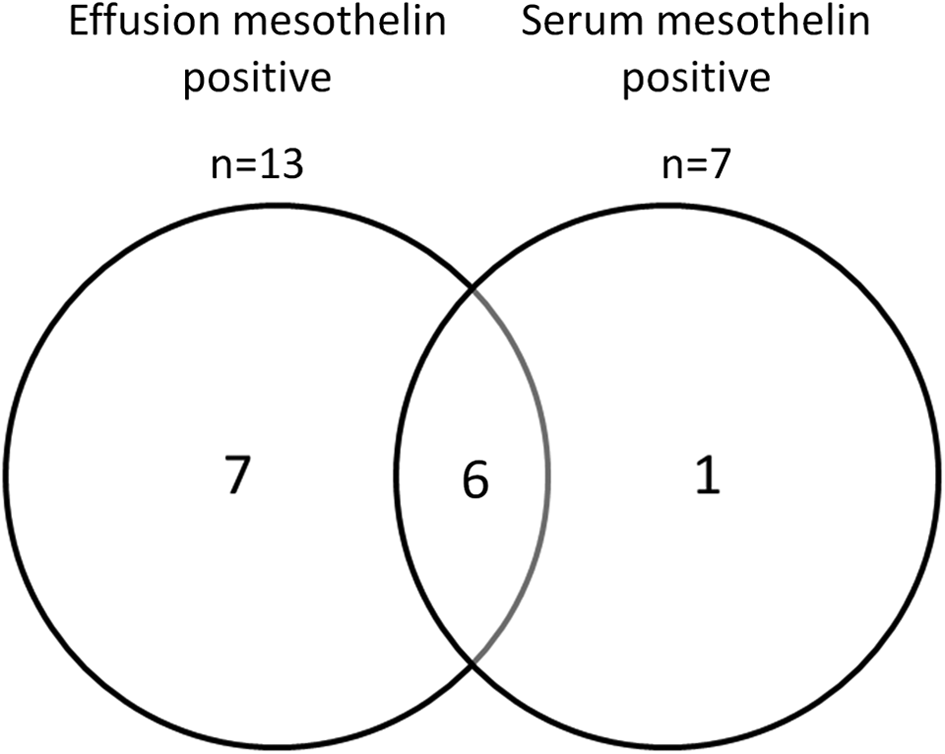
Venn diagram showing the number of cases with elevated mesothelin in the serum and the effusion.

Soluble mesothelin concentrations in effusions collected at thoracoscopy presented relative to final patient diagnosis. Black diamonds—thoracoscopy results as indicated, gray diamonds—thoracoscopy results suspicious of malignancy, and clear diamonds—thoracoscopy results nonmalignant.

Serum soluble mesothelin concentrations in samples collected at thoracoscopy presented relative to final patient diagnosis. Black diamonds—thoracoscopy results as indicated, gray diamonds—thoracoscopy results suspicious of malignancy, and clear diamonds—thoracoscopy results nonmalignant.
At the time of data analysis, 16 patients with MPM had died and 8 patients remained alive. The median survival duration of the patients who died after diagnosis was 7.5 (IQR 4.5-12.9) months. The patients surviving at the end of study had been followed for a median duration of 18.6 (range 2-24) months. There was no difference in median survival of patients with MPM based on mesothelin level at the time of thoracoscopy (Figure 6).

Survival of patients with malignant pleural mesothelioma (MPM) segregated on pleural effusion mesothelin concentration above or below the 20 nmol/L threshold.
Non-MPM Malignant Cases
Three patients had pleural malignancy not due to MPM: 2 lung adenocarcinomas and 1 primary pleural lymphoma. The pleural fluid mesothelin levels were all below the 20 nmol/L cutoff for MPM at 18.6, 10.5, and 9.5 nmol/L, respectively. Serum mesothelin levels were available from 2 patients; again neither exceeded the serum cutoff of 2.5 nmol/L (at 1.6 and 0.58 nmol/L).
Cases of Benign Etiology
Nine patients had a benign effusion. Pleural effusion mesothelin concentrations in this group of patients were all below the threshold of 20 nmol/L. Serum mesothelin was performed in 7 of these 9 cases which were all below 2.5 nmol/L. Of the 9 patients, 2 had died at time of censor, 1 from heart failure and 1 from pneumonia, neither had developed cancer before death. The other 7 patients were followed up for a mean of 30 (SD 15.2) months with no evidence of pleural cancer.
Discussion
This study is the first to evaluate the use of mesothelin as an adjunct diagnostic test in patients undergoing thoracoscopy for the investigation of an undiagnosed pleural effusion. Raised mesothelin levels predicted, with high accuracy, the eventual diagnosis of MPM even in cases where thoracoscopic biopsy failed to provide a conclusive finding. A negative mesothelin level however did not exclude MPM. Pleural fluid mesothelin was more sensitive than its measurement in serum.
Diagnosis of MPM continues to be challenging. Most patients present in advance stages with symptomatic pleural effusions. Pleural aspiration, a simple procedure that neither requires sedation nor requires extensive monitoring, is commonly the first diagnostic test performed. In prospective studies of patients presenting with a new pleural effusion, mesothelin has a sensitivity of approximately 70% at a specificity of greater than 90%. 9,11,14,15 This has led to mesothelin being advocated to be an adjunct diagnostic test in clinical guidelines. 12
Although specialist units have reported high (˜70%) diagnostic returns of MPM from pleural fluid cytology, 16 most centers found a much lower yield. 11,17 This is in part because the cytologic appearances of benign, reactive, and malignant mesothelial cells are similar. Most pathologists prefer to define MPM from tissue biopsies showing evidence of invasion of deeper structures by mesothelioma cells. Thoracoscopy is advocated as the procedure of choice in international guidelines in the workup of undiagnosed, cytology-negative pleural effusions. 18 Thoracoscopy however is an invasive procedure, and it is not uncommon to come across cases whereby thoracoscopy is considered too high a risk and therefore deemed an unsuitable intervention for an individual. This is particularly true in patients with significant underlying comorbidities and/or frailty—a patient profile that occurs not infrequently. Furthermore, thoracoscopic biopsies have a reported false-negative rate of ∼10%. 4 –6
This current study is the first to evaluate the role of mesothelin in this stage of the diagnostic algorithm. The patients in this study were representative of cases with MPM having increased difficulty for diagnosis who had in the majority of cases already undergone nondiagnostic procedures. In this setting, a positive mesothelin test, either from pleural fluid or serum, strongly predicted pleural malignancy, especially MPM. The test was particularly valuable in patients whose biopsy was inconclusive or negative; a positive mesothelin level should raise concerns that the negative biopsy was due to sampling error. In such cases further investigations, such as repeat thoracoscopy or imaging-guided biopsy, or at least close surveillance, are needed. Clinicians and patients with an elevated mesothelin level should not be reassured, even if thoracoscopy did not reveal evidence of malignancy.
Mesothelin can be measured in pleural fluid and/or blood. Mesothelin is elaborated by mesothelioma cells into the pleural cavity and then absorbed into the systemic circulation. Pleural fluid mesothelin concentrations in MPM are therefore usually 10- to 20-fold higher than in corresponding serum. It is therefore not surprising that most mesothelin-positive cases were captured in pleural fluid assessments. 9,19 However, like other studies, in a minority of patients, the serum but not the pleural fluid level of mesothelin reached the diagnostic threshold.
A negative serum or pleural fluid mesothelin level is of little clinical value, as false negatives can occur. Only approximately 50% of patients with MPM have a positive mesothelin level at diagnosis, and sarcomatoid MPM often have low expression of mesothelin. However, estimates of diagnostic accuracy can be confounded because of the presence of antecedent symptoms associated with benign asbestos-related conditions. For example, in the current study, in 1 case with concurrent asbestosis and pleural plaques at the time of thoracoscopy biopsy, a diagnosis of MPM was established 3 years later. Active ongoing work focuses on improving mesothelin diagnostic sensitivity by adjusting for renal excretion or combining with additional biomarkers.
This study was performed in a center with extensive experience in cytological diagnosis, 16 which limited the size of the cohort available for study. Furthermore, the study was performed in an area with a relative high incidence of MPM, thus application of the findings to nonendemic areas needs validation.
In summary, our data showed that measuring mesothelin in pleural fluid (and serum) can contribute to the workup of a pleural effusion in patients undergoing thoracoscopy, especially if the biopsy did not provide a definitive diagnosis of the effusion. This is reflective of a real-life clinical practice, where biomarkers would be used in conjunction with clinical, radiological, and histocytological data. Future studies on biomarkers of MPM should continue to define and optimize their role in clinical management pathway.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
