Abstract
Medical thoracoscopy (MT) is a procedure that involves access to the pleural space with an endoscope allowing direct visualization of the pleural space and intrathoracic structures while aiding in obtaining tissue or performing interventions under direct visual guidance. This article reviews the technique, applications, and complications of MT.
Introduction
Pleural effusions are frequently encountered conditions in pulmonary medicine. Approximately 1.5 million new pleural effusions are diagnosed and around 180 000 thoracentesis are performed annually in the United States alone. 1,2 Thoracentesis provides a definitive or presumptive diagnosis in 59% to 73% of cases, and additional diagnostic investigations are often needed. 3,4 Thoracoscopy is a procedure that involves access to the pleural space with an endoscope and allows direct visualization of the pleural space and intrathoracic structures and aids in obtaining tissue or performing interventions under direct visual guidance. Thoracoscopy is broadly divided into minimally invasive medical thoracoscopy (MT) and surgical thoracoscopy, also called “video-assisted thoracic surgery” (VATS). The recent rapid growth in the field of interventional pulmonology along with the evolution of sedation techniques and development of flex–rigid thoracoscopes has brought MT to the forefront. Several centers have opened “pleural disease” programs, and MT is a key component of the interventional pulmonology programs. 5,6
History of MT
Thoracoscopy was documented in a report dated 1866 when Richard Cruise examined the pleural space in a girl with empyema. The thoracoscopy was officially introduced in 1910 by a Swedish internist, Hans Christian Jacobaeus, who used a cystoscope to evaluate the pleural space. From 1910 to the 1930s, Jacobaeus provided extensive descriptions of thoracoscopy technique. For that reason, he is recognized as the father of modern thoracoscopy. 7 Accordingly, this technique was initially named after Jacobaeus as “Jacobaeus operation.” 8 Since 1923, “pleuroscopy” was the most commonly used definition, and in later years, the term “medical thoracoscopy” has been used.
In the preantibiotic era, MT became central in the treatment of pulmonary tuberculosis to break down pleural adhesions as therapeutic management for lung collapse. With the discovery of streptomycin by Waksman and Schatz in 1943, the main indication for MT became obsolete and its use rapidly declined. In the 1970s, physicians, such as Brandt and Loddenkemper in Germany and Boutin in France, revived the use of MT primarily to provide quality specimens for the diagnosis of unexplained exudative pleural effusions. Over the past 2 decades, the interest in MT has increased as the incidence of pleural diseases has continued to rise. The first commercially available flexible–rigid (or “flex–rigid”) thoracoscope was developed in the late 1990s as an alternative to the traditional rigid instruments. These thoracoscopes offer controls similar to those of flexible bronchoscopes offering better maneuverability and improved visualization of the rigid thoracoscopes. 9
Technique of MT
Preprocedural Checklist
Medical history and physical examination
Medication review to ensure anticoagulants or antiplatelet agents has been appropriately held before the procedure. In patients with normal renal function, anticoagulation fully resolves usually after 5 half-lives (Table 1). 10
Basic laboratory studies: prothrombin time/international normalized ratio, platelets, blood urea nitrogen, and creatinine (Table 2).
Chest imaging (chest X-ray or computed tomography of the chest).
Focused pleural ultrasound examination to assess the volume and characteristics of the pleural effusion and to mark the site for pleural space entry.
Multidisciplinary review of cases by pulmonologists and thoracic surgeons.
Informed written consent.
Preoperative fasting (6 hours).
Duration of Anticoagulant Medications Withhold Before Procedure.

Laboratory Cutoff to Minimize Bleeding.

Abbreviations: BUN, blood urea nitrogen; INR, international normalized ratio.
Staff, Location, and Equipment
The procedure should only be undertaken by a properly trained interventional pulmonologist and thoracic surgeons. A minimum of 2 other suitably trained health-care personnel are required for assistance, monitoring, and administration of the intravenous (IV) sedation. The procedure room must have pulse oximetry, noninvasive blood pressure recording, electrocardiogram monitoring, resuscitation equipment, oxygen source, height-adjustable operating table, suction equipment, trolley to hold instruments, and facilities to display patient’s radiographs.
Rigid Versus Flex–Rigid Thoracoscope
Rigid thoracoscopy has been the traditional method to perform MT. Rigid thoracoscopy provides larger biopsy samples, which typically provides better samples than those provided by flex–rigid thoracoscope (Figure 1). However, with limited available data, flex–rigid thoracoscopy has been found to have similar safety profile and diagnostic yield. 11 Table 3 compares rigid versus flex–rigid thoracoscopy.

Flex–rigid thoracoscope and 11-mm Outer Diameter (OD) trocar.
Rigid Versus Flex–Rigid Medical Thoracoscopy.
The rigid thoracoscopy consists of various equipment including trocar, telescope, light source, and biopsy forceps. Trocars usually range in size from 5 to 13 mm in external diameter and are made from single-use disposable plastic or stainless steel. The rigid telescope is made of stainless steel and is 27 to 31 cm in length with a diameter of 7 to 12 mm. The rigid telescope can be straight (0°) or oblique (typically 30° and 50°) angle of vision. A cold (xenon) light source with camera is attached to the eyepiece of the telescope. Mini thoracoscopy refers to the use of rigid thoracoscope with small diameters of 2 to 3 mm. These thoracoscope may have similar diagnostic yield to the larger instruments but require a second port of entry for the biopsies. 9,12
The flex–rigid thoracoscope has similar handset as that of the flexible bronchoscopes. This handset design facilitates its adoption by pulmonologists already familiar with the operation of flexible bronchoscopes. The flex–rigid thoracoscope has a distal tip that can be flexed and extended (160° and 130°), respectively. These are introduced through 8- to 10-mm outer diameter plastic trocar. Biopsies are performed via instruments introduced through the 2.4- or 2.8-mm working channel. One of the most commonly used autoclavable flex–rigid model (Olympus LTF 160 or 240, Tokyo, Japan) has a 7 mm outer diameter, 22 cm proximal rigid shaft, and 5 cm flexible distal tip.
Patient Position, Preparation, and Procedural Monitoring
The patient is placed in the lateral decubitus position with the affected side up and the arm above the head. An alternative upper limb position used in Europe is to have the uppermost arm elevated in a “sling.” A pillow or roll, placed under the patient’s lateral thorax at a level just below the axilla, helps to spread the contralateral ribs, making it easier to insert the trocar and cannula and minimizing discomfort during manipulation of the thoracoscope. The procedure is typically performed with the operator facing the patient to allow optimal exposure of the posterior parietal pleura. Laterality should be confirmed and marked prior to the procedure. This can be done by rechecking a recent chest radiograph and performing a bedside pleural ultrasonographic examination immediately before commencing the procedure. We use cefazolin 1 g IV 30 to 60 minutes before pleuroscopy for surgical infection prophylaxis.
The patient should be monitored throughout the procedure with pulse oximetry, noninvasive blood pressure measurements, and electrocardiogram tracing. Capnometer can be utilized to monitor for hypoventilation, if available. All the operative team members must clean their hands with a standard surgical scrub technique and then put on a sterile gown and gloves. The skin over the entire hemithorax of interest including the axilla should be prepared with an alcohol-based skin-sterilizing solution. A sterile drape should be placed over the patient, leaving a small exposed area through which the examination is performed. A second application of alcohol-based skin-sterilizing solution should then be applied to the operative area and allowed to dry.
Analgesia and Sedation
Medical thoracoscopy, compared to VATS, is commonly performed under moderate sedation in addition to local anesthesia. Sedation induced by using rapidly acting, IV benzodiazepine (eg, midazolam), administered with a dose of 1 to 5 mg during the procedure. Intravenous analgesic agent such as fentanyl 25 to 100 μg can be used to titrate according to the patient’s pain scale following local anesthesia application. 13
The procedure can be performed under monitored anesthesia care with IV propofol with a faster onset of action and a rapid recovery compared to conscious sedation. General anesthesia may be preferable in some special indications such as idiosyncratic or allergic sensitivities to local anesthetics and in anxious or uncooperative patients, including children.
Local anesthesia has to be administered carefully from skin, subcutaneous tissue, intercostal muscle down to the parietal pleura, and at the caudal rim of the upper and the cranial rim of the lower rib to anesthetize the intercostal nerve as well as the periosteum of the ribs. Local anesthesia is induced using up to 20 mL of lidocaine 0.5% to 1%. The dose of infiltrated lidocaine should not exceed 3 mg/kg body weight to avoid toxicity. 14 Lidocaine combined with 1 in 100 000 epinephrine may aid visualization by preventing blood from oozing onto the thoracoscope tip during the procedure, although there is no specific evidence to support its use.
Chest Ports/Site of Entry/Insertion of Trocar and Introducer
The recommended site of chest entry is the fourth or fifth intercostal space in the midaxillary line, within the “safe triangle” (Figure 2). The area lies above a line horizontal to the nipple in male, which is bordered by latissimus dorsi posteriorly and pectoralis major anteriorly. 13 Thoracic ultrasound is used immediately prior to the procedure to identify the safest and most appropriate site for trocar insertion, avoiding areas of lung adhesion to the chest wall (Figure 3).

Safe triangle for the recommended site of chest entry.
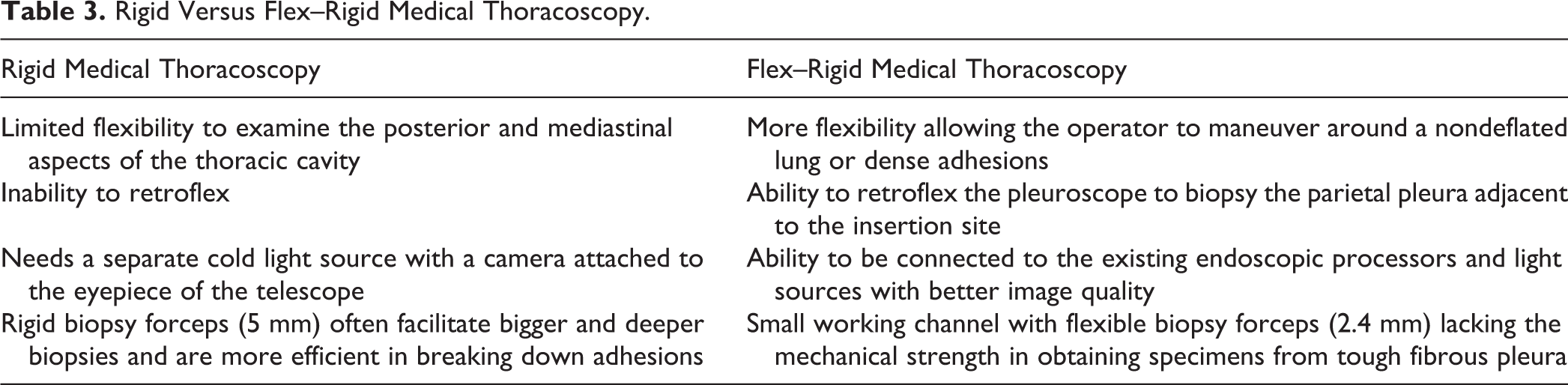
Transthoracic ultrasound view of a patient with malignant pleural effusion demonstrating the liver on the right, consolidated lung on the left, and plural effusion with septations.
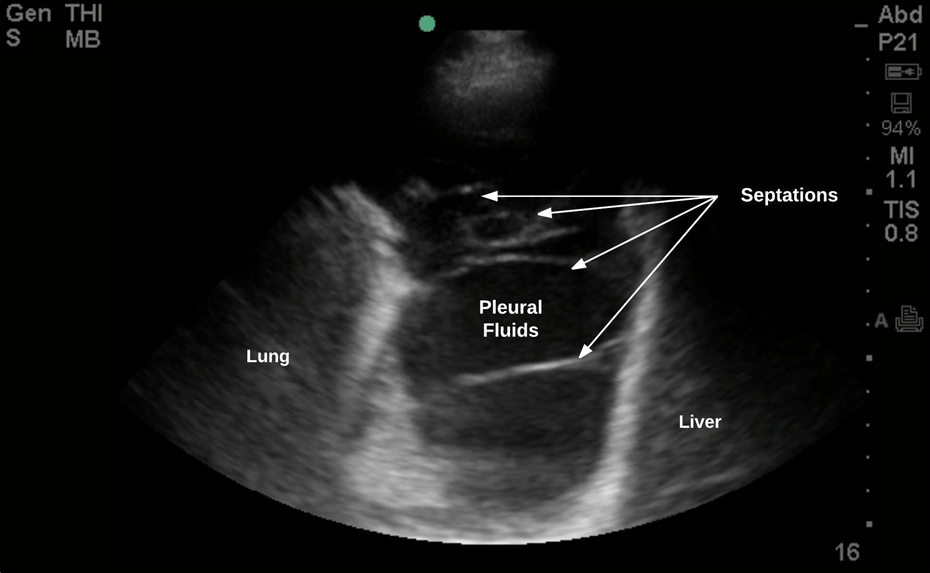
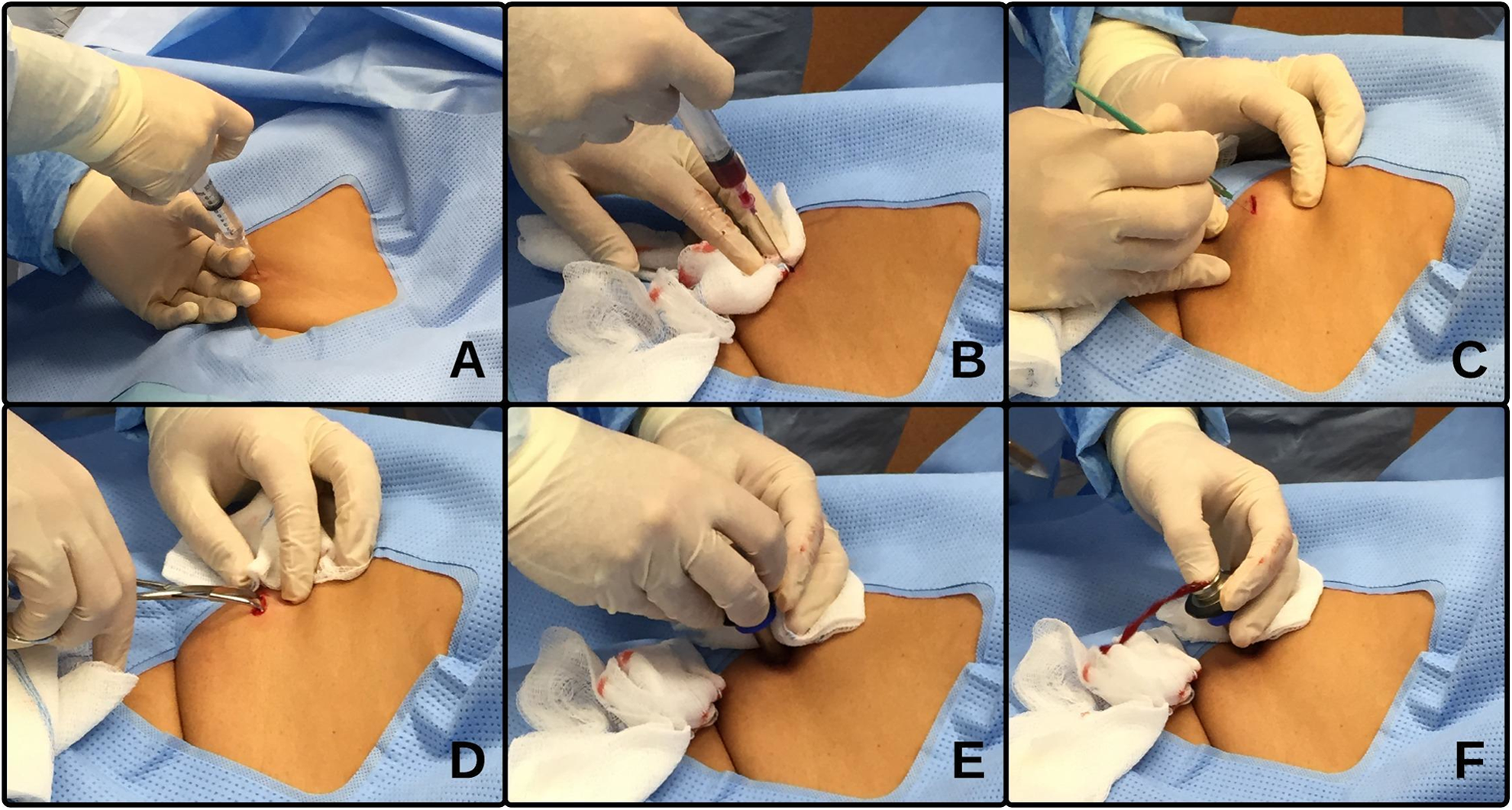
An intradermal anesthetic bleb should initially be raised, and the intercostal muscles and parietal pleura then infiltrated with local anesthetic (Figure 4A). 15 In cases of pleural effusion, an initial aspiration of pleural fluid with a needle should be confirmed (Figure 4B). About a 1-cm horizontal incision should be made with the scalpel through the skin and subcutaneous tissue, appropriate to the size of the trocar to be used (Figure 4C). The incision should be in the middle of the selected intercostal space following the orientation of ribs. Blunt dissection with blunt dissecting forceps should then be performed through the chest wall and into the pleural cavity (Figure 4D). After the parietal pleura has been penetrated, it should be dissected to create a small window for trocar entry. The 7-mm trocar/introducer port should then be inserted into the pleural space via corkscrew motion (Figure 4E). The cannula should lie about 1 to 3 cm within the pleural space and should be held well in position (Figure 4F). The thoracoscope is then placed in the cannula and advanced into the pleural cavity under direct vision through the trocar (Figure 5). If necessary, the pleural fluid is removed with a suction catheter. During aspiration of a large amount of pleural effusion, care must be taken to let air pass freely into the thorax down the sides of the suction catheter. This maneuver creates a pneumothorax to prevent lung reexpansion by maintaining normal intrapleural pressure.
The side of the pleural effusion identified with thoracic ultrasound and marked (A) infiltration of skin, subcutaneous tissue, intercostal muscle, and parietal pleura with local anesthetic; (B) aspiration of pleural fluid with a needle confirming pleural effusion; (C) making the incision with the scalpel through the skin and subcutaneous tissue, appropriate to the size of the trocar to be used; (D) blunt dissection with blunt dissecting forceps through the chest wall and into the pleural cavity; (E) insertion of trocar/introducer port into the pleural space via corkscrew motion; and (F) advancement of thoracoscope into the pleural cavity under direct vision through the trocar.
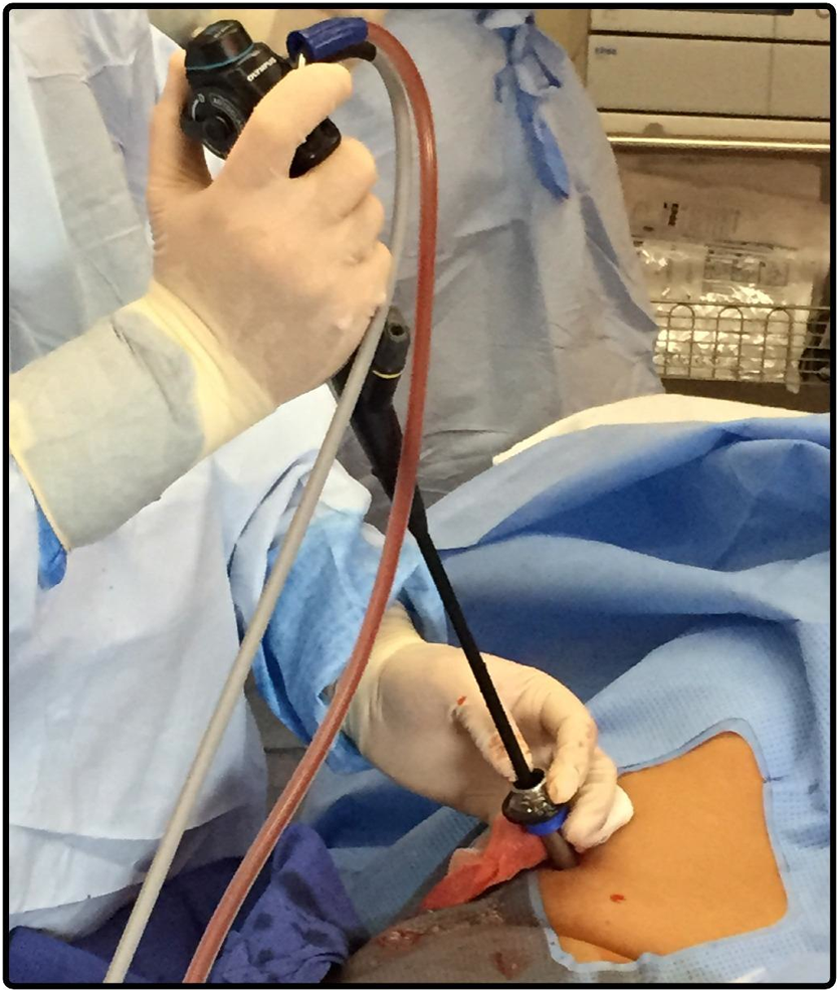
Thoracoscope advanced into the pleural cavity under direct vision through the trocar.
Second Entry Port
A single-port access is usually sufficient for diagnostic pleuroscopy, pleural biopsy, and talc poudrage using a flex–rigid pleuroscope. 9,12 A second port might be necessary to drain complex loculated fluid collections, to facilitate adhesiolysis, to obtain larger pleural biopsies, for “pinch” lung biopsy, for sampling abnormal lesions around the first entry site, and to control bleeding. Similarly, double-port access may be necessary to evaluate the pleural space completely when the rigid telescope is used, especially if the posterior and mediastinal aspects of the hemithorax are inaccessible because of partial collapse of lung or when the lung parenchyma is adherent to the chest wall. Some operators routinely use a second entry port as it improves visualization of the pleural space. The second port should be in line with the first and should be separated from it by about 2 intercostal spaces. A 5-mm trocar is typically used for the second port site as the flex–rigid scope has a 7-mm outer diameter.
Induction of Pneumothorax
Safe thoracoscopy requires a large pleural cavity between the lung and the chest wall. In cases where there is little pleural fluid, a pneumothorax should be induced. This may be performed with the Boutin needle. The sharp “cutting” outer needle should be used to penetrate through most of the chest wall, except the pleura, and the blunt inner needle should then be used to puncture the pleura. The blunt inner needle is then withdrawn and air should be heard to hiss into the pleural cavity. The placement of needle in pleural space is confirmed either by listening or, more reliably, by the use of a pressure manometer. The potential complication of this procedure is bleeding, and the risk is considerably higher in cases of small pleural space. Hence, MT should only be performed by an experienced physician where induction of a pneumothorax is necessary and in patients with good lung function that can compensate for the loss of lung volume caused by the pneumothorax.
Visualization of the Pleural Space
The pleural cavity can be inspected directly or indirectly by video through the thoracoscope. The exploration of the pleural space should be performed systematically maneuvering the thoracoscope. One method is to start at the apex and successively examine the costal pleura, the diaphragm, and the mediastinal pleura to end back at the apex. Adequate assessment of the diaphragmatic pleura and costodiaphragmatic recesses is particularly important. Photographic camera or video recorder should be utilized to record any significant macroscopic pleural changes.
For the rigid pleuroscope, oblique telescopes are valuable in difficult cases to ensure adequate pleural inspection. Usually, 50° oblique angle viewing thoracoscope is utilized for the initial visualization of the pleural cavity. It should be rotated systematically for the proper exploration of the pleural space. Subsequently, a straight viewing (0°) telescope should be introduced for more careful examination and for planning biopsy sites. Complete examination of the pleural cavity can sometimes be obstructed (or complicated) by fine spider web-like adhesions, which can be mechanically separated. Fibrous bands or vascular adhesions should be avoided but if necessary can be cauterized by electrocautery.
Pleural Biopsy
Suspicious areas are biopsied through the working channel of the thoracoscope under direct vision using the optical biopsy forceps. The optimal number of biopsies needed is unclear, but typically 2 to 6 biopsies of a suspicious pleural lesion are necessary to establish the diagnosis. When the suspicion of malignancy is high but no obvious tumor is seen, it is recommended to take biopsies from multiple sites where the pleura appears abnormal. Sufficient quantities of tissue must be obtained especially if hormonal receptor studies are required for tumors such as carcinoma of the breast, however, repeated biopsies from the same site should be avoided. Pleural biopsies should only be taken from the parietal pleura to avoid the risk of prolonged air leak. Biopsy should be performed over a rib, which can be touched and felt with biopsy forceps to avoid the neurovascular bundle (Figure 6). A lateral “lift and peel” technique can be utilized to obtain large, 1 to 2 cm biopsies that cleave along the subpleural plane avoiding the blood vessels. In this technique, the edge of the pleural surface is lifted and gently pulled sideways to obtain biopsies. Pleural biopsy may be painful even when the patient is adequately sedated. There is a lack of strong evidence regarding the IV administration of short-acting potent analgesic prior to parietal pleural biopsy but may be considered. Biopsies should be placed in formalin for histology and in saline for culture, particularly for tuberculosis.

A, Multiple pleural densities visualized during thoracoscopy. B, Thoracoscopic pleural biopsy obtained.
Electrosurgical Biopsy of the Parietal Pleura
Electrocautery has been incorporated with the flexible forceps biopsy to enhance its yield. Full-thickness parietal pleural biopsies can be achieved using the insulated tip (IT) diathermic knife during flex–rigid pleuroscopy. In one study, the reported diagnostic yields were 85% with IT knife, compared with 60% with flexible forceps. The IT knife was notably useful when smooth, thickened lesions were encountered, of which nearly half were malignant mesothelioma. 16
Talc Poudrage
Thoracoscopic chemical pleurodesis is the most widely reported method of instillation of sclerosants into the pleural cavity. It is used mainly for the treatment of malignant or chronic recurrent nonmalignant pleural effusions and persistent or recurrent pneumothorax. Thoracoscopic talc poudrage is considered as the preferred technique of pleurodesis for all patients with good performance status for malignant effusions. 17 Talc poudrage is more frequently performed through the single port after removal of the thoracoscope and can be performed via a second port. Prior to the insufflation, all pleural fluid should be removed. The talc is then insufflated into the pleural space through a catheter (or cannula) placed in the working channel of the thoracoscope/pleuroscope.
In the case of 2-port procedure, the first port is left open to allow air to escape during talc insufflation, thereby avoiding a tension pneumothorax. Although an optimal dose of talc for poudrage has not been established, a dose of 5 g (4-8 g or 8-12 mL) of sterile talc is recommended. For pneumothorax cases, 2 to 3 g of talc is usually sufficient. Only dry, sterile “graded” talc with median particle size of greater than 25 μm should be used, as smaller particles may result in the deterioration in gas exchange and even acute respiratory distress syndrome. 18
Various talc delivery devices are available, such as a spray atomizer, a bulb syringe, or a spray catheter introduced through the working channel of the flex–rigid pleuroscope. Uniform distribution of the talc on all pleural surfaces is confirmed by direct vision. Talc application might be painful, and an additional analgesic before beginning the insufflation or direct lidocaine spray to the chest wall pleura via catheter is recommended.
Postthoracoscopy Care and Chest Drain Insertion
At the end of diagnostic thoracoscopy, a chest tube is inserted and air is aspirated. In order to reduce the risk of subcutaneous emphysema, a chest tube should be chosen that is approximately the same or bigger caliber of the insertion port used; a 7-mm port requires a drain of at least 20F. The patient’s respiratory rate, temperature, pulse, blood pressure, oxygen saturation, and pleural fluid drainage should be monitored and recorded every 15 minutes for the first hour following local anesthetic thoracoscopy. The chest tube can be removed once the lung has fully expanded, and there is no evidence of an active air leak. The patient may then be discharged after a brief observation in a recovery area.
If talc pleurodesis or lung biopsy is performed, the patient is hospitalized for a period of monitoring and chest tube drainage. Pain is common following thoracoscopy, particularly if talc pleurodesis has been performed, and analgesics are frequently required.
Comparison of MT to Video-Assisted Thoracoscopic Surgery
Medical thoracoscopy is regarded as a mini-invasive and less expensive procedure compared to VATS. Table 4 compares MT (rigid and flex–rigid) to VATS.
Medical Thoracoscopy Versus Video-Assisted Thoracoscopic Surgery.

Clinical Applications
Exudative Pleural Effusion of Unknown Etiology
Medical thoracoscopy is now increasingly used in the diagnosis of pleural effusion of unknown etiology. In a retrospective analysis of patients who underwent diagnostic thoracentesis over a 19-year period, the overall accuracy of pleural fluid cytology was 59%. 4 Moreover, in a recent randomized controlled trial comparing the efficacy of closed pleural biopsy and MT in the diagnosis of exudative pleural effusion of unknown etiology, MT had a diagnostic yield of 86% and complication rate of 10% compared to closed pleural biopsy (62% and 17%, respectively). 19 Thus, despite repeated pleural fluid sampling and closed pleural biopsy, the etiology of pleural effusion remains unknown in up to 25% to 40% of patients. 20,21 In a systematic review evaluating the diagnostic accuracy of MT for patients with pleural effusion of undetermined etiology, MT (semirigid) had a pooled sensitivity and specificity of 97% and 100%, respectively. 11 Although the overall diagnostic yield of rigid MT is higher compared to semirigid MT in undiagnosed pleural effusion on the intention-to-treat analysis, both modalities had a similar yield when pleural biopsies were obtained successfully. 22 In a recent study, VATS yielded a diagnostic accuracy of 92% in patients with undiagnosed exudative pleural effusion, which is comparable to MT. 23 The British Thoracic Society (BTS) guidelines recommend thoracoscopy as the next diagnostic test in patients with exudative pleural effusion of unknown etiology after an inconclusive thoracentesis. 24
Malignant Pleural Effusion
Malignant pleural effusion (MPE) is considered as a very common cause of exudative pleural effusion. The overall diagnostic sensitivity of thoracentesis is around 60%. 20 The yield of pleural fluid cytology from the first specimen is 51%, increases an additional 7% from the second specimen and only 2% from the third. 11 Thus, additional investigation is clearly needed in such a situation of a nondiagnostic thoracentesis in which MPE is a consideration (or is suspected). Thoracoscopy (MT or VATS) is highly successful in achieving diagnosis when pleural fluid cytology is nondiagnostic as well as in treating MPE through loculation breakdown, chemical pleurodesis, and/or tunneled pleural catheter (TPC) insertion. The diagnostic yield of MT is around 95% in patients with MPE and has a success rate of 90% in achieving pleurodesis. 19 Medical thoracoscopy is considered a less invasive procedure compared to VATS, requires moderate sedation, requires a single port of entry, and can be done in the endoscopy suite. Although rigid MT can obtain a larger specimen, the diagnostic yield of rigid and flex–rigid is similar in malignant pleural disease. 20
Malignant Pleural Mesothelioma
The diagnostic yield of thoracentesis in malignant pleural mesothelioma (MPM) had been reported low with a sensitivity around 30%. 25 A recent study from Western Australia, an area with high incidence of mesothelioma, found that the sensitivity of fluid cytology in the hands of an experienced cytopathologists approached 73%. 26 Thus, the diagnosis of MPM is challenging and often requires pathological diagnosis. Closed pleural biopsy has a very low sensitivity (16%) 27 and generally has a limited value in the diagnostic workup for MPM. Thoracoscopic biopsy techniques such as MT have high diagnostic sensitivity approaching 98% 28 and allow treating patients in the same setting with chemical pleurodesis and/or TPC insertion. It is important to mention that catheter tract metastases can develop in about 10% of patients (mostly with the diagnosis of mesothelioma). Symptoms are usually mild and usually respond to radiotherapy. 29
Tuberculous Pleural Effusion
Tuberculous (TB) pleural effusion occurs as the initial presentation in approximately 5% of patients with Mycobacterium tuberculosis infection. 30 Acid-fast bacilli smear and culture of pleural effusion have a very low yield of <10% and <30%, respectively. 30 The diagnostic accuracy of TB pleural effusion can be increased by adding a pleural fluid test such as adenosine deaminase (ADA). A recent systematic review and meta-analysis determined that the overall diagnostic sensitivity and specificity of ADA were 86% and 88%, respectively, 31 but results should be interpreted in the context of pretest probability of the disease. Also, closed pleural biopsy has an overall sensitivity of 80% but increases to 93% when combined with high ADA pleural fluid level in areas with endemic TB. 32,33 Medical thoracoscopy has also been widely used in the diagnostic workup of TB pleural effusion with a sensitivity of 93% in developed countries and increasing to 98% in endemic countries. 33,13 The most common visual findings of TB pleural effusion on MT are diffuse miliary nodules, necrosis, and adhesions. 34 Medical thoracoscopy is also useful for loculated pleural effusions and adhesiolysis. 35 Moreover, MT can provide larger quantities of tissues for culture in suspected multidrug-resistant TB.
Empyema and Complicated Parapneumonic Effusion
Pleural infection is associated with a high morbidity and mortality, and the incidence of related hospitalization was 5.98 per 100 000 in 2008. 36,37 The initial treatment of empyema and complicated parapneumonic effusion consists of antibiotics and chest tube drainage. Furthermore, the instillation of intrapleural tissue plasminogen activator/deoxyribonuclease therapy to breakdown loculations and adhesions reduced the need for surgical intervention and the length of hospital stay. 38,39 Despite therapy mentioned above, around 4% to 8% 38,39 of patients might require further surgical intervention. Medical thoracoscopy was retrospectively analyzed in 2 studies for patients referred with multiloculated empyema. 40,41 Medical thoracoscopy was successful in 86% to 91% of patients, and 6% to 14% required further surgical interventions such as VATS and thoracotomy. 40,41 The complication rate from MT was 7% to 9% (cutaneous fistula, pneumothorax, hemothorax, and subcutaneous emphysema). 40,41 The use and safety of MT in pleural infection are still not defined, and more data are needed to clarify its role in such population.
Pneumothorax
Pneumothorax is commonly encountered by pulmonologists. The role of MT had been explored in primary (no underlying lung disease) and secondary pneumothorax (predisposing lung disease). The initial approach usually consists of aspiration or chest tube drainage; however, further treatment such as pleurodesis might be indicated in many cases with recurrence or unresolving pneumothorax. Tschopp et al 42 compared the recurrence rate of pneumothorax in primary spontaneous pneumothorax treated with pleural drainage versus MT with talc pleurodesis. Those who underwent MT had a recurrence rate of 5% compared to 34% in the pleural drainage group. 42 Furthermore, MT can visualize apical subpleural blebs or bullae under white light MT and pleural porosity (diffuse visceral pleural lesions) under fluorescein-enhanced autofluorescence MT. It has been suggested that pleural porosity rather than blebs may be responsible for air leak. 43 Patients with chronic obstructive pulmonary disease (COPD) are at increased risk of developing spontaneous pneumothorax and might be a poor surgical candidate for VATS. Lee et al 44 prospectively evaluated patients with severe COPD with an average forced expiratory volume in 1 second (FEV1) of 41% of predicted who underwent MT and talc pleurodesis. The success rate of achieving pleurodesis was 95% at a median follow-up of 35 months. However, around 10% of the patients died within 30 days attributed to severe COPD, ischemic heart disease, and pneumonia. This is in comparison with another small retrospective trial showing mortality rate of around 9% in a patient with COPD undergoing VATS for secondary spontaneous pneumothorax with a mean FEV1 of 48%. 45 However, further studies are needed to investigate the role of MT in patients with secondary pneumothorax who are poor surgical candidates.
Contraindication
Table 5 lists the absolute and relative contraindications to MT.
Medical Thoracoscopy Contraindications.

Complications
Medical thoracoscopy is a relatively safe procedure with a very low mortality rate. In 2010, BTS Pleural Disease Guideline reported an overall mortality of 0.34%. 13 In studies that only reported diagnostic MT, the mortality rate was 0%. 13 On the other hand, therapeutic MT with talc poudrage had a mortality rate of 0.69%. 13 It is important to mention that one study used nongraded talc and contributed to majority of mortality rate. 46 Major complications such as empyema, hemorrhage, port site metastasis, bronchopleural fistula, postoperative pneumothorax, and pneumonia occurred in about 1.8%, whereas minor complications such as subcutaneous emphysema, minor bleeding, skin site infection, hypotension during procedure, atrial fibrillation, and fever occurred in 7.3% according to the recent BTS Pleural Disease Guideline. 13 Moreover, a small series (29 patients with 6 who had previous talc pleurodesis) reported that repeat MT along with pleurodesis in the majority of cases on the same ipsilateral site was feasible without increased morbidity or mortality. 47 Furthermore, flex–rigid MT might be even safer than rigid MT. A meta-analysis that included 17 studies using flex–rigid MT reported no associated mortality and around 1.5% complication rate. 48 In studies comparing flex–rigid to rigid MT directly, the former had a lower complication rate (around 3% vs 5%). 49
Injury to the intercostal artery (ICA) is an uncommon complication during thoracoscopy but can potentially be life threatening. Pleural hemorrhage has been reported to occur in about 4% of cases of thoracoscopies. 50 Slight bleeding almost always resolves spontaneously. If bleeding continues or ICA has been biopsied inadvertently, the bleeding area should be compressed. In case of massive or continued bleeding, thoracic surgical consultation should be obtained immediately for the possible repair of ICA. Angiographic techniques have also been found to be useful to accurately localize and embolize the lacerated ICA to control the bleeding. 51
Medical Thoracoscopy Training and Competency
There is no general consensus among international societies about what is considered an adequate training in MT. The American College of Chest Physician (ACCP) suggested that 20 supervised MTs are needed before trainees are considered competent, with additional 10 procedures every year afterward to maintain competency. 52 The BTS Pleural Disease Guideline outlined basic and advanced MT in levels I and II, but the required training to gain competence at each level was not specified. 13 However, as with all procedures, there is a learning curve, individualized according to operators’ own speed of acquiring knowledge, before trainees could achieve competence.
Conclusion
Medical thoracoscopy is an overall safe procedure with very low complication and mortality rate when performed by trained pulmonologists. The application of MT in pleural diseases is supported by studies showing high diagnostic yield and effective therapeutic intervention. Medical thoracoscopy appears to be valuable in patients who are not surgical candidates or are at an increased risk of complications from more invasive procedures such as VATS. Although such a minimally invasive procedure is still underutilized, it is expected that MT will be widespread as interventional pulmonology programs are now training more pulmonologists in this procedure.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
