Abstract
Background. The most prevalent microvascular consequence of type 1 diabetes (T1D) is diabetic nephropathy (DN). Aim of the Study. To find the clinical, anamnestic, and genetic markers that characterize and forecast early diabetic nephropathy in T1D children. Methods. One hundred four children with T1D and DN between the ages of 2 and 17 were surveyed. Stepwise logistic regression models and linear regression models were used. Results. BMI, systolic blood pressure, concurrent kidney pathology, anamnesis viral infections, ESR level, serum cholesterol, blood urea, number of DKA episodes/year, and GFR were determined to be predictors of early DN in children with T1D. Bcl-xL, caspase-3, and HIF-1alfa were discovered to predict DN among all previously identified variables influencing apoptosis. Conclusion. BMI, systolic blood pressure, concurrent kidney disease, anamnesis viral infections, ESR level, serum cholesterol, blood urea, number of DKA episodes/year, GFR, apoptotic and hypoxia markers were discovered as variables predicting early DN.
Introduction
A major epidemic in the modern world is diabetes mellitus (DM).1-4 Between 25 and 40 percent of people with type 1 diabetes experience diabetic nephropathy (DN), one of the most common microvascular consequences of diabetes mellitus (T1D). End stage renal disease (ESRD) in adults is most frequently brought on by this one factor in the West. In diabetic nephropathy, the renal glomeruli undergo pathological alterations that cause albuminuria, hypertension, and a gradual loss in renal function.2-4 The earliest feature of DN is typically microalbuminuria. 5
Historically, the clinical context has used the measurement of microalbuminuria (30-300 mg/g) to test for diabetic nephropathy. Evidence, however, points to the development of renal pathological alterations, such as nephromegaly and glomerular basement membrane thickening, soon after the diagnosis of diabetes and considerably earlier than the detection of microalbuminuria. 6
Numerous clinical variables, such as the length of diabetes, puberty, age at onset, family history of insulin resistance, type 1 and type 2 diabetes, genetic factors, race/ethnicity, modifiable, glycemic (metabolic) control, smoking, hyperlipidemia, intrauterine exposure, obesity, pregnancy, and social status, have been discussed in terms of factors predicting the development of DN. 7
In tubular cells, hyperglycemia causes the production of free radicals and oxidative stress. Reactive oxygen species are thought to be crucial mediators of a number of biological processes, such as apoptosis, extracellular matrix deposition, and proliferation. Many different cell types, including proximal tubular epithelial cells, are stimulated to undergo apoptosis by high glucose concentrations. 8 Uncertainty surrounds the mechanism by which hyperglycemia triggers apoptosis. The induction of apoptosis in HK2 cells by a high glucose concentration of 30 mmol/L for 18 to 48 hours has been demonstrated using an in vitro model.8,9 It has been demonstrated that prolonged exposure of proximal tubular epithelial cells (1-13 days) to a hyperglycemic environment inhibits cell proliferation and causes growth arrest or cellular ap-optosis.8-13
The phosphorylation/inactivation of Bcl-2 in microtubules treated with apoptotic drugs has been demonstrated in prior studies to be mediated by the serine/threonine kinase, mTOR. In order to keep track of the organism’s genetic programing, Bcl-2 is crucial. A family of positive and negative apoptosis regulators called Bcl2 related proteins. Bcl-2 and its close homolog Bcl-XL are anti-apoptotic, whereas BAD or BAX are pro-apoptotic members of the Bcl-2 family.14,15 It has been demonstrated that Bcl-2 blocks the release of cytochrome C from mitochondria, hence preventing the activation of caspase 9, the initiator caspase. As a physiological process during normal cell cycle progression or as a defense mechanism after being activated by various stimuli and stress, several kinases, including JNK, p38, and cdc2/cyclin B kinase, have been observed to phosphorylate/inactivate Bcl-2. The antiapoptotic action, which causes the release of cytochrome C from the mitochondria and the activation of downstream caspases, is inhibited by the phosphorylation or inactivation of Bcl-2.14-18
The primary regulators of oxygen homeostasis in response to hypoxia are hypoxia-inducible factors (HIFs). Moreover, hyperglycemia causes methylglyoxal to build up, which facilitates the destabilization of HIF-1. As early as 3 days following the introduction of diabetes, hypoxia is shown in animal models, primarily in the medullary region. Tubular atrophy and interstitial fibrosis are caused by hypoxia in the renal tubules, which can exacerbate glomerular disease as diabetic nephropathy develops. The promotion of extracellular matrix expansion by tubular hypoxia leads to further reductions in oxygen delivery and the start of a vicious loop that aids in the onset of diabetic nephropathy. 18
Children with T1D and early-stage DM have not yet been thoroughly examined in terms of the effects of apoptosis and hypoxia. Also, in terms of those predicting the onset and progression of albuminuria as well as subsequent illness progression, indicators of chronic hypoxia and apoptosis were not examined in children with T1D and DN.
In order to determine potential indicators that can predict DN in children with T1D, we evaluated clinical, laboratory, instrumental, anamnestic data, markers of chronic hypoxia and apoptosis in pediatric patients with T1D, as well as early stages of DN.
Material and Methods
Patients Characteristics
The study conducted during 2015 to 2020 and included 104 children (62 boys and 42 girls) aged 2 to 17 years with T1D with/without DN followed in the Endocrinology unit. Group entitled T1D were patients with diagnosed T1D and no signs of DN. Patients with T1D who also had DN for an ≤1 year made up the group called DN group. 32 healthy individuals who showed no symptoms of proteinuria or accompanying illnesses made up the control group. All patients and their families have given their informed consent. The local ethics committee gave its approval to the study (Protocol №142). Children (under the age of 12) and/or their parents signed each informed consent, which was then recorded in the patient’s medical history. The criterias for including and excluding patients from the study are listed in the supplemental material.
All patients have a thorough examination that includes standard procedures (physical examination, blood pressure measurement, blood tests, analysis of urine sediment, renal ultrasonography, ECG, etc.). Urinary microalbumin excretion was assessed in 24-hour urine collection samples. Glomerular filtration rate (GFR) is a measurement of kidney health. This is the Schwartz formula for kids and teenagers aged 1 to 17. 19
All patients with T1D with/without DN received treatment with insulin in basal-bolus regimen. All patients were visited every 3 months. Chronological age, the length of diabetes, weight, height, BMI, blood pressure, Hb1Ac, serum cholesterol, complete blood count, urinalysis, indicators of kidney and liver function, and blood pressure were all recorded at each follow-up hospital visit and taken for the study analysis from medical records. Blood samples for the apoptosis markers analysis taken at the follow-up visit to hospital.
Immunoblotting for Detection of BcL-xL, Caspase-3, HIF-1alfa
Measurement of intracellular hypoxia response and apoptosis indicators is done using plasma samples. SDS-PAGE was used to resolve proteins that had been dissolved in Laemmli sample buffer in polyacrylamide gels before they were transferred to a polyvinylidene difluoride membrane. Membranes were then immunoblotted with the Bcl-xL and HIF-1 Ab (Cell Signaling Technology, Danvers, MA USA) and the caspase-3 Ab (Cell Signaling Technology, Danvers, MA USA) for an hour at room temperature. A loading control was performed using the actin mouse mAb. Chemiluminescent substrate ECL allowed for the visualization of the protein bands. By using densitometric analysis, the protein content was quantified.
Statistics
The statistics presented as means, SEM, and, when appropriate, frequencies and percentages. To determine the importance of differences, an ANOVA was used, followed by the post hoc Kruskal-Wallis test. To evaluate the researched parameter’s potential to predict steroid resistance, regression analysis was conducted. The study of factor correlations uses Pearson correlation. Data processed with the Windows version of GraphPad Prism 9.0 (USA, San Diego, CA). Statistics are deemed significant with P values < .05.
Results
Apoptosis and Chronic Hypoxia Markers in Children With T1D and DN
The demographic and clinical variables data of the patients included into the study described in Table 1.
Clinical Characteristics of Patients.

P < .05. **P < .01. ***P < .001.
Using immunoblotting, the expression of proapoptotic and antiapoptotic factors was examined as a measure of intracellular hypoxia. Caspase-3 and Bcl-xL were chosen as pro- and anti-apoptotic indicators, respectively. HIF-1a has been investigated as a chronic hypoxia marker.
Bcl-xL levels in the T1D group were 151.90.9 a.u. and 172.30.9 a.u., respectively, substantially lower than in the control group (P < .001). Bcl-xL levels in the DN patient group were measured at a level of 145.52.23 a.u., which was considerably lower than the values in the T1D group and the control group (P < .0001), respectively (Figure 1A).

Levels of anti-apoptotic (A) and pro-apoptotic (B) markers in children with T1D and DN.
Children with T1D had levels of caspase-3, a last effector in mitochondrial apoptosis, measured at 137.41.55 a.u. (P < .0001 compared to control group value) while children with DN had levels of 147.02.62 a.u. (P < .0001 compared to control group value; P < .05 compared to T1D group value). Children from the control group had caspase-3 levels that were recorded at 110.92.48 a.u (Figure 1B). HIF-1alfa levels as a measure of intracellular hypoxia were examined in all patients. Children with T1D had significantly greater levels of HIF-1alfa than the control group (P < .01) and the group with DN (P < .0001) − 164.01.42 a.u., 140.22.8 a.u., and 185.12.47 a.u., res-pectively (Figure 2).

Level of HIF-1 in children with T1D and DN.
Identification of Clinical, Biochemical, and Metabolic Variables That Define and Forecast Early DN in T1D Children
A step-by-step selection process was used to create logistic regression models for the probabilities of DN among numerical covariates in children with nephrotic syndrome. potential predictors that join the model at 0.05. Six models were made: (I) Basic clinical factors: disease progression, gender, BMI, systolic and diastolic blood pressure; (II) Basic anamnestic parameters: existence of concurrent endocrine pathology, presence of diabetic neuropathy, relatives with T1D, and viral infections (rubella, CMV, mumps, measles) in anamnesis; (II) basic laboratory assessments of inflammation, including the PLT count, WBC count, ESR level, and albumin/globulin ratio; (IV) Basic biochemical measurements (serum cholesterol, ALAT, and ASAT); (V) indicators of kidney state and diabetes management (serum creatinine and urea, blood sugar, Hb1Ac, frequency of DKA events per year, and GFR); (VI) - analysis of the markers of hypoxia HIF-1alfa, caspase-3, and BcL-xL, which regulate apoptosis. The ability of each model to define early DN was evaluated using receiver operating characteristics curve and area under the curve (AUC) studies (Figure 3).

Receiver operating characteristics curve predicting DN in T1D children. (I) basic clinical factors—disease course, gender, BMI, systolic BP, diastolic BP; (II) basic anamnestic parameters: presence of concomitant kidney pathology, T1D in relatives, viral infection (rubella, CMV, mumps, measles) in anamnesis, presence of concomitant endocrine pathology, presence of diabetic neuropathy; (III) basic laboratory tests reflecting inflammatory status—PLT count, WBC count, ESR level, albumin/globulin ratio; (IV)—Basic biochemical parameters (serum cholesterol, ALAT, ASAT); (V) markers of kidney function and diabetes course and compensation (serum creatinine and urea, blood glucose, Hb1Ac, number of DKA episodes/year, GFR); (VI)—apoptosis regulating factors BcL-xL, caspase-3 and marker of hypoxia HIF-1alfa analyzed.
The clinical indicators of early DN in children with T1D were identified using multivariate linear regression analysis. Disease duration, gender, BMI, and systolic and diastolic blood pressure are among demographic factors that were taken into consideration for the analysis. BMI (β .02, SE 0.009, 95% CI −0.04 to −0.004, P < .05) and systolic blood pressure value (β .03, SE 0.003, 95% CI 0.02-0.03, P < .0001) were the 2 factors that predicted the early stages of DN. None of the additional characteristics examined were shown to be predictors of early DN, including disease course (β .0008, SE 0.01, 95% CI −0.02 to 0.02), male gender (β .004, SE 0.03, 95% CI −0.06 to 0.07, P = 0.88), and diastolic blood pressure (β-.006, SE 0.005, 95% CI −0.02 to 0.0005, P = .25) (Table 2).
Basic Clinical Parameters Predicting Early DN in Children With T1D.

Note: The bold lines mean statistical difference (P < 0.05).
The anamnestic determinants of early DN in children with T1D were identified using multivariate linear regression analysis. The following criteria were taken into consideration: the presence of concurrent renal disease, T1D in relatives, viral infection (rubella, CMV, mumps, measles), concurrent endocrine pathology, and the presence of diabetic neuropathy. Concomitant kidney pathology (β .37, SE 0.14, 95% CI 0.09 to 0.64, P .01) and viral infections in anamnesis (β .34, SE 0.11, 95% CI 0.12 to 0.56, P .01) were factors indicating early stage of DN. T1D in relatives (β .11, SE 0.11, 95% CI −0.11 to 0.34, P = .32), concurrent endocrine disorders (β .05, SE 0.12, 95% CI −0.2 to 0.029, P = .68), existence of diabetic neuropathy (β −.06, SE 0.11, 95% CI −0.23 to 0.16, P = .58), and allergies (β .011, SE 0.17, 95% CI −0.24 to 0.45, P = .54) were not discovered to be predictors of early DN in children (Table 3).
Basic Anamnestic Parameters Predicting Early DN in Children With T1D.

Note: The bold lines mean statistical difference (P < 0.05).
PLT count, WBC count, ESR level, and albumin/globulin ratio were examined using logistic regression analysis as potential markers predicting DN in T1D patients. These basic laboratory values represent inflammatory status. In children with T1D, the ESR level was discovered to be a predictor predicting early DN (OR: 1.1; OR 95% CI: 1.02-1.21; P .05). PLT count (OR: 0.1; OR 95% CI: 0.99-1.01; P = .65), WBC count (OR: 1.02; OR 95% CI: 0.81-1.28; P = .9), and albumin/globulin ratio (OR: 0.18; OR 95% CI: 0.02-1.2; P = .11) did not prove to be significant DN predictors (AUC: 0.71; SE: 0.05; 95% CI: 0.6) (Table 4).
Basic Laboratory Parameters Predicting Early DN in Children With T1D.
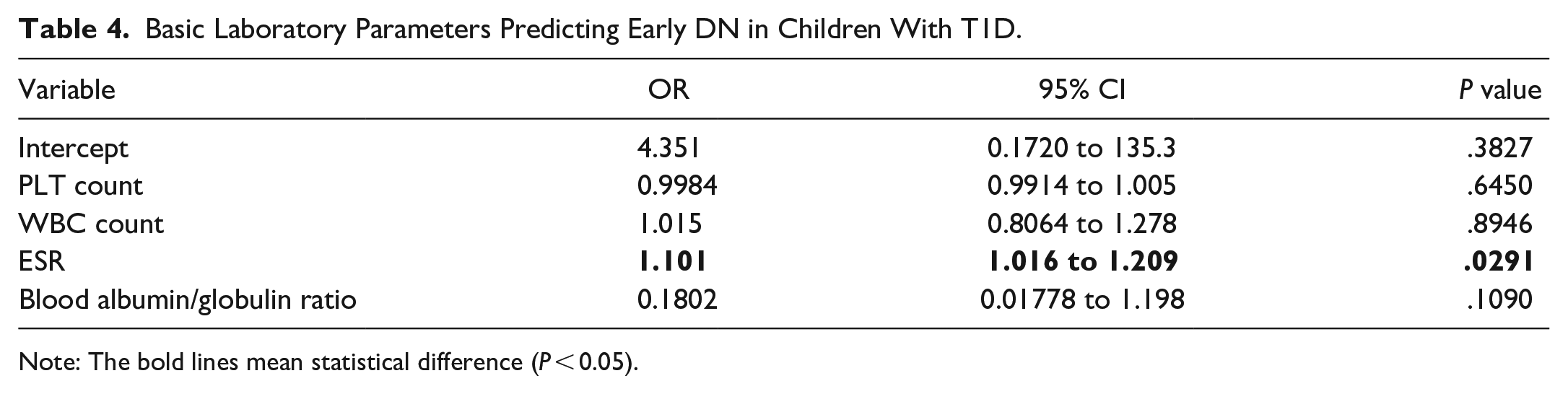
Note: The bold lines mean statistical difference (P < 0.05).
Serum cholesterol, ALAT, and ASAT levels were chosen as the second group in the logistic regression analysis of fundamental biochemical parameters. In children with T1D, serum cholesterol level was chosen as a predictor predicting early DN (OR: 2.7; OR 95% CI: 1.8-4.42; P .0001). The results of DN (AUC: 0.83; SE: 0.05; 95% CI: 0.74-0.91; P .001) have not been found to be significantly predicted by ALAT level (OR: 1.03; OR 95% CI: 0.98-1.09; P = .27) or ASAT level (OR: 1.00; OR 95% CI: 0.07-1.04; P = .99) (Table 5).
Basic Biochemical Parameters Predicting Early DN in Children With T1D.

Note: The bold lines mean statistical difference (P < 0.05).
Markers of renal function and diabetes course and compensation (serum creatinine and urea, blood glucose, Hb1Ac, number of DKA episodes/year, GFR) were the next group of biochemical characteristics added to the logistic regression model.
Blood urea level (OR: 0.62; OR 95% CI: 0.38-0.98; P = .04), number of DKA episodes/year (OR: 3.89; OR 95% CI: 2.37-7.53; P .0001), and GFR (OR: 0.95; OR 95% CI: 0.9-0.99; P .05) were chosen as significant factors predicting early DN in children with T1D. Blood glucose level (OR: 1.14; OR 95% CI: 0.91-1.45; P = .28), Hb1Ac (OR: 0.9; OR 95% CI: 0.67-1.19; P = .48), and S-Cr (OR: 1.02; OR 95% CI: 0.96-1.07; P = .58) were not discovered to be significant DN predictors (AUC: 0.98; SE: 0.02; 95% CI: 0) (Table 6).
Kidney Function and Glucose Metabolism Parameters Predicting Early DN in Children With T1D.

Note: The bold lines mean statistical difference (P < 0.05).
BcL-xL, caspase-3, and a marker for hypoxia are factors that regulate apoptosis. HIF-1alfa was examined in terms of the variables that predicted early DN. Bcl-xL (OR: 0.88; OR 95% CI: 0.82-0.93; P .0001), caspase-3 (OR: 0.93; OR 95% CI: 0.87-0.98; P .01), and HIF-1alfa (OR: 1.08; OR 95% CI: 1.03-1.13; P .01) were all identified as significant DN predictors (AUC: 0.92; SE: 0.03; 95%) (Table 7).
Apoptosis and Hypoxia Markers Predicting Early DN in Children With T1D.

Note: The bold lines mean statistical difference (P < 0.05).
BcL-xL, caspase-3, HIF-1alfa, and Hb1Ac were revealed to be the main characteristics determining DN in T1D patients and were correlated using the Pearson correlation approach (Figure 4). Substantial negative correlations between the anti-apoptotic factor BcL-xL and the pro-apoptotic factor caspase-3 (r = − 0.85, P < .05) and the anti-apoptotic factor BcL-xL and Hb1Ac (r = −0.68, P < .05) were found. Hb1Ac and the cellular hypoxia marker HIF-1alfa showed a significant positive correlation (r = .79, P < .01), as did Hb1Ac and the pro-apoptotic protein caspase-3 (r = .6, P < .05).
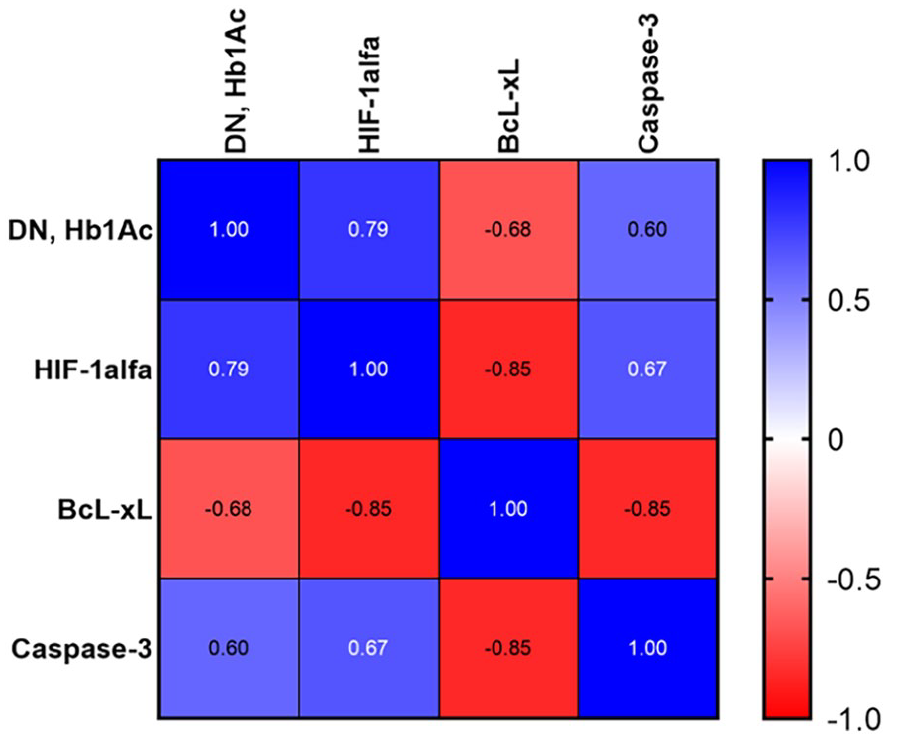
Heat map correlation between factors regulating apoptosis, hypoxia marker and Hb1Ac in children with DN.
Discussion
One of the most prevalent chronic disorders affecting kids and teenagers is T1D. It has long been recognized that diabetes, particularly its long-term consequences related to DN, is significantly linked to an increase in mortality. DN’s development has experienced a protracted “quiet phase.” End-stage renal disease (ESRD) continues to be mostly brought on by DN worldwide. 20 The key early prognostic indicator of further complications is microalbuminuria. 3
Male sex and higher mean HbA1c were linked to macroalbuminuria, 2 risk variables that encourage the onset and progression of diabetic nephropathy. Higher mean HbA1c, higher mean triglycerides, older age, and higher systolic blood pressure were the most important risk factors for incident decreased GFR.7,21 Yet not enough research has been done on the variables that predict the early stage of DN linked with microalbuminuria, particularly in juvenile patients.
In this study, we focused on the early detection of microalbuminuria in children with T1D and the early confirmation of DN. Studies of apoptosis and hypoxia markers were carried out concurrently with analyses of fundamental clinical parameters, anamnestic data, and anamnestic information.
With an average disease duration of 4.9 to 6.5 years, this group is referred to as T1D. A year following the first recorded occurrence of albuminuria, children with DN were observed as part of the DN group. In both the T1D and DN groups, there were no variations in the patients’ gender distribution, average age, BMI, or heart rates. Systolic blood pressure in the T1D group was noticeably lower than in the DN group. No variations were seen in diastolic blood pressure.
ESR levels and the albumin/globulin ratio were altered in the DN group, indicating that the pro-inflammatory status of those patients. These findings are consistent with other evidence that demonstrate the pro-inflammatory condition in DN. It has recently been discovered that the synergistic effects of interferon gamma (IFN-gamma) and the innate inflammatory cytokines TNF-alpha and IL-1beta appear to be responsible for the inflammation of pancreatic beta cells in T1D. Nitric oxide (NO) is produced as a result of the elevation of inducible nitric oxide synthase (iNOS) caused by the combined effects of these inflammatory chemicals. 22 A significant contributor to future kidney and vascular damage, serum cholesterol was observed in the DN group to be above normal levels at the same period. Patients with DN in this group have. Based on evaluation of S-Cr and GFR levels, patients from the group with DN have a slight impairment of renal function.
The regulation of apoptosis by the BCl-2 family has long been known, that is, in experimental forms of diabetes mellitus.14-17,23 In the kidney tissue of kids with T1D and DN, we measured the presence of proteins from the apoptotic family. Our findings indicate that children with DN had higher levels of caspase-3 and lower levels of BcL-xL, 2 anti-apoptotic factors, than children with T1D. BcL-xL and caspase-3 were found to be important predictors and descriptors of DN, according to a logistic regression model. HIF-1alfa, a crucial transcriptional factor controlling numerous biological activities, was evaluated in all patients with T1D and DN. The constitutively expressed HIF-1 and the rate-limiting HIF1 form the heterodimer known as HIF-1. 18
Under the ongoing influence of MAU and glucose, DN is linked to persistent low-grade chronic inflammation. 24 By increasing the inner mitochondrial membrane’s permeability, which results in the release of cytochrome C, hypoxia can trigger apoptosis. Our data in line with literature data. 25 Our findings indicate that the amount of HIF-1alfa was higher in DN than T1D. HIF-1alfa was also discovered to be a role in the definition and prediction of DN in children.
We hypothesize that in children with DN, hypoxia brought on by Hb1Ac production may be another component triggering the mitochondrial apoptotic pathway. The enhanced oxygen affinity of glycosylated hemoglobins has been demonstrated. It was recently discovered an association between low oxygen saturation and poor glycemic management. 26 Many triggers can cause intrinsic apoptosis, also referred to as mitochondrial apoptosis. The Bcl-2 protein family regulates the intrinsic apoptotic pathway including albumin.27-29 Summarized picture of the pathway of factors controlling apoptosis under influence of chronic glucose and albumin exposure in children with early DN given in Figure 5 (picture was created our-self based on literature data).

Summarized picture of the pathway of factors controlling apoptosis under influence of chronic glucose and albumin exposure in children with early DN.
As a result, we draw the conclusion that individual analysis of apoptosis-controlling factors, such as BcL-xL, caspase-3, and HIF-1alfa, along with routine evaluation of BMI, systolic blood pressure, the presence of concurrent kidney pathology, viral infections in anamnesis, ESR level, serum cholesterol, blood urea, number of DKA episodes/year, and GFR, may have clinical significance in predicting and defining We suggest a simplified model of the variables predicting early DN in T1D children (Figure 6).

Summarized scheme of the Clinical, laboratory, anamnestic, apoptosis-controlling factors predicting early DN in children with T1D.
There are several restrictions on this study that must be discussed. We conducted a cross-sectional, patient-limited pilot trial at a single site. Potential limitations of this study is that study did have a sample size calculation.
The benefit of enrolling patients is that they were examined for a wide range of clinical, laboratory, and anamnestic markers concurrently with the assessment of apoptosis and signs of hypoxia. The ability of our panel to differentiate between DN and T1D (AUC values) is strong enough.
Conclusions
We conclude that individual analysis of apoptosis-controlling factors, such as BcL-xL, caspase-3, and HIF-1alfa in combination with routine evaluation of BMI, systolic blood pressure, anamnestic data, that is, the presence of concurrent kidney pathology, past viral infections, ESR level, serum cholesterol, blood urea, number of DKA episodes/year, and GFR, may have clinical significance in predicting and defining early DN in T1D children.
Supplemental Material
sj-docx-1-gph-10.1177_2333794X231214456 – Supplemental material for Apoptosis-Controlling, Clinical, Laboratory, Anamnestic Factors in Prediction of the Early Stage of Diabetic Nephropathy in Children
Supplemental material, sj-docx-1-gph-10.1177_2333794X231214456 for Apoptosis-Controlling, Clinical, Laboratory, Anamnestic Factors in Prediction of the Early Stage of Diabetic Nephropathy in Children by Ievgeniia Burlaka in Global Pediatric Health
Footnotes
Acknowledgements
We acknowledge the assistance of Endocrinology unit of the Clinical Pediatric Hospital No. 6 (Kyiv, Ukraine).
Author Contributions
The author confirms responsibility for study desing, conception, data collection, analysis and interpretetion of results, manuscript preparation.
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Statement
This research was conducted in accordance with the Declaration of Helsinki 1964. The study was approved by the Bogomolets National Medical University local ethics committee (Protocol No. 142).
Patient Consent Statement
Written informed consent was obtained from all study participants before taking part in this research.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
