Abstract
Diuretic therapy, commonly used in the newborn intensive care unit, is associated with a variety of electrolyte abnormalities such as hyponatremia, hypokalemia, and hypochloremia. Hypochloremia, often ignored, is associated with significant morbidities and increased mortality in infants and adults. Clinicians respond in a reflex manner to hyponatremia than to hypochloremia. Hypochloremia is associated with nephrocalcinosis, hypochloremic alkalosis, and poor growth. Besides, the diuretic resistance associated with hypochloremia makes maintaining chloride levels in the physiological range even more logical. Since sodium supplementation counters the renal absorption of calcium and lack of evidence for spironolactone role in diuretic therapy for bronchopulmonary dysplasia (BPD), alternate chloride supplements such as potassium or arginine chloride may need to be considered in the management of hypochloremia due to diuretic therapy. In this review, we have summarized the current literature on hypochloremia secondary to diuretics and suggested a pragmatic approach to hypochloremia in preterm infants.
Introduction
Bronchopulmonary dysplasia (BPD) is one of the most common morbidities in extremely premature infants. Our understanding of the pathophysiology of BPD is still evolving. 1 Hence, several pharmacological agents, such as caffeine, diuretics, bronchodilators, and systemic corticosteroids, are being used in the management of BPD.2,3 Although long term benefits of diuretics in BPD are still debatable, the diuretics are still used extensively in the management of BPD in preterm infants.3,4 Diuretics, both furosemide, and combination of chlorothiazide and spironolactone, may improve airway resistance, specific airway conductance, decrease pulmonary edema, and improve lung compliance in the short term.5-7 Though strong evidence for the routine use of diuretics in the management of BPD is lacking,8,9 the perceived benefits of diuretics 10 in a condition that imposes severe morbidity in this vulnerable population have led to their widespread use.
Nevertheless, after much debate, once a clinician considers diuretics for the management of BPD, he faces the prospect of electrolyte abnormalities such as hyponatremia, hypokalemia, and hypochloremia. It is a common knee jerk reaction to initiate sodium chloride supplements 11 with or without potassium to correct these electrolyte derangements. However, few questions that invariably pop up are: Is this the ideal approach as it may negate the purpose of diuretic therapy? Should we add a potassium-sparing diuretic? Is it necessary to correct hypochloremia even in the absence of hyponatremia?
As chloride cannot be dealt in exclusion of sodium, we will briefly discuss hyponatremia and its significance during diuretic therapy.
Hyponatremia
Hyponatremia is a fairly common clinical problem in preterm infants. Most authors have defined hyponatremia as serum sodium levels of <130 mEq/L. 12 Hyponatremia is seen in up to 30% of very low birth weight infants in the first week after birth (early onset) and the incidence is much higher thereafter, up to 25% to 80% as per various reports (late onset hyponatremia).12,13 Renal capacity to regulate sodium absorption and excretion depends on the gestational age and postnatal age. 14 Increased urinary losses, due to impaired absorption of sodium in the renal tubules, is considered as one of the major reasons for hyponatremia in very low birth weight (VLBW, babies with birth weight <1500 g) babies. Hence, diuretic use may be one of the major potentiating factors for late onset hyponatremia in many of these preterm infants. 15 Unlike in adults it is not clear if there is higher incidence of hyponatremia associated with thiazide diuretic use in preterm infants. 16
Studies have associated hyponatremia with poor outcomes such as seizures,17,18 poor growth15,19, increased duration of hospital stay, 20 hearing loss, 21 cerebral palsy, 22 and increased mortality.23,24 Hyponatremia associated with diuretic use has not been well studied. Nevertheless, it is frequently treated with sodium chloride supplements, in up to 60% to 100% of cases as per some reports. 11 However, due to the concern that interstitial edema is a contributing factor in the pathophysiology of BPD, the role of sodium chloride supplements becomes questionable.3,11,25 Hence, one should exercise caution while routinely using sodium chloride supplements.
Hypochloremia
Hypochloremia is defined as serum chloride level <96 mMol/L. 26 Though the true prevalence of hypochloremia in preterm infants is unknown, some studies have reported incidence of 16.3% with loop diuretics. 27 There is paucity of literature on the significance of hypochloremia in preterm infants. Most of the recent studies on diuretics usage in preterm infants do not discuss hypochloremia11,28 and even the Cochrane review on diuretics use for CLD only briefly mentions hypochloremia as a complication. The current review therefore, addresses this important issue with existing evidence, and suggest a careful approach to the management of this important dyselectrolytemia during diuretic therapy.
The Physiological Importance of Chloride
Chloride plays vital roles, including acid-base equilibrium, gastric hydrochloric acid secretion, modulation of the renin-angiotensin-aldosterone system, and thus the systemic blood pressure, and Hamburger shift in carbon dioxide transport at the venous end of the capillaries. 29
The chloride anion is exchanged for bicarbonate anion in the cortical collecting duct, and hypochloremia increases bicarbonate absorption resulting in metabolic alkalosis. The chloride anion seems to have a significant effect than sodium on the renin-angiotensin system (RAAS). When sodium is infused with bicarbonate without any chloride, the effect on the renin-angiotensin system was minimal. 30 However, there is good evidence that increased chloride delivery to the macula densa inhibits, while reduced chloride delivery stimulates renin release.31,32 It will be interesting to see if systemic hypertension that is seen in infants with bronchopulmonary dysplasia 33 is related to hypochloremia and associated RAAS stimulation, at least in part.
Clinical Importance of Chloride
Dyschloremia (hypo and hyperchloremia) is well studied in adults than in preterm infants. Hence, adult studies on dyschloremia may throw some light on the importance of chloride in clinical conditions. Several studies36-43 have shown that dyschloremia is associated with increased morbidity in adults. A recent meta-analysis of high- versus low-chloride content in perioperative and critical care fluid resuscitation concluded that there was a weak but significant association between higher chloride content fluids and unfavorable outcomes. 36 Moreover, chloride rich fluids resulted in volume-dependent increased vasopressor need 37 and possibly increased need for blood products. 38 Studies have also shown that changes in serum chloride concentration, independent of serum sodium and bicarbonate, are associated with increased risks of acute renal injury, morbidity, and mortality. 39
Moreover, hypochloremia and low normal serum chloride levels, even without overt hypochloremia, are reported to be associated with increased mortality in the general population as well. 39 A pilot study demonstrated that supplementation with lysine chloride not only corrected chloride levels but also reduced the NT-pro-BNP levels suggesting that hypochloremia may represent more than just a marker of disease severity; rather, it may be amenable to therapeutic modification. 47 There is an ongoing trial studying if therapeutic intervention with lysine chloride improves the outcomes in heart failure patients. 26 Thus, there is compelling evidence that hypochloremia is associated with poor prognosis in adult patients.
In another study evaluating the mortality risk in chronically ventilated bronchopulmonary dysplasia infants, it was noted that the degree of hypochloremia was a useful predictor of survival even after controlling for other major confounding factors like mean airway pressure and inspired oxygen concentration. 49
The compensatory hypoventilation may increase the duration or time to wean off of mechanical ventilation.53,54 Therefore, treating metabolic alkalosis by maintaining normal chloride levels may help to disrupt this vicious cycle.
Some have reported even poor cognitive functioning at 2 years of age in the infants exposed to chloride deficient formula in infancy. 60 Extended assessment at 9 to 10 years of age showed that infants who experienced hypochloremic metabolic alkalosis were at risk for deficits in language skills that require expressive language abilities. 61 The underlying mechanisms for growth failure and neurodevelopmental abnormalities associated with Cl-deficient diet is not studied.
Diuretic resistance is defined as a failure to achieve the therapeutically desired reduction in edema or blunted natriuresis despite a full dose of diuretic.62,63 Hypochloremia exaggerates this diuretic resistance. Hanberg et al, 47 in their study in acute heart failure patients, have noted that hypochloremia was associated with poor diuretic response (OR = 7.3, 95% CI 3.3-16.1, P < .001). The diuretic efficiency, which was calculated as the increase in sodium output per doubling of the loop diuretic dose, was also substantially decreased in patients with hypochloremia. PROTECT trial involving 2033 patients observed that lower chloride was associated with decreased diuretic response. The diuretic response, which was defined as weight change on day 4 per 40 mg of intravenous furosemide (or equivalent doses) administered from baseline to day 3 was significantly lower with hypochloremia. This trial also reported that there was less weight change, a need for higher diuretic doses, a smaller percentage change in BNP levels from baseline, and the more frequent requirement for adjuvant thiazide diuretics or inotropes during hospitalization in patients with hypochloremia. The trial also showed that unlike the chloride levels, sodium levels did not correlate with mortality. 64
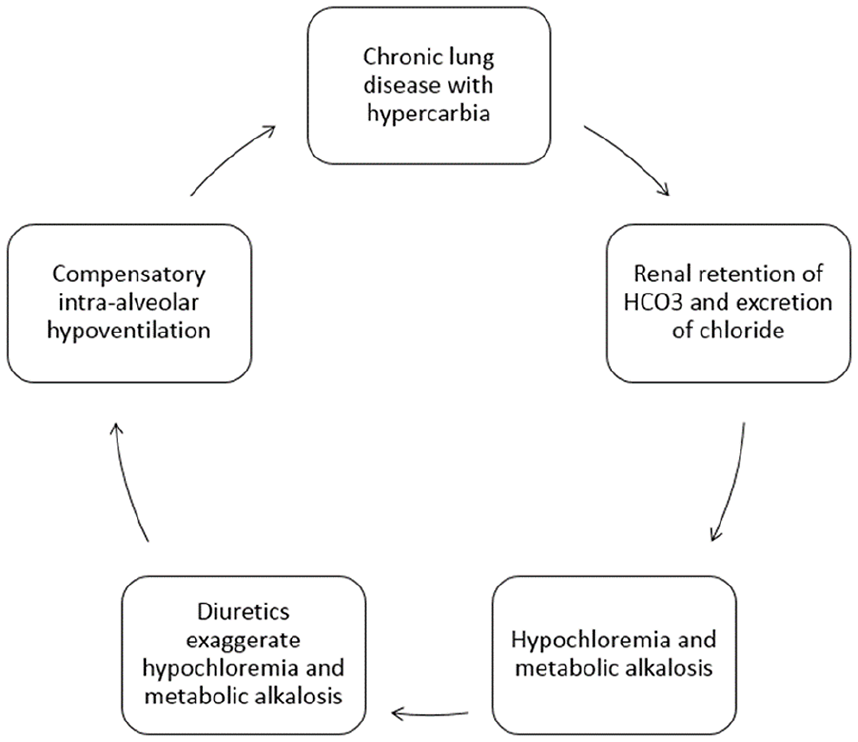
Vicious cycle of hypoventilation, hypercarbia, hypochloremic alkalosis, and hypoventilation.
As previously discussed, hypochloremia is also involved in renin release at macula densa in the distal tubule through the NKCC2 (Na-K-Cl cotransporter) pathway. Increased chloride delivery to macula densa inhibits renin release, while lower chloride stimulates renin release 32 and thus increased fluid retention and increased need for diuretics. WNK (with no lysine K) is a serine-threonine kinase that regulates Na-K-Cl and Na-Cl cotransporters in the kidneys. Binding of chloride to the active site of WNK in the thick ascending loop of Henle and distal convoluted tubule inhibits autophosphorylation and therefore decreases the availability of both sodium-potassium-chloride cotransporter and sodium-chloride cotransporter. This interaction consequently reduces renal salt reabsorption. Thus, hypochloremia may enhance fluid absorption (Figure 2). Hypochloremia may also reduce glomerular filtration rate, and thus decreasing the delivery of bicarbonate to the distal nephron and attenuating further Na and fluid losses. 41

Effects of hypochloremia at renal level.
Since hypochloremia is associated with poor growth, nephrocalcinosis, and increased mortality, there is a compelling need to maintain the chloride levels within the physiological norms. Besides, normal chloride levels are also essential for the optimal diuretic response.
Management of Hypochloremia
It is important to consider other causes of hypochloremia like pyloric stenosis, Bartter syndrome, adrenal insufficiency, pseudohypoaldosteronism, cystic fibrosis, cerebral salt wasting syndrome, chloride diarrhea), or hypochloremia due to water excess (eg, SIADH), as management in each condition is different. The current review focuses on the management of hypochloremia secondary to diuretic therapy.
In the management of hypochloremia, one should consider holding of diuretics temporarily, use of higher ratio of Cl to acetate in TPN etc. before Cl supplementation is considered. Chloride administration promotes bicarbonate excretion through chloride-bicarbonate exchange pumps in the nephron. 65 Treatment of hypochloremic alkalosis is mainly in the form of administration of potassium chloride, sodium chloride, arginine chloride, or acetazolamide.
Many neonatologists prefer to use sodium chloride supplementation as the first-line therapy for correcting hyponatremia and hypochloremia. Sodium supplementation can also create unwanted side effects. Adult data suggests that high dose sodium supplementation induces increased arterial tone by impairing endothelial function 66 or by diminishing nitric oxide bioactivity. 67 Long-term sodium intake also induces structural changes in the arterial wall and activation of the renin-angiotensin system. 68 Evidence is sparse to conclude if sodium supplementation has similar implications in the neonatal population. There are some reports, however, of significantly thicker and stiffer systemic arteries in preterm infants with BPD, compared to gestational age and sex-matched preterm infants with no BPD or term infants. 69 There is also a high incidence of systemic hypertension in infants with BPD,70,71 which is associated with a significantly longer duration of home oxygen therapy and significantly higher mortality. It needs to be further studied if this phenomenon is the result of sodium supplementation or hypochloremia.
Spironolactone is a synthetic steroid and a competitive antagonist of the aldosterone receptor. Aldosterone acts on the distal tubule and collecting duct to increase reabsorption of sodium in exchange for potassium and hydrogen ions. The blockade of the mineralocorticoid receptor by spironolactone decreases potassium secretion, and it also decreases sodium and chloride reabsorption in the principal cells. 73 The natriuresis and the resultant diuresis produced by spironolactone accounts for less than 2% of the filtered sodium, making it a very weak diuretic.
Combining spironolactone with chlorothiazide therapy does not seem to change the clinical outcomes in neonates compared to monotherapy with chlorothiazide. Hoffman et al 74 compared the diuretic therapy as a combination with chlorothiazide alone. They randomly assigned 33 preterm infants with chronic lung disease to a 2-week course of thiazide with spironolactone versus thiazide alone. The authors did not find any difference in pulmonary compliance, resistance, tidal volume, or fraction of inspired oxygen between the groups.
Hypokalemia with diuretic use is multifactorial in origin. Thiazide diuretics increase distal sodium delivery, leading to enhanced sodium reabsorption in the collecting tubule, which favors the secretion of potassium. Thiazides also lower the luminal calcium concentration along distal tubules. This activates epithelial Na channels (which are inhibited by calcium) and favors potassium secretion by increasing the electrochemical gradient for potassium secretion. 75 Increased urinary potassium excretion associated with diuretic-induced hypomagnesemia and metabolic alkalosis also may contribute to hypokalemia. 76 Finally, hyperaldosteronism, which may be associated with hyponatremia, also may promote potassium loss. 77
It is a common practice among neonatologists to add spironolactone to the diuretic regimen to decrease the degree of hypokalemia and hyperkaliuria with chronic diuretic therapy. The impact of spironolactone on potassium management has not been well understood.8,28 The effect of spironolactone on potassium homeostasis seems to be paradoxical. It is suggested that spironolactone may increase serum potassium concentrations initially, with the potential for decreases in serum potassium during prolonged therapy. 78
Routine use of spironolactone may not be more efficacious than potassium supplementation for the maintenance of serum potassium concentrations while on diuretic therapy. Hoffman et al 74 did not find any significant difference in serum electrolytes, the need for potassium chloride, or sodium chloride supplementation between the groups in their randomized study comparing chlorothiazide with a combination of chlorothiazide and spironolactone. A similar experience has been reported in the pediatric population as well. Moffett et al, 79 in a retrospective analysis of 448 patients in the pediatric cardiac intensive care unit, reported that Spironolactone supplementation did not reduce the need for potassium supplementation. Likewise, Hobbins et al 80 reported that spironolactone did not change the potassium values compared to patients on potassium supplements in 21 infants with congestive heart failure while on concomitant digoxin and chlorothiazide therapy. This lack of effect on potassium may be due to age-related end organ resistance of the distal convoluted tubule in the kidney to aldosterone in the premature infant population.81,82 Thus, despite the addition of the potassium-sparing diuretic, many infants may still require dietary supplementation with potassium chloride.
Besides, the addition of potassium-sparing diuretic prevents the clinician from providing chloride supplementation in the form of potassium chloride because of the risk of hyperkalemia. Thus, the addition of spironolactone may not only be ineffective in improving the clinical outcomes but also may interfere with electrolytes supplementation to improve chloride homeostasis.
Potassium supplementation decreases urinary calcium loss. The study showed that potassium supplementation in children with hypercalciuria significantly decreased urinary calcium excretion. 83 Similarly, potassium chloride, when added to hydrochlorothiazide, significantly decreased urinary calcium excretion in adults. 84 It appears that with potassium supplements, the hyponatremia may also get corrected to some extent. This correction is thought to be due to the shift of sodium from the intracellular to extracellular space, increasing the serum sodium concentration.85-87
Sodium inhibits calcium uptake by the membrane vesicles in the distal convoluted tubules. 88 So when sodium supplementation is used to treat thiazide-induced hyponatremia, it may lead to increased sodium concentrations in the distal tubular lumen and may result in increased urinary loss of calcium, thus minimizing the calcium sparing effect of thiazides.89,90
The calcium conserving property makes potassium chloride the preferred choice rather than spironolactone or sodium chloride to treat the diuretic-induced electrolyte disturbances like hypokalemia, hypochloremic metabolic alkalosis, and even mild hyponatremia. However, close monitoring of serum potassium levels is needed to prevent inadvertent hyperkalemia.
Since Arginine Cl requires hepatic conversion for full activity, infants with hepatic dysfunction may require alternate therapy. Monitor for hyperkalemia especially when combined with other conditions or medications like potassium chloride and ACE inhibitors that can cause hyperkalemia.
Summary
Chloride plays a vital role in several physiological functions in the body. Hypochloremia is associated with increased morbidity and mortality in adults and preterm infants as well. Due to possible role of hypochloremia in the morbidities associated with BPD such as nephrocalcinosis, hypochloremic alkalosis and poor growth, clinicians should be paying close attention to chloride levels to optimize the outcomes in preterm infants. Besides, the evidence suggests that hypochloremia may be associated with diuretic resistance, which makes maintaining chloride levels in the physiological range even more sense. Since sodium supplementation counters the renal absorption of calcium and lack of evidence for spironolactone role in diuretic therapy for BPD, alternative chloride supplements such as potassium or arginine chloride may need to be considered as the first line of therapy in the management of hypochloremia due to diuretic therapy.
Footnotes
Author Contributions
RKT: Review of the data and wrote a portion of the manuscript, compilation of data, and reference, review and submission of the manuscript in the required format; PA: conceive, design and wrote the portion of the manuscript that led to this submission, figure generation, review of the manuscript; SR: review of the data and wrote portion of the manuscript, review of the manuscript, and figure generation; ND: review of the data and wrote portion of the manuscript, review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
