Abstract
In this article, we report a case of tuberculosis spondylodiscitis in a 2-year-old child. Imaging of her spine showed a paraspinal abscess. The diagnosis of spinal tuberculosis remains difficult, and we discuss its salient features and current management within the pediatric population.
Introduction
According to the Global Tuberculosis Report 2017 by the World Health Organization, tuberculosis (TB) is the ninth leading cause of death worldwide, and the leading cause from a single infectious agent. In 2016, the estimated incidence of TB globally was 10.4 million, with an estimated 1.3 million TB deaths among human immunodeficiency virus–negative people, and an additional 374 000 deaths among human immunodeficiency virus–positive people. 1 Locally in Singapore, 2310 new cases of TB were reported in 2016, giving an incidence rate of 41.2 per 100 000 population. A total of 83.5% of the new cases had pulmonary TB with or without extrapulmonary involvement, while 16.5% had extrapulmonary TB exclusively. 2
Skeletal TB accounts for 1% to 2% of all TB cases, of which spinal TB contributes to approximately half of skeletal TB cases, followed by TB of the hip and the knee. The commonest site of spinal TB infection is the thoracic region, followed by the lumbar and the cervical regions.3,4 Spinal TB has been postulated to arise from hematogenous spread from a primary site, which is often unknown. Pulmonary TB is seen in less than 50% of cases with skeletal TB. Due to the high vascularity of the vertebral column, the spine is the commonest site of skeletal TB infection. In younger children, the disc is often involved because of the anastomosis between the vertebral endplate and disc.4,5 Hematogenous spread of the infection leads to destruction of the vertebrae body, and when extensive, may result in vertebral collapse. With further spread, the infection extends into the adjacent disc space, with contiguous infection of multiple adjacent vertebral bodies. 3 Other than bony changes, the infection may also spread into soft tissues and result in paraspinal abscesses. These may be large and displace adjacent structures, including abdominal organs. Chronic abscesses can result in fistulation to distant structures, resulting in intrathoracic or subcutaneous abscesses. 6
Case Report
A 2-year, 3-month-old female presented to our institution with a 4-month history of mid-back prominence and concerns of running slower than her peers. Further history revealed a previous febrile illness, 5 months prior to our consult. It was associated with upper respiratory symptoms, hip pain, and refusal to walk. At that point, she was seen and investigated by a pediatrician in the community. Her investigations showed a raised erythrocyte sedimentation rate (ESR) of 75 mm/h, an unremarkable full blood count, white blood cell count of 10.79 × 109/L, and C-reactive protein (CRP) of 14 mg/L. A plain radiograph of her affected hip was reported as normal, and she received symptomatic treatment with resolution of her fever and symptoms within 2 weeks. During our consult, there was no history of fever after she recovered from her initial febrile illness, respiratory symptoms, loss of weight or appetite, and night sweats. Her immunizations were up to date, including a dose of Bacille Calmette-Guérin at birth. There was no travel history, and no significant animal, soil, or contaminated water contact. However, she had significant contact history with her maternal grandfather from China, who had possible pulmonary tuberculosis. Two years ago, he had lived and cared for her for 4 months, during which he had a chronic cough. He was previously investigated in China, where a plain radiograph of his chest showed an opacity in the lung field and had not received any antituberculosis treatment.
On clinical examination, she was alert, afebrile, and comfortable at rest. She had a nontender, mid-back prominence at the thoracolumbar region of her spine. There were no overlying skin changes. Neurological examination and gait were normal, with no signs of cord compression. She did not have significant lymphadenopathy, and examination of the cardiovascular, pulmonary, and abdominal systems were unremarkable.
A plain radiograph of her spine showed scoliosis and kyphosis of 65° of the thoracolumbar spine (Figure 1a; open arrow). A magnetic resonance imaging (MRI) of her spine showed destruction from the 10th to 12th thoracic vertebrae (T10 to T12) with associated loss of anterior vertebral height and a gibbus deformity (Figure 1b; open arrow). There was also a 4.6 × 3.3 × 4.3 cm rim-enhancing collection spanning the 8th to 12th thoracic vertebrae (T8 to T12; Figure 1b; arrow), which was posteriorly displacing the dural sac, but with no evidence of severe cord compression or cord signal abnormality.

(a) Lateral radiograph of the spine shows kyphosis of the thoracolumbar region (open arrow). Sagittal (b) and axial (c) postcontrast magnetic resonance imaging of the spine reveals a rim-enhancing collection (arrow in b, arrow in c), with destruction of T11, and displacement of the dural sac posteriorly.
Our patient was subsequently admitted and underwent a computed tomography–guided drainage of the spinal collection, with frank pus aspirated. During her admission, she was referred to the infectious diseases specialist for further workup and management. Her full blood count, CRP, and ESR were unremarkable (hemoglobin level of 11 g/dL, total white blood cell count of 12.8 × 109/L, platelet count of 343 × 109/L, CRP 1.1 mg/L, and ESR 20 mm/h). Her liver function tests and renal panel were also unremarkable. A plain radiograph of her chest showed clear lung fields with no consolidation. Aerobic and aerobic blood cultures were negative. The pus which was drained showed polymorphs of 1+ with no bacterial growth, cytology was negative for malignant cells, and fungal smear and culture were both negative. Acid-fast bacilli (AFB) smear was positive 1+ from the pus drained, but negative for AFB culture; TB nucleic amplification test (SDA ProbTec) from the pus was positive, and interferon-γ release assay (IGRA; T-SPOT.TB) from blood was negative. Her T-SPOT.TB was not repeated.
As her T-SPOT.TB was negative, and results of her TB nucleic amplification acid test were initially pending, she was started on a 4-drug antituberculous regime (pyrazinamide, rifampicin, isoniazid, and ethambutol), as well as clarithromycin for possible nontuberculous mycobacteria infection. Clarithromycin was stopped, when her TB nucleic acid amplification (NAA) test returned positive.
During her outpatient consult with the infectious disease specialist, she received a tuberculin skin test, which was positive at 15 mm. Household contact screening was negative for tuberculosis infection. She was seen subsequently in 6 weeks, where she remained well with no anorexia or weight loss and was continued on her 4-drug antituberculous regime. She completed 8 weeks of pyrazinamide and ethambutol and will complete a total of 9 months of rifampicin and isoniazid. On follow-up with the orthopedic specialist, the gibbus deformity over her thoracolumbar region remained, and she measured less than the third centile for height. Thankfully, her gait and neurological examination were both normal. A repeat plain radiograph of her spine 1 month postdischarge showed a stable kyphosis. She is currently managed with a spine brace (Figure 2) and may possibly require posterior spinal instrumentation in the event of further vertebral collapse. Written consent for publication was obtained from the patient’s parents. This study was approved by Singhealth Centralised Institutional Review Board (CIRB Reference Number: 2015/2024).
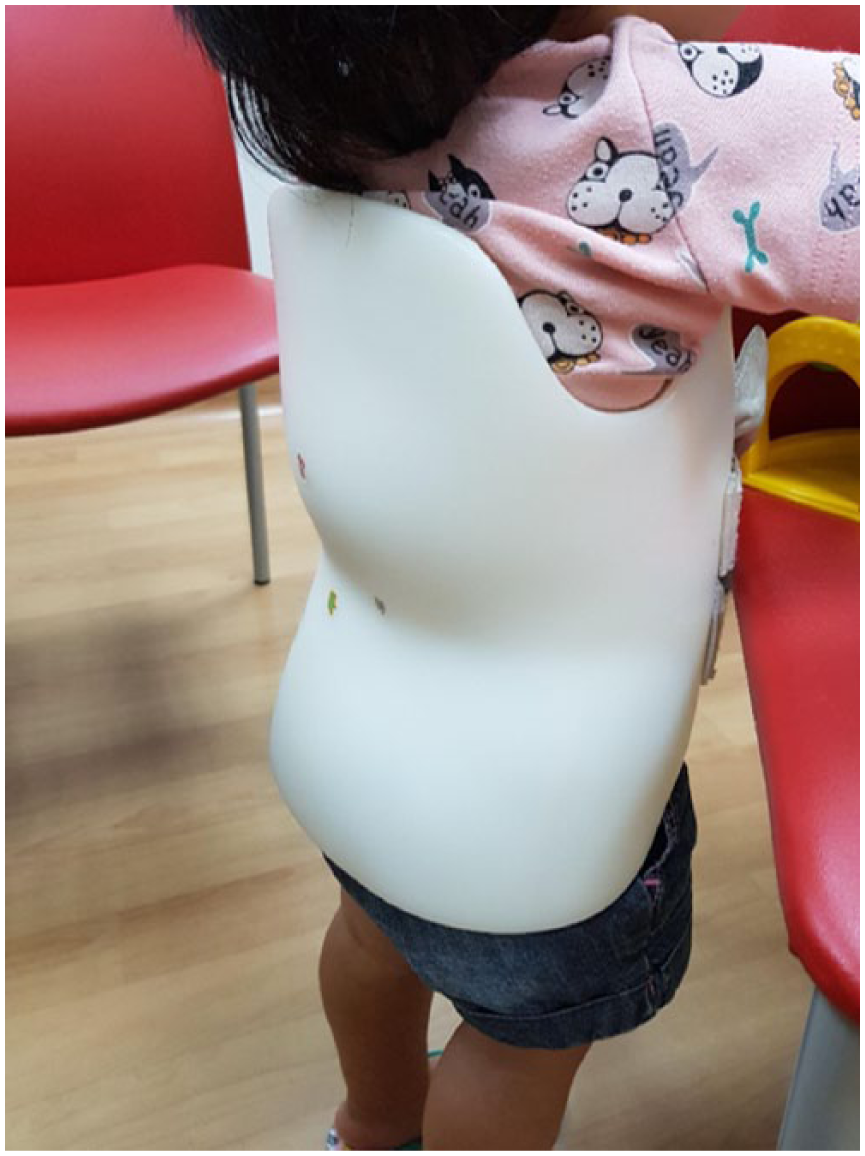
Our 2-year old patient with TB spondylodiscitis wearing a spinal brace during outpatient follow-up.
Discussion
In our young patient, her diagnosis of spinal TB was established through several modalities. She had a positive TB contact, a gibbus deformity, and an MRI that was suggestive of spinal TB. Further investigations that contributed to her diagnosis included a positive tuberculin skin test, AFB-smear positive specimen, and a positive NAA test for TB.
Presenting clinical features in children with spinal TB include systemic symptoms of fever, anorexia, weight loss, night sweats, as well as localized symptoms of back pain, spinal deformity, and neurological symptoms such as paraparesis.4,7,8 Due to relatively small-sized case series and reviews, the frequency of neurological involvement in children with spinal TB reported varies widely between 3% and 57%.7-10 Authors have previously reported a high rate of systemic involvement, and over half presenting with neurological symptoms—highlighting the importance of suspecting spinal TB in children with both systemic symptoms and back pain.4,7,11 However, as seen in our patient who presented with back pain and a gibbus deformity, without systemic symptoms, the diagnosis of spinal TB can be easily missed if thought of in association with systemic symptoms. Important aspects of history that should be sought include TB contact, previous TB, and travel history to places where TB is endemic, which proved valuable in our patient.
Radiological evidence of spinal TB is best seen with MRI, as it is able to demonstrate soft tissue masses, discs, and spinal cord involvement. It is able to detect early changes in the disease process and demonstrate the extent of soft tissue involvement. Precise demonstration of the level of cord compression also helps in planning for those who require surgical intervention. Plain radiographs are insensitive in detecting early changes, and only detect vertebral changes only when at least 50% of the vertebrae is destroyed.3,4,6 Late in the disease, plain radiographs may show extensive calcification, loss of vertebral body height, and bony erosions. Computed tomography imaging is useful in guiding percutaneous drainage, and demonstrating bony involvement of the posterior elements that are difficult to visualize on plain radiographs.3,4,6,12
Isolation of Mycobacterium tuberculosis complex by culture from the source of infection remains the gold standard, but is often negative in children—where isolation rate is fewer than 50% of children with pulmonary TB, and even less so in extrapulmonary TB. 13 As seen in our patient, a negative culture cannot be used to exclude TB, and other forms of diagnostic testing should be employed. Aside from isolation by culture, a task force supported by the American Thoracic Society, the Centers for Disease Control and Prevention, and the Infectious Diseases Society of America recommends performing AFB smear microscopy, NAA tests, and histological examination from sites of suspected extrapulmonary TB. 14 When evaluated in patients with suspected pulmonary TB, compared with AFB smear microscopy alone, the NAA increases the positive predictive value above 95% when performed in AFB smear-positive specimens and provides more confidence in ruling in the diagnosis of TB. 15 However, information regarding NAA testing for nonrespiratory specimens is limited, often reported as having lower specificities and sensitivities. 16 In our patient, in addition to an AFB smear-positive and a positive NAA test, her tuberculin skin test (TST) was positive at 15 mm. In a review of children with spinal TB in a major UK centre, 7 90% of their cohort had a positive TST, of which all were above 10 mm in diameter. Their median age was 9.7 years, and ranged from 3.4 to 15.9 years. Given the high positivity yield of a TST in this cohort, it should be considered as a valuable test in children with suspected spinal TB, especially in children below 5 years old, where testing with IGRAs may be less accurate.
QuanfiFERON-TB GOLD In-Tube and T-SPOT.TB are the 2 preferred IGRAs for detection of TB infection. The specificity of IGRAs is reported to be higher than TST in many clinical settings, as the antigens used are not found in Bacille Calmette-Guérin or most pathogenic nontuberculous mycobacteria. 13 However, published data demonstrate the consistency of IGRAs in children aged 5 years and older, with limited data on its use in children under 5 years of age, and concerns about inadequate sensitivity. It has been suggested that IGRA sensitivity is diminished in young children, possibly due to a blunted immune response in the young. 17 The nonreactive T-SPOT.TB in our patient, aged 2 years and 3 months, demonstrates the limitations of the use of IGRA in young children.
There is no single consensus on the treatment of spinal TB in children, and current recommendations are largely based on adult studies. In Singapore, our practice guidelines recommend a 9-month anti-TB regime for patients with musculoskeletal TB. 18 This includes an intensive phase of 2 months of rifampicin, isoniazid, pyrazinamide, and ethambutol, followed by a continuation phase of rifampicin and isoniazid. Other guidelines recommend regimes ranging between 6 and 12 months. Specifically, the 2016 National Institute of Health and Care Excellence guidelines on tuberculosis recommends a 6-month regime for treatment of spinal TB without central nervous system involvement 19 ; the 2016 Joint American Thoracic Society, the Centers for Disease Control and Prevention, and the Infectious Diseases Society of America guidelines cited evidence for anti-TB regimes between 6 and 12 months 20 ; the American Academy of Pediatrics recommends a duration of 9 to 12 months 3 ; while the World Health Organization, in their 2014, second edition of their guidance on the management of TB in children, recommend a 12-month regime. 21
There has been no clear evidence on the use of steroids in spinal TB, with some guidelines recommending its use in the presence of central nervous system involvement.12,19 Surgical intervention in the management of spinal TB is also unclear, with no recommendations from several guidelines,13,21 and recommendations for surgical consideration in the presence of spinal instability, ongoing deterioration, or need for cord decompression.19,20
Conclusion
Spinal TB in children is uncommon, and its diagnosis is often delayed. They may present with or without systemic symptoms associated with localized symptoms of back pain or spinal deformity. Complications of cord compression may also be present and should always be evaluated in all suspected cases of spinal TB. An MRI of the spine is crucial in its diagnosis, and provides important information regarding the disease extent and soft tissue involvement. There is no consensus on the treatment of spinal TB in children as yet, and at our institution, we employ an anti-TB regime of at least 9 months duration, with close follow-up to monitor its progress.
Footnotes
Author Contributions
JLKG: Contributed to design; contributed to analysis and interpretation; drafted manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. CYC: Contributed to analysis and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. SZRL: Contributed to analysis and interpretation; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. KBLL: Contributed to analysis and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy. NWHT: Contributed to conception and design; contributed to analysis and interpretation; critically revised manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
