Abstract
Long-acting reversible contraception, which include etonogestrel subdermal hormonal implants, has high efficiency rates in preventing teen pregnancy. Although these numbers are increasing, teens have the lowest long-acting reversible contraception usage rate of any group, which the American Congress of Obstetricians and Gynecologists addressed with recommendations for Nexplanon placement for teens. The American Congress of Obstetricians and Gynecologists has recommended support for increased use and training of practitioners and pediatricians to gain momentum. Safety remains a concern as increased usage by practitioners and pediatricians may lead to more adverse outcomes. Despite the improvements to Nexplanon as a preloaded insertion device to prevent deep placement and malalignment, there are few known published adverse outcomes. Although different products, a review of the adverse outcomes reported with Norplant removal may closely reflect today’s concerns with Nexplanon. The 3 case studies in this article are examples of similar adverse outcome with Nexplanon removal as those reported with Norplant.
Teen and young adults have higher rates of unintended pregnancy than do any other age group. According to the Centers for Disease Control and Prevention, sexual risk behavior rates of 41% are reported for high school students prior to graduating from high school. 1 There is an increased risk of poor health outcomes for both teen mothers and their children. This public health concern remains constant. Although there is a steady decline in unwanted teen and young adult pregnancy rates, there is an inverse rate of long-acting reversible contraception (LARC) use among teens and young adults. LARC use does not rely on user adherence for effectiveness, and despite an initial decline in the 1970s, there is resurgence in use since 2010 among all females, 2 making this a sound contraceptive option.
Long-acting reversible contraception, which include intrauterine devices (IUDs) and etonogestrel subdermal hormonal implants, are gaining popularity due to high efficiency rates in preventing teen pregnancy. 2 Use of IUDs since 2002, for adolescents and adults ages 15 to 44 years, has increased 83%, while implant use tripled (0.3% to 0.8%). 2 Although these numbers are increasing, teens have the lowest LARC usage rate of any group, 3 which the American Congress of Obstetricians and Gynecologists (ACOG) addressed with recommendations for both IUDs and Implanon placement for teens and young adult females. 4 The ACOG has recommended support for increased use and training of practitioners and pediatricians to gain momentum in adolescent and young adult access.5,6 Safety remains a concern as these recommendations for increased training and usage by practitioners and pediatricians may lead to more adverse outcomes with insertion and removal with these devices.
Despite the improvements to Nexplanon as a preloaded insertion device to prevent deep placement and malalignment, there are few known published adverse outcomes. Although different products, a review of the adverse outcomes reported with Norplant removal may closely reflect today’s concerns with Nexplanon, and these include implanted fibrous tissue, broken implants, deep placement or poorly aligned implant, placement into muscle tissue, and migration.7,8 The following 3 case studies are examples of how certified adolescent medicine providers have placed Nexplanon and identified similar adverse outcome with removal as those reported with Norplant (see Figures 1-4).
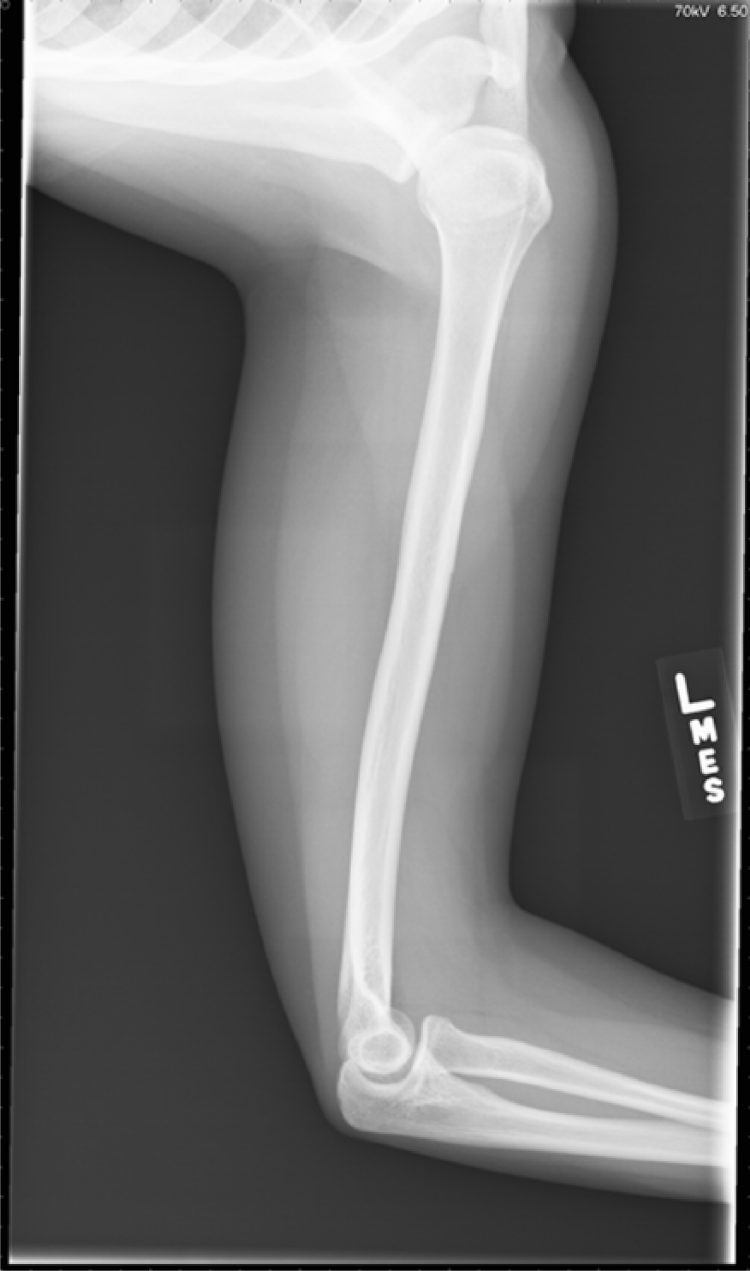
X-ray confirming thin radiopaque foreign body which projects over the anteromedial cortex of the mid left humeral diaphysis.

Fluoroscopic image of radiopaque foreign body projecting over the medial cortex of the right humerus

Nexplanon broken device.

Nexplanon bent device.
Case 1
A 17-year-old female with systemic lupus erythematous requested placement of Nexplanon for abnormal vaginal bleeding. She requested placement April 2014, by a certified and experienced adolescent medicine fellow. The patient maintained use of the Nexplanon for 8 months, at which time she requested removal due to persistent vaginal bleeding. Removal was complicated by an inability to palpate the device within the left arm. She was scheduled for ultrasound evaluation and removal, but the device was unable to be located with ultrasound imaging. A laboratory evaluation was completed that confirmed etonogestrel levels of 225.5 g/mL and the presence of the device. Imaging studies were requested with X-ray and the device was identified as a cylindrical radiopaque foreign body measuring 4 to 5 cm in length identified superficial to left bicep muscle at level of deltoid tuberosity. The device had migrated from the site of orginal placement.
General surgery was consulted for removal but due to the location of the Nexplanon intraoperatively, general surgery requested emergent assistance from a plastic surgeon as the device was located around the neurovascular components of the arm. The exact location of the device was not easily identified and fluoroscopy was utilized by plastic surgery for removal. The implant was located deep in the fascia within the bicep muscle and required dissection prior to removal. The procedure time in the operating room was more than 3 hours. There was discussion in regard to aborting the procedure and leaving the device in place as there were ongoing concerns for nerve damage, but due to the assistance of plastic surgery the device was safely removed (Figure 1 & 2).
Case 2
A 17-year-old female who requested Nexplanon insertion in December 2015 for contraception. Placement was completed by a certified gynecologist without event, but due to prolonged heavy vaginal bleeding removal was requested 6 months later. Removal occurred in the clinic exam room. Palpation of the device was obtained and removal was completed. On removal it was identified that the device had fractured into 2 pieces. The second piece was not immediately located at the insertion location, but superior to the original insertion site. Although there is documentation in the literature that the fracture of Nexplanon can occur during removal, this device was in 2 separate locations, which supported fracturing at insertion. Removal of the second fragment of the Nexplanon required repeat local anesthetic and a second incision for removal. The pieces were measured for completion and no further fragments were identified (Figure 3).
Case 3
A 20-year-old female who requested Nexplanon September 2012, for prolonged vaginal bleeding and contraception. This device was placed by a certified and experienced adolescent medicine fellow. Removal was requested due to persistent breakthrough vaginal bleeding and the 3-year expiration of contraception. The patient’s device was removed, but found to be bent, which prolonged timely removal. The device measurement was consistent with insertion measurements. No identified fragments were visualized or palpable. It was noted that the patient refused placement of a second implant and elected to return to Depo-Provera injections.
Although the etonogestrel subdermal hormonal implant is an effective form of contraception, specifically for the noncompliant adolescent who is sexually active with multiple partners, it may have limitations and complications for removal for the general practitioner. Placement may occur in the community, but removal may require centers of excellence and a surveillance protocol. Due to possible adverse outcomes with these devices, it may be valuable to implement a surveillance protocol to evaluate placement of the implanted device. The protocol would require yearly palpation of the device with measurement and imaging studies with ultrasound if unable to assess the device or if unable to adequately measure. Ultrasound-guided imaging should be used for removal if the device is not accessible on palpation in the clinic (Figure 4).
Footnotes
Acknowledgements
Acknowledge the staff in the Divisions of Adolescent Medicine and Adolescent Gynecology for their recommendations and providing unique cases toward this report.
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
