Abstract
Children and adolescents with long-term physical conditions (LTPCs) are at greater risk of developing psychosocial problems. Screening for such problems may be undertaken using validated psychometric instruments to facilitate early intervention. A systematic review was undertaken to identify clinically utilized and psychometrically validated instruments for identifying depression, anxiety, behavior problems, substance use problems, family problems, and multiple problems in children and adolescents with LTPCs. Comprehensive searches of articles published in English between 1994 and 2014 were completed via Medline, Embase, PsycINFO, CINAHL, and Cochrane CENTRAL databases, and by examining reference lists of identified articles and previous related reviews. Forty-four potential screening instruments were identified, described, and evaluated against predetermined clinical and psychometric criteria. Despite limitations in the evidence regarding their clinical and psychometric validity in this population, a handful of instruments, available at varying cost, in multiple languages and formats, were identified to support targeted, but not universal, screening for psychosocial problems in children and adolescents with LTPCs.
Introduction
More than 10% of children and adolescents worldwide are affected by long-term physical conditions (LTPCs), including asthma, diabetes, and epilepsy. 1 These individuals are more prone to a range of psychosocial problems including depression, anxiety disorders, behavior disorders, and posttraumatic disorder.1-9 The prevalence of formal psychiatric disorder in children with LTPCs is estimated at between 29% and 34%, 10 and pediatricians often lack the confidence to identify such disorders. 11 Medical complications of psychiatric problems include poorer treatment adherence, increased hospitalization, and the development of long-term complications.12,13 Although some studies have shown that children with LTPCs such as cancer can cope well,14,15 others have shown they experience more emotional and behavioral problems, even following the completion of treatment. 16
Children with LTPCs often minimize distress when asked directly, and parental depression, which is more common in such families, can contribute to the underreporting of children’s mental health symptoms by caregivers.17-20 Symptoms of psychological problems in these children are likely to overlap not just with each other but also with those of their physical conditions.21,22 For instance, somatic symptoms such as low energy, loss of appetite, and difficulty getting to sleep can be both features of depression and side-effects of chemotherapy. Even subclinical psychological symptoms in children with LTPCs can be associated with significant emotional and relational problems. 23 Early intervention requires the timely identification of psychosocial problems. 24 Despite World Health Organization criteria 25 being fulfilled for the screening of many such problems in this population, there are no well-known formal screening programs for identifying psychosocial difficulties in children and adolescents with LTPCs. Currently, psychosocial screening is often undertaken in pediatric settings using nonvalidated techniques such as HEEADSSS assessment. 26 Over the past few decades, a number of psychometric instruments have been developed to identify problems in single or multiple psychosocial domains. Many of these have been used in children with LTPCs, but their psychometric properties with this group have not formally been evaluated. 10
Previous reviews of psychometric instruments for identifying psychosocial problems in children and adolescents have focused on the clinical utility and psychometric properties of such instruments in the general population. Given that children and adolescents with LTPCs are a higher risk group and that cutoff scores designed for use with the general population may lead to an over- or underestimation of true rates of problems in this cohort, this systematic review was undertaken to identify psychometric instruments that have been used in studies of children and adolescents with LTPCs and to assess their utility as screening tools from both clinical and psychometric viewpoints. Specifically, this review was designed to identify suitable instruments for identifying (a) depression, (b) anxiety, (c) behavior problems, (d) substance use problems, (e) family problems, and (f) multiple problems in this clinical population.
Methods
Literature Search Strategy
Articles detailing the use of psychometric instruments for either identifying or measuring change in one or more of the 6 types of psychosocial problems mentioned above, that had been published in English between 1994 and 2014, were sourced via Medline, Embase, PsycINFO, CINAHL, and Cochrane CENTRAL databases accessed between December 20 and 31, 2014 (see the appendix); from reference lists of articles identified from the database searches; and from previous reviews of psychometric instruments for use with children and adolescents.27,28 Abstracts were reviewed by 2 authors (HT and HM), and complete articles were reviewed and a subset identified for data extraction and analysis by all 4 authors (HT, HM, KM, and KG). The study protocol was registered with PROSPERO on January 19, 2015 (Registration Number: CRD42015016021).
Evaluation of Instruments
Psychometric instruments were compared on the basis of clinical properties, including the type of LTPCs with which they had been tested, the time required for completion, available formats, and cost for their use. In addition, they were compared according to their psychometric properties within the child and adolescent LTPC population. Based on the recommendations of previous studies,27-29 the “ideal screening instrument” for each condition was expected to have been tested against a gold standard for screening or identifying cases of psychological disorder in one or more populations of children and adolescents with LTPCs (either an in-depth sophisticated clinical interview with an empathic and experienced interviewer or a scale that had been demonstrated to be as good as such an interview). It was also expected to possess good sensitivity (the probability of having a positive test result among those patients who have a positive diagnosis), specificity (the probability of having a negative test result among those patients who have a negative diagnosis), positive predictive value (the probability of having a positive diagnosis among those patients having a positive test result), and negative predictive value (the probability of having a negative diagnosis among those patients having a negative test result). Finally, it was expected to have good validity (eg, internal consistency Cronbach’s α > 0.8 29 ) and reliability (eg, interrater reliability > 0.4 30 ) and clear cut points for case identification in children and adolescents with LTPCs. As a meta-analysis was not planned, no formal assessment of risk of bias was undertaken.
Results
Results are presented in accordance with PRISMA guidelines. 31 A total of 4105 abstracts were extracted and reviewed using the search strategy described above, and 57 potential screening instruments were identified (Figure 1). Of these, 13 instruments were subsequently excluded as they were found to either have been used only in children without LTPCs or adult populations, or because they only included quality of life measures. Forty-four suitable scales were evaluated as outlined in Table 1. Further details regarding these scales can be found via the manuals and websites listed in Table 2.

PRISMA flow chart.
Clinical and Psychometric Properties of Identified Instruments.
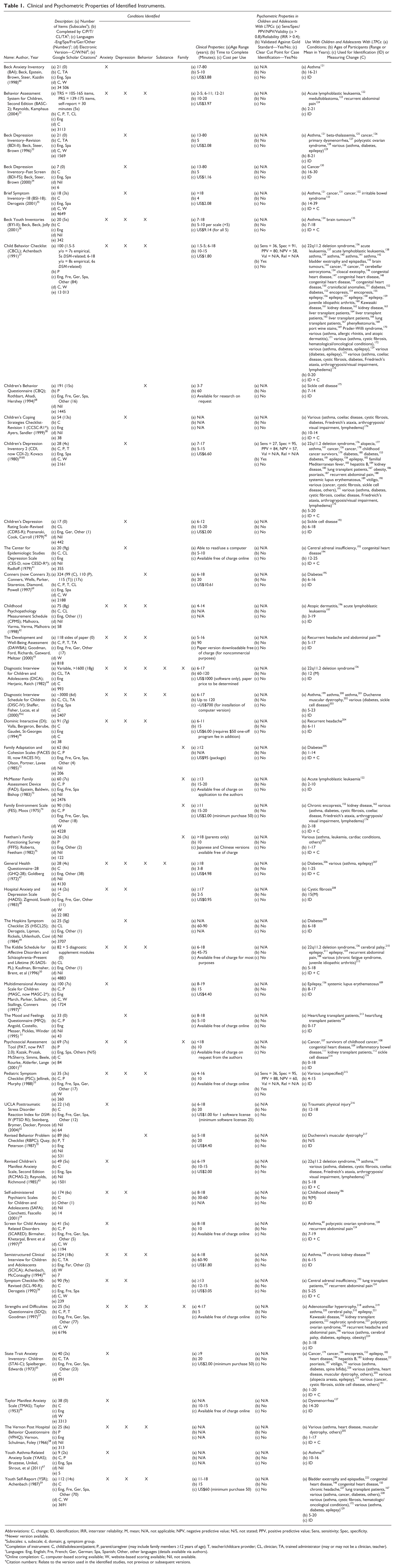
Abbreviations: C, change; ID, identification; IRR, interrater reliability; M, mean; N/A, not applicable; NPV, negative predictive value; N/S, not stated; PPV, positive predictive value; Sens, sensitivity; Spec, specificity.
Newer version available.
Subscales: s, subscale; d, domain; g, symptom group.
Completion of instrument: C, child/adolescent/patient; P, parent/caregiver (may include family members ≥12 years of age); T, teacher/childcare provider; CL, clinician; TA, trained administrator (may or may not be a clinician, teacher).
Languages: Eng, English; Fre, French; Ger, German; Spa, Spanish; Other, other languages (details available via authors).
Online completion: C, computer-based scoring available; W, website-based scoring available; Nil, not available.
Citation numbers: Relate to the version used in the identified studies, not previous or subsequent versions.
Key Websites or References for Identified Instruments.

Newer version available.
Depression
Twenty-eight instruments for identifying depression in children and adolescents with LTPCs were found by our search (Table 1). These included the BASC-2, 32 BDI-II, 33 BDI-FS, 34 BSI 18, 35 BYI-II, 36 CBCL, 37 CCSRC-R1, 38 CDI, 39 CDRS-R, 40 CESD, 41 CPMS, 42 DAWBA, 43 DICA, 44 DISC-IV, 45 DI, 46 GHQ-28, 47 HADS, 48 HSCL 25, 49 K-SADS-PL, 50 MFQ, 51 PAT, 52 PSC, 53 SAFA, 54 SCICA, 55 SCL-90-R, 56 SDQ, 57 VPHQ, 58 and YSR. 59 Of these, the only instruments to have been psychometrically investigated by Canning 10 in a single sample of 112 children and adolescents with multiple LTPCs, aged 9 to 18 years from a tertiary care medical center in the United States, were the CBCL, CDI, and PSC, all of which were compared with the DISC-IV intensive structured clinical interview as a gold standard. In this study, all 3 instruments demonstrated low sensitivity, positive predictive value, and negative predictive value, but high specificity.
Anxiety
Twenty-eight instruments for identifying anxiety in children and adolescents with LTPCs were identified by our search (Table 1). These included the BAI, 60 BASC-2, 32 BYI-II, 36 CBCL, 37 CPMS, 42 DAWBA, 43 DICA, 44 DISC-IV, 45 DI, 46 GHQ-28, 47 HADS, 48 K-SADS-PL, 50 MASC, 61 PAT, 52 PSC, 53 PTSD RI, 62 RCMAS, 63 SAFA, 54 SCARED, 64 SCICA, 55 SCL-90-R, 56 SDQ, 57 STAI-C, 65 TMAS, 66 VPHQ, 58 YAAS, 67 and YSR. 59 None of these instruments had been validated as a screening tool for anxiety in the target population, either against a gold standard or other instrument. Nor had any sensitivity, specificity, positive predictive values, or negative predictive values been reported by any of the authors of these studies.
Behavior Problems
Eighteen instruments for identifying behavior problems in children and adolescents with LTPCs were found by our search (Table 1). These included the BASC-2, 32 BYI-II, 36 CBCL, 37 CBQ, 68 Conners, 69 CPMS, 42 DAWBA, 43 DICA, 44 DISC-IV, 45 DI, 46 GHQ-28, 47 K-SADS-PL, 50 PSC, 53 RBPC, 70 SCICA, 55 SDQ, 57 VPHQ, 58 and YSR. 59 Of these, the CBCL, SDQ, and YSR were the most commonly used, and only the CBCL had specifically been validated with this population. 10
Substance Use Problems
Only 2 instruments for identifying substance use problems in children and adolescents with LTPCs were found by our search, namely, the DICA 44 and DISC-IV 45 (Table 1). Neither of these instruments was purpose-designed as an instrument for rating substance use problems and both identified these issues as part of a broader DSM-IV 71 aligned assessment process in research settings. Neither instrument had been validated as a screening tool for substance use problems in the target population, either against a gold standard or other instrument, and no sensitivity, specificity, positive predictive values, or negative predictive values have been reported by any of the authors of these studies.
Family Problems
Seven instruments for assessing family problems were identified by our search, namely, the DICA, 44 FACES III, 72 FAD, 73 FES, 74 FFFS, 75 PAT, 52 and SDQ 57 (Table 1). None of these instruments had been validated as a screening tool for family problems in the target population, either against a gold standard or other instrument. Nor had any sensitivity, specificity, positive predictive values, or negative predictive values been reported by any of the authors of these studies.
Multiple Problems
Of the instruments we found, the DICA 44 was the only one that identified all 5 types of problem, namely, depression, anxiety, behavior, substance use problems, and family issues. The DISC, 45 GHQ-28, 47 and SDQ 57 being broad screening instruments identified 4 of these problems (the first two excluding family issues, the third excluding substance use problems). The combination of depression, anxiety, and behavior problems was identified by the BASC-2, 32 BYI-II, 36 CBCL, 37 CPMS, 42 DAWBA, 43 DI, 46 K-SADS-PL, 50 SCICA, 55 VHPQ, 58 and YSR. 59 The combination of depression, anxiety, and family problems was identified by the PAT. 52 Overall, none of our identified instruments proved to be a clinically viable instrument for easily identifying all of these problem areas in children and adolescents with LTPCs.
Discussion
Children and adolescents with LTPCs remain at greater risk of developing psychosocial problems. Despite enthusiasm from public health and funding bodies to routinely identify and address common childhood mental health problems as early as possible in high-risk groups,76-79 there is inadequate evidence to recommend doing so using currently available psychometric instruments.80,81 Targeted screening using some of these tools is probably more valid. Of the 44 potential instruments evaluated by us, none met the criteria for an “ideal screening instrument” outlined prior to the commencement of the review and most had only had confirmation of their psychometric properties within the general population.
Previous reviewers of psychometric instruments for children and adolescents have had varying views, as outlined below, partly due to differences in focus and partly due to when their reviews were undertaken. Myers,27,82 Brookes, 28 Stocking, 83 and Quittner 84 have conducted the most comprehensive reviews of instruments for identifying depression and anxiety. Myers 82 recommended the Reynolds Adolescent Depression Scale (RADS 85 ) and Reynolds Child Depression Scale (RCDS 86 ) for the identification of depression in the general population, and a combination of the clinician-rated CDRS-R 40 and patient-rated CDI-2 87 for identifying depression in clinical populations, the latter instruments being more sensitive to clinical change. Both Brookes 28 and Stocking 83 identified significant limitations in the KSADS, 50 DISC, 45 DICA, 44 BDI, 33 Hamilton Depression Rating Scale (HDRS 88 ), and Montgomery Asberg Depression Rating Scale (MADRS 89 ) for identifying depressive symptoms, and the BDI-II, 33 CDI-2, 87 CES-D, 41 and RADS 85 in identifying “caseness.” A recent consensus statement on the identification of anxiety and depression in children and adolescents with cystic fibrosis 88 recommended that the Patient Health Questionnaire (PHQ-9 90 ) should be routinely used to screen children with the condition over the age of 12 years as it is brief, reliable, has valid optimal cutoff scores for detecting psychological symptoms that map onto DSM-5 91 criteria, and is free and available in all major languages. Unfortunately, no studies of children and adolescents with LTPCs using the PHQ-9 were identified by our search, leaving us unable to comment on this recommendation. The BDI-FS 34 was designed for “evaluating symptoms of depression in patients reporting somatic and behavioral symptoms that may be attributable to biological, medical, alcohol, and/or substance abuse” and has been shown to be better than the PHQ-9 at discriminating between depressive and somatic symptoms. 92 Although most studies have focused on its use in primary care and only one study in children with LTPCs was identified by us, it shows some promise.
Myers and Brookes favored the MASC and SCARED for identifying anxiety, due to their clear constructs, adequate internal psychometric properties, ability to discriminate between anxiety and depression, response formats that should detect treatment effect, short screening forms, and parallel parent-report forms. Myers and Brookes disagreed on the value of the RCMAS 63 and STAI C, 65 with the latter favoring these instruments. Brooks and Kutcher additionally identified the CBCL, 37 K-SADS-PL, 50 and ADIS-C/P 93 as viable instruments for detecting anxiety. Quittner recommended the GAD-7 94 for identifying anxiety in children over the age of 12 years with cystic fibrosis.
Comprehensive reviews of instruments for identifying behavior disorders in children and adolescents95,96 have previously recommended the Conners, 69 Swanson Nolan and Pelham IV Questionnaire (SNAP-IV), 97 Attention Deficit Disorder Evaluation Scale (ADDES-2 98 ), and ADHD Symptom Rating Scale (ADHD-SRS 99 ) for identifying combined/hyperactive symptoms of ADHD; the Brown Attention Deficit Disorder Scale (BADDS 100 ) for identifying inattention; the Eyberg Child Behavior Inventory (ECBI 101 ), the Sutter-Eyberg Student Behavior Inventory–Revised (SESBI-R 102 ), and the New York Teacher Rating Scale for Disruptive and Antisocial Behavior (NYTRS 103 ) for assessing broad constructs of disruptive behavior disorder; and the Antisocial Process Screening Device (APSD 104 ) for evaluating youth with conduct disorder.
A number of well-validated, specific, and brief instruments exist for identifying substance use problems in young people including the CRAFFT 105 substance abuse screening test, recommended by Pilowsky 106 following a recent review of screening instruments for adolescent substance abuse in primary care settings; the Personal Experience Short Questionnaire (PESQ 107 ), recommended by Farrow 108 during a similar review for the Washington State Division of Alcohol and Substance Abuse; and newer instruments such as the Substances and Choices Scale (SACS 109 ) and the Teen Addiction Severity Index (T-ASI 110 ). Despite their lack of use and psychometric validation with children and adolescents with LTPCs, their specific design for identifying substance use problems, cost, and ease of use probably make them better choices for the targeted identification of such problems in clinical settings compared with the DICA 44 or DISC-IV. 45
The FACES III, 72 FAD, 73 FES, 74 and FFFS 75 were exclusively designed to assess family functioning, and despite lack of psychometric validation in children and adolescents with LTPCs, they had all been shown to be of some clinical use in this population. Out of all the identified instruments, the PAT 2.052,111,112 is the most extensively researched and promising screening instrument for systemic issues within families of children and adolescents with LTPCs. It is linked to a triaging system, based on the Pediatric Psychology Preventative Health (PPPH) model 113 to ensure appropriate referrals are made, and information provided to the treating team. It has been researched in families of children with conditions such as cancer, 52 congenital heart disease, 114 inflammatory bowel disease, 115 and kidney transplants. 116 While it has shown good discrimination in terms of family and parental psychosocial difficulties and behavior problems, it has not specifically been researched as a screener for childhood or adolescent anxiety or depression.
This review provides a snapshot of instruments that have been used in children and adolescents with LTPCs and some information regarding their nature. There are a number of other considerations to be factored in when deciding which screening instruments to use for identifying psychosocial problems in this population, when to use such instruments, and how to do so. All scales are not built equal. Briefer scales such as the MFQ designed for quick identification of conditions are less comprehensive, but more practical to use in clinical settings than comprehensive assessment questionnaires such as the DISC-IV. 117 Although clinician-rated scales have been shown to be more accurately predict outcomes than self-report scales, the former are more commonly used, are more relevant to patient-centered care, 118 and the 2 scales are best used in combination for optimum result. Newer scales are more accurate than older scales, particularly in discriminating between overlapping constructs such as anxiety and depression. 29 However, the former have a longer track record and clinicians may be more familiar with them. If identification of “cases” rather than symptoms is important, checklists that are aligned with diagnostic manuals such as the DSM-5 71 are probably more useful than those that rate symptoms continuously using different paradigms. Online or electronically available scales allow for efficient data analysis, but can be costly and off-putting for those with less familiarity with technology. Finally, acceptability and validity of scales in different languages and cultures is important to establish as some instruments such as the GAD7 have been shown to be less accurate in some groups (eg African Americans) than others. 119
Limitations of this review include the fact that only instruments used in studies of children and adolescents with LTPCs were included in the main analysis and other newer and potentially useful scales that have been not similarly researched may have been excluded. In addition, few instruments had psychometric data pertaining to the target population and assumptions of efficacy had to be made for most instruments based on their properties within the general population. Strengths of the review include the wide range of LTPCs with which identified instruments had been used and the correlation of our findings with those of key reviews of these instruments in the wider population to enable recommendations for clinicians and researchers that are based on the most up-to-date evidence.
Overall, in our opinion, the best instruments identified by us for targeted screening for psychosocial problems in children and adolescents with LTPCs are as follows. For depression, the clinician-rated CDRS-R 40 and patient-rated CDI-2, 87 BDI, 32 and PHQ-9 90 are the easiest to use and best regarded instruments, with the BDI-FS 34 showing promise. For anxiety, the self/parent-rated MASC-2, 61 SCARED, 64 and GAD-7 94 all have satisfactory appeal. Behavior problems are best identified using the parent-rated SDQ 57 and CBCL, 37 and ADHD is best identified using the self/parent/teacher-rated Conners-3. 69 Substance use problems are best screened for using the well-established self-rated CRAFFT 105 and PESQ 107 or newer but easier to use scales such as the SACS 109 and T-ASI. 110 Family problems are best identified using the parent-rated PAT 2.0, 52 and finally, depending on their combination, multiple problems may be screened for using a limited range of instruments including the parent-rated BASC-3, 32 SDQ, 57 and PAT 2.0. 52
Just as important as screening is what comes after it. Care pathways and provision of high-quality care should be in place before the implementation of any targeted or universal screening programme. 120 Future research should include more in-depth evaluation of existing instruments in children and adolescents with LTPCs and the development of more specific instruments for identifying psychosocial problems in this population.
Conclusions
For now, clinicians should continue to be vigilant regarding the greater likelihood of psychosocial problems in children and adolescents with LTPCs and should only use recommended instruments in a targeted manner to support clinical judgment within an established continuum of care.
Author Contributions
HT: Contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; agrees to be accountable for all aspects of work ensuring integrity and accuracy.
HM: Contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript.
KG: Contributed to analysis and interpretation; drafted the manuscript; critically revised the manuscript.
KM: Contributed to conception and design; contributed to acquisition, analysis, and interpretation; drafted the manuscript.
Footnotes
Appendix
Acknowledgements
The authors thank Anne Wilson for her assistance with data extraction and the Starship Foundation New Zealand for supporting some of KG’s time on this project (Grant number SF985).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Part of this study was funded by the Starship Foundation New Zealand, Grant SF985.
