Abstract
Hepatic glycogen storage diseases (GSDs) are genetic diseases associated with fasting hypoglycemia. Periodic intake of uncooked cornstarch is one of the treatment strategies available for those disorders. For reasons that are still not clear, patients with hepatic GSDs may be overweight.
Aims:
To assess nutritional status and body composition in patients with hepatic GSDs receiving uncooked cornstarch.
Methods:
The sample included 25 patients with hepatic GSD (type Ia = 14; Ib = 6; III = 3; IXα = 1; IXβ = 1), with a median age of 11.0 years (interquartile range [IQR] = 9.0-17.5), matched by age and gender with 25 healthy controls (median age = 12.0 years, IQR = 10.0-17.5). Clinical, biochemical, and treatment-related variables were obtained from medical records. Nutritional status and body composition were prospectively evaluated by bioelectrical impedance.
Results:
Patients and controls did not differ with regard to age and gender. Height was significantly reduced in patients (median = 1.43 m, IQR = 1.25-1.54) in comparison to controls (median = 1.54 m, IQR = 1.42-1.61; P = .04). Body mass index for age z-score and fat mass percentage were higher in patients (median = 1.84, IQR = 0.55-3.06; and 27.5%, IQR = 22.6-32.0, respectively) than in controls (median = 0.86, IQR = −0.55 to 1.82; P = .04 and 21.1%, IQR = 13.0-28.3; P = .01, respectively). When patients were stratified by type, those with GSD Ia had significantly higher adiposity (median fat mass = 28.7%, IQR = 25.3-32.9) than those with GSD III and GSD IXα/β (median fat mass = 20.9%, IQR = 14.9-22.6; P = .02).
Conclusions:
Our findings suggest that patients with hepatic GSD on treatment with cornstarch, especially those with GSD Ia, exhibit abnormalities in nutritional status and body composition, such as short stature and a trend toward overweight and obesity.
Keywords
Introduction
The glycogen storage diseases (GSDs), or glycogenosis, are a group of genetic diseases characterized by abnormalities in enzymes that regulate glycogen synthesis and degradation. 1 Glycogen, stored mainly in muscle and liver tissue, serves as a readily accessible source of energy 2 to maintain glucose homeostasis during fasting. 3
Different GSD types exist and are classified accordingly to the organs affected, with a broad range of clinical manifestations, and gene/enzyme involved (Table 1). In hepatic GSDs, endogenous glucose production is impaired; thus, the main clinical consequence is fasting hypoglycemia. 1 Hepatomegaly is another common manifestation shared across nearly all GSD types. Symptoms vary depending on the specific form of GSD including hyperlactatemia, hyperlipidemia, hyperuricemia, cardiomyopathy, abnormal liver enzymes, hypotonia, and cirrhosis. 1,2,10,11
Classification of Hepatic Glycogen Storage Diseases and Summary of Their Characteristics.a
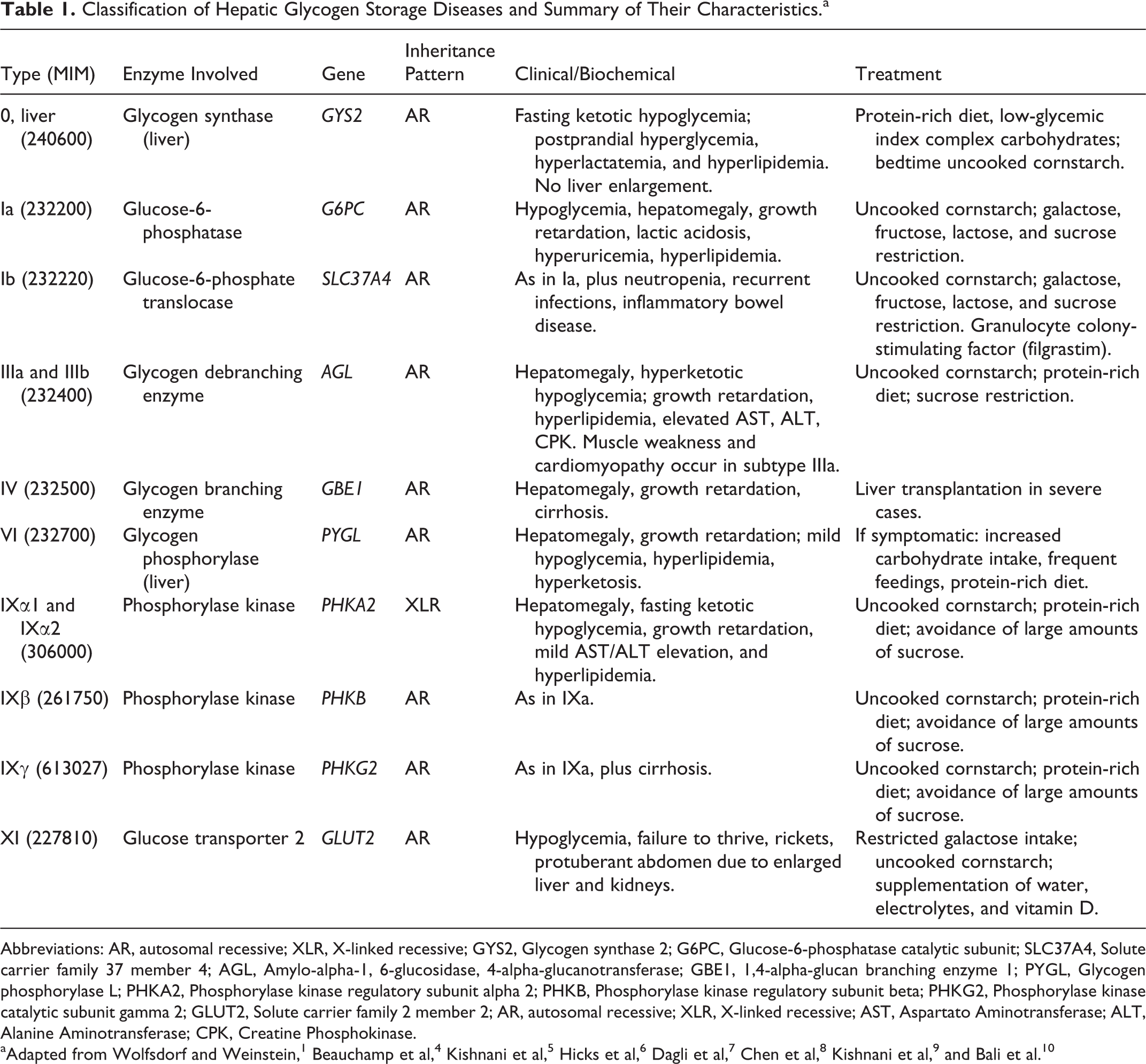
Abbreviations: AR, autosomal recessive; XLR, X-linked recessive; GYS2, Glycogen synthase 2; G6PC, Glucose-6-phosphatase catalytic subunit; SLC37A4, Solute carrier family 37 member 4; AGL, Amylo-alpha-1, 6-glucosidase, 4-alpha-glucanotransferase; GBE1, 1,4-alpha-glucan branching enzyme 1; PYGL, Glycogen phosphorylase L; PHKA2, Phosphorylase kinase regulatory subunit alpha 2; PHKB, Phosphorylase kinase regulatory subunit beta; PHKG2, Phosphorylase kinase catalytic subunit gamma 2; GLUT2, Solute carrier family 2 member 2; AR, autosomal recessive; XLR, X-linked recessive; AST, Aspartato Aminotransferase; ALT, Alanine Aminotransferase; CPK, Creatine Phosphokinase.
The primary treatment goal in hepatic GSDs is to prevent hypoglycemia, avoid long-term complications, and ensure adequate growth. 12 One of the treatment approaches is dietary and may involve frequent, periodic administration of uncooked cornstarch (UCCS) and continuous nocturnal gastric drip feeding (CNGDF). 13 Liver transplantation can be considered in cases of severe hepatic cirrhosis, hepatic dysfunction, and/or hepatocellular carcinoma, 7 and kidney transplantation when there is renal insufficiency. 10
Considering the clinical heterogeneity of hepatic GSD, dietary recommendations need to be adapted to the needs of each type of diseases and the age of the patient. The nutritional recommendations for GSD I, III, and IX are presented in Table 2. Daily caloric intake should be prescribed and monitored closely, as insufficient energy provision will be unable to correct metabolic derangements and, consequently, leads to growth impairment. On the other hand, excessive UCCS intake may lead to obesity. 12
Dietary Recommendations for Hepatic GSD I, III, and IX.a
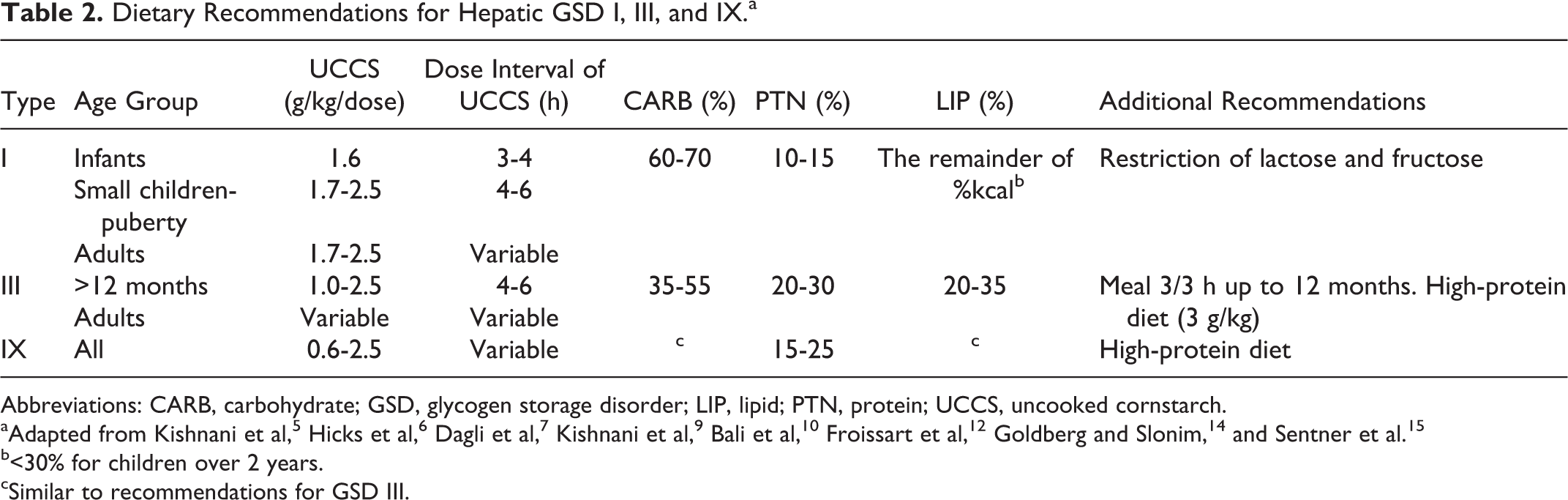
Abbreviations: CARB, carbohydrate; GSD, glycogen storage disorder; LIP, lipid; PTN, protein; UCCS, uncooked cornstarch.
aAdapted from Kishnani et al, 5 Hicks et al, 6 Dagli et al, 7 Kishnani et al, 9 Bali et al, 10 Froissart et al, 12 Goldberg and Slonim, 14 and Sentner et al. 15
b<30% for children over 2 years.
cSimilar to recommendations for GSD III.
There are several reports of overweight in patients with hepatic GSDs, particularly in GSD Ia, 9,16,17 however, the etiology of this finding remains incompletely elucidated. On the one hand, dietary treatment with a complex carbohydrate can reduce the risk of short-term and long-term complications, 18 on the other hand, it could be associated with overweight and, eventually, excess adiposity. Short stature has also been reported in GSD types I, III, and IXα/β. 2,4,6,11,19
Although changes in nutritional status appear to be frequent among patients with hepatic GSDs, knowledge gaps remain regarding these aspects as well as regarding the body composition of these patients. Within this context, the present study sought to evaluate nutritional status and body composition in patients with hepatic GSDs in comparison to healthy controls, through a review of clinical data and bioelectrical impedance analysis (BIA), in an attempt to improve the current understanding of the association of these aspects with GSD.
Methods
Study Design
This cross-sectional study used a convenience sampling strategy to enroll patients with hepatic GSDs and healthy controls matched for age and gender. All patients were recruited from the Medical Genetics Service at Hospital de Clínicas de Porto Alegre (SGM-HCPA), Brazil. The 25 healthy controls had a median age of 12.0 years (interquartile range [IQR] = 10.0-17.5) and were recruited from the community of the same hospital. This study was approved by the HCPA Research Ethics Committee (protocol number: 14-0120) and conducted in accordance with the provisions of the Declaration of Helsinki. All participants or their legal guardians signed an informed consent term.
Sample
The sample included 25 patients receiving UCCS treatment, with a median age of 11.0 years (IQR = 9.0-17.5), of whom 14 had GSD Ia, 6 had GSD Ib, 3 had GSD III, and 2 had GSD IXα/β (Tables 3 and 4). All patients included in this study had a diagnosis of hepatic GSD confirmed by enzymatic and/or genetic analysis (median age at diagnosis = 8.5 months; IQR = 6-21.5) and are seen at the Outpatient Metabolic Disorders Clinic at SGM-HCPA (median age at treatment onset = 12 months; IQR = 6-35.2) every 3 to 6 months. Clinical variables (GSD type and comorbidities), biochemical parameters (serum lactate, glucose, triglycerides, and cholesterol levels), and treatment parameters (dose of UCCS administered) were obtained through a review of the medical records for the patients’ latest visits (up to 3 months before inclusion in the study).
Nutritional Status and Body Composition in Patients With Hepatic Glycogenosis Receiving Uncooked Cornstarch Therapy and Controls—Summary of the Main Findings.a
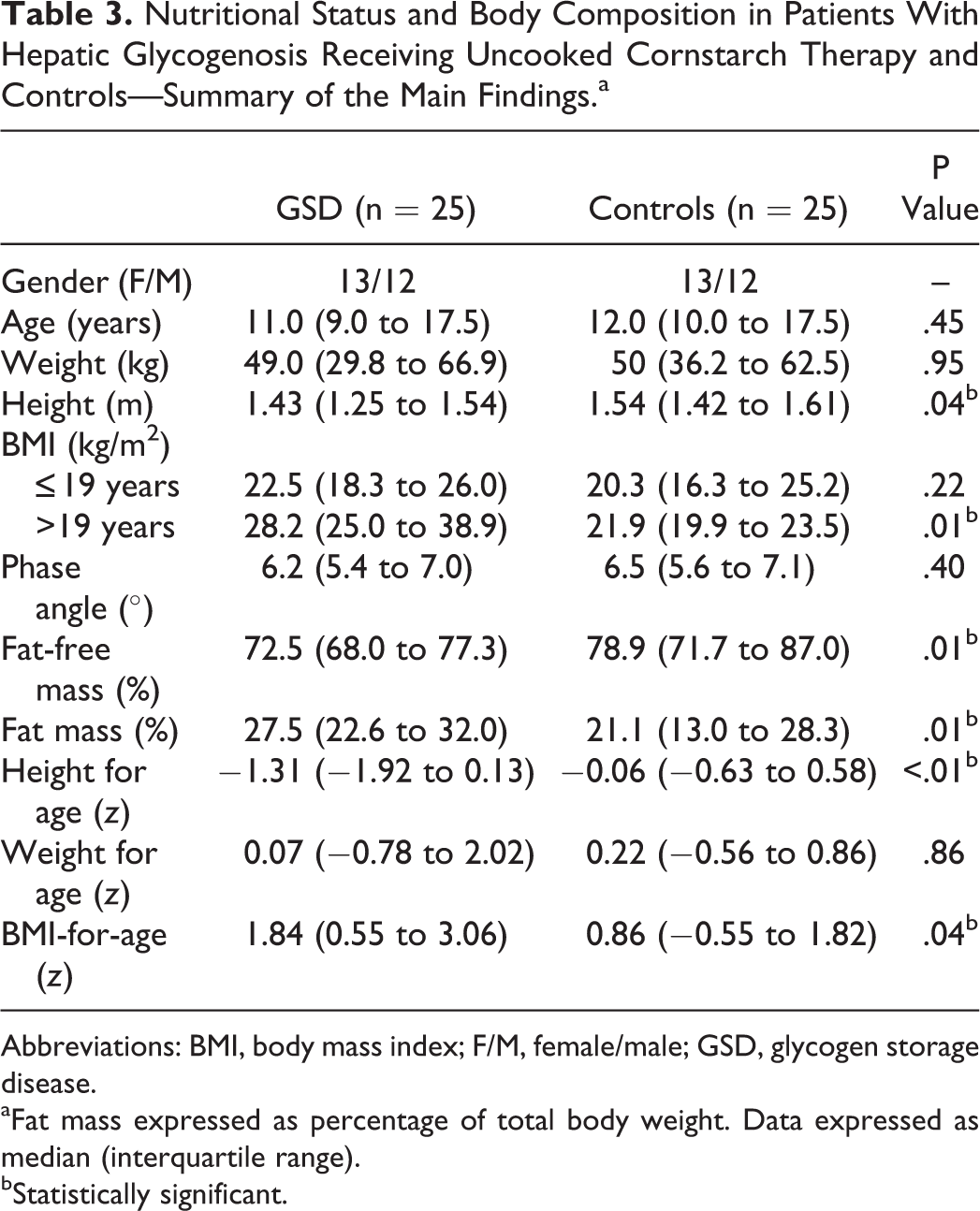
Abbreviations: BMI, body mass index; F/M, female/male; GSD, glycogen storage disease.
aFat mass expressed as percentage of total body weight. Data expressed as median (interquartile range).
bStatistically significant.
Nutritional Status and Body Composition According to the Type of Hepatic Glycogenosis—Summary of the Main Findings (N = 25).a
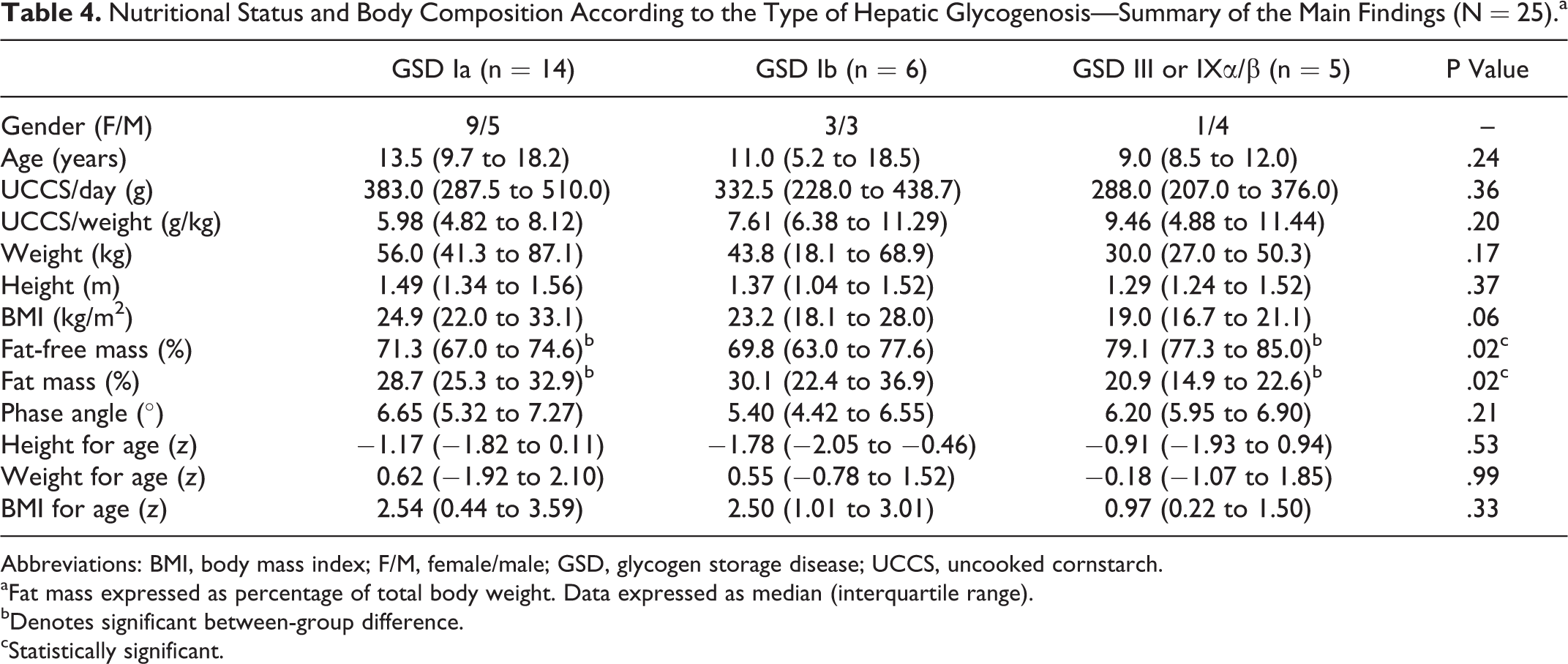
Abbreviations: BMI, body mass index; F/M, female/male; GSD, glycogen storage disease; UCCS, uncooked cornstarch.
aFat mass expressed as percentage of total body weight. Data expressed as median (interquartile range).
bDenotes significant between-group difference.
cStatistically significant.
Anthropometric Measurements
Weight was measured using digital scales (resolution 0.1 kg; Model 2096PP/2; Toledo, São Paulo, Brazil), while height was measured with a wall-mounted stadiometer (precision 0.1 cm; Harpenden; Holtain, Crymych, Wales, United Kingdom). These measurements were obtained while participants were in standing position and wearing minimal clothing. Body mass index (BMI) was calculated as weight (kg) divided by height in meter square (m2) and classified as underweight, normal weight, overweight, or obese, per the World Health Organization (WHO) criteria. 20,21 In patients aged <19 years, nutritional status was calculated using BMI for age z scores calculated in WHO Anthro version 3.2.2 and WHO Anthroplus version 1.0.4. 22,23
Bioelectrical Impedance Analysis of Body Composition
To estimate body composition (fat mass and fat-free mass), a BIA system was used (Biodynamics 450, version 5.1; Biodynamics Corporation, Seattle, Washington), with resting-tab electrocardiogram electrodes (Conmed Corporation, Utica, New York), as described elsewhere in the literature. Participants were instructed to refrain from physical activity or intake of caffeine-enriched foods and beverages in the 8 hours preceding assessment as well as to empty their bladders and remove all metal objects before starting the test. 24 Due to the need to maintain normal glucose levels, patients could not be asked to fast for 8 hours. Therefore, a 3-hour fast was defined as standard for all patients. Controls underwent the same preparation and were given the same instructions but with an 8-hour fast. During the test, participants remained in the supine position, with the limbs outstretched away from the body, while 4 electrodes were applied in the following distribution: 1 on the wrist, 1 on the hand, 1 on the ankle, and 1 on the foot (all on the right side of the body). The adiposity (percentage fat mass) was calculated according to the Obesity Medicine Association 25 reference values for adults and McCarthy et al 26 reference values for children. Bioelectrical impedance analysis also provided data on phase angle (PA), a variable derived from the relationship between resistance (R), reactance (Xc), and impedance (Z) (PA = arctang Xc/R) to the passage of a low-amplitude, high-frequency (50 kHz) electrical current during the procedure. 27 The cutoff of Bosy-Westphal et al 28 was used to evaluate PA.
Statistical Analysis
All analyses were carried out using SPSS Statistics, version 21.0 (IBM Corp, Armonk, New York). Categorical variables were expressed as absolute and relative frequencies, and continuous variables, as medians and IQRs. The Mann-Whitney U test was used for comparison between the case and control groups. The Kruskal-Wallis test, followed by a post hoc analysis, was used for comparison between subgroups of patients with different GSD types. Spearman coefficients were used to test for correlation. The significance level was set at 5%.
Results
The 25 patients included belonged to 24 unrelated families. The rate of parental consanguinity was 20.8%. Four patients (16%) had clinical diagnosis of inflammatory bowel disease; all had a diagnosis of GSD type Ib and were overweight or obese at the time of assessment. Three patients (12%), all with GSD Ia, were being treated for hypothyroidism; of these, 2 (67%) had excess weight and 1 had normal weight.
Median daily UCCS intake was 6.48 g/kg body weight (IQR = 5.11-9.97), for a median total of 360 g/day (IQR = 265-447). The UCCS doses were administered every 3 to 6 hours. The median total cholesterol and triglyceride levels in plasma, available for 24 patients, were 167.5 mg/dL (IQR = 146.2-206.0) and 197.0 mg/dL (IQR = 119.7-407.5), respectively. The median serum glucose level was 86.5 mg/dL (IQR = 80.5-95.2, n = 22), while the median lactate level was 1.78 mmol/dL (IQR = 1.22-2.69, n = 20).
Table 3 reports anthropometric parameters and body composition data for patients and controls. Bioelectrical impedance analysis revealed that 16 (64%) patients (3 with normal weight, 3 overweight, and 10 obese) and 7 (28%) controls (1 with normal weight, 2 overweight, and 4 obese) had excess adiposity. Eight (32%) patients (6 with GSD Ia and 2 with GSD Ib) and 5 controls (20%) had PA values below the cutoff. Phase angle correlated strongly with fat-free mass in controls (r s = 0.7; P < .01) and moderately in patients (r s = 0.4; P = .03). Figure 1 shows the distribution of nutritional status in the sample, stratified by BMI classification. 20,21 High rates of excess weight (overweight = 28%; obesity = 40%) were found among patients. Conversely, most controls had normal weight (60%), while 9 had excess weight (overweight = 20%; obesity =16%).
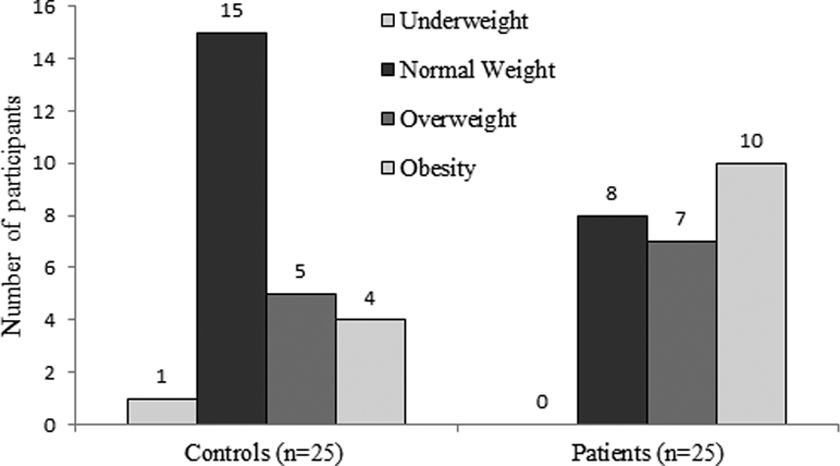
Stratification of patients by GSD type revealed a significant difference only for fat mass percentage; in this sample, patients with GSD Ia had greater adiposity than those with GSD III or IXα/β (Table 4).
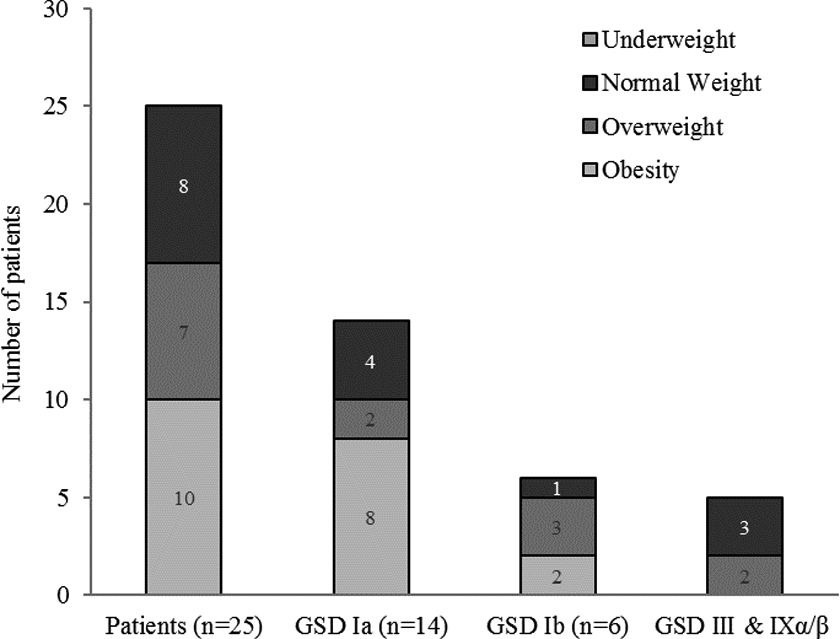
Regarding stratification of nutritional status by GSD type (Figure 2), we found a heterogeneous distribution of BMI values: 2 of 5 patients with GSD III and IXα/β were overweight, while 5 out of 6 with GSD Ib and 10 out of 14 with GSD Ia had excess weight (overweight or obesity).
Discussion and Conclusion
This was the first study to evaluate body composition by BIA in patients with hepatic GSD on treatment with cornstarch. We chose BIA for evaluation of body composition because it is a noninvasive, low-cost, user-friendly method widely employed in clinical practice. Bioelectrical impedance analysis is a predictive technique for assessment of body composition whereby the passage of a low-amplitude, high-frequency electrical current through the body, particularly through the lowest-resistance compartment, allows direct measurement of resistance and reactance values, which are used to calculate impedance and PA; finally, it estimates total body water content, extracellular and intracellular water, fat-free mass, and fat mass. 29 –31
In a review, Andreoli et al 32 suggested that, compared to other techniques, BIA is a good tool for estimation of body composition in isovolemic patients. The estimated error for prediction of total body water content and fat-free mass by BIA is 2% to 4%. To achieve optimal precision or even improve the precision of this method, it is essential that measurement conditions be standardized. 33
It is well-known that nonfasting state interferes with BIA findings. However, patients with GSD cannot be deprived of food for long periods without risking hypoglycemia. According to Kyle et al, 24 food and drink intake 2 to 4 hours before the test can reduce body impedance by 4 to 15 Ω, which represents an error of less than 3%. Thus, although an 8-hour fast is recommended, shorter periods can be acceptable both in clinical practice and in research settings.
A high frequency of obesity in patients with GSD was found by Chen et al 16 in a study of 13 GSD I patients in treatment (UCCS or CNGDF) for more than 5 years. The results of this evaluation showed that 10 of 13 patients were classified as obese according to percentage of ideal body weight for length percentile. Similar findings were described by Santos et al 17 in a cross-sectional study that evaluated patients with GSD I treated with UCCS, z scores of BMI for age showed that 16 of 21 patients were overweight. Short stature has also been reported by several authors as common problems in the hepatic GSD. 2,4,6,11,19
In agreement with the literature, our findings showed that most patients had excess weight and adiposity, with higher BMI, BMI for age z scores, and body fat percentage than controls as well as lower median height than controls. Although anthropometric parameters revealed a stature deficit in the patient group, as in the Weinstein and Wolfsdorf 18 study, the median z score for height (−1.31; IQR = −1.92 to 0.13) was within the expected range according to WHO standards. 21 The pathophysiology of short stature in hepatic GSDs has yet to be elucidated. However, studies suggest that the carbohydrate overload for treatment can contribute not only to excess weight 5,10,12,34 but also to better height scores in these patients. 17,18 Although our treated patients had a short stature, suggesting that the use of UCCS does not heal the low height.
Comparison of body composition revealed that fat mass was significantly greater in patients than in controls overall and was significantly greater in patients with GSD Ia than in patients with GSD III and IXα/β (P = .02). Patients with GSD III and IXα/β were those who most resembled controls in terms of fat mass percentage.
Body mass index is widely used for assessment of nutritional status, but the underestimation of obesity is a major limitation of this method. 35 In 1 study, BIA was used to evaluate body composition in 1244 European children, from 2004 to 2006. The authors concluded that, in the pediatric population, fat-free mass and fat mass measurements provide a better understanding of growth and changes in body composition than BMI. 36 This discrepancy was also observed in our sample, in which excess adiposity was identified even in some patients classified as having normal weight by BMI.
Patients with GSD Ia, Ib, and III/IX were not found to differ regarding the daily amount of UCCS received (in g or in g/kg), but patients with GSD Ia were found to have higher fat mass than patients with GSD III/IX. So, although many authors suggest that excess weight and body fat in patients with GSD are associated with UCCS excessive intake, 5,10,12,34 our findings did not confirm this hypothesis. Another factor that may have been associated with excess weight, although not evaluated in this study, is the low engagement in physical activity. Uncertainty to whether physical activity is allowed and what dietary changes would be required may contribute to higher rates of sedentary behavior in this population.
Another BIA parameter that plays an important role in assessment of nutritional status is PA. 37 –39 A positive correlation between PA and fat-free mass has been described both in healthy individuals and in pathological conditions. 40 –42 We observed similar correlation in our control sample but not in patients with hepatic GSD, despite statistical significance (P = .03). Therefore, we suggest that PA is not a good predictor of fat-free mass in hepatic GSDs.
This parameter has also been associated with inflammation 43 and may be useful in the assessment of patients with hepatic GSDs, particularly GSD Ib, because of their greater susceptibility to inflammatory conditions. 44 There was no difference in PA between patients and controls, however, the median PA was lower in patients with GSD Ib than in patients with other types of GSD, suggesting possible predictive value in this subpopulation.
In conclusion, our findings suggest that Brazilian patients with hepatic GSDs have a shorter median height than healthy controls but still within normal range, and a tendency toward overweight and obesity, as demonstrated both by BMI (especially in patients with GSD type I) and by BIA-measured adiposity. Prospective studies with larger samples, taking into account measurement of anthropometric parameters as well as adiposity, may contribute to the further elucidation of the impact of these conditions on nutritional status and height. Furthermore, we believe greater research attention should be given to achieving a better understanding of the heterogeneous impacts of different GSD types on nutritional status, which could help prevent comorbidities associated with excess weight and adiposity and, thus, improve patient’s quality of life.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by CNPq (Call for Proposals No. 31/2013—Metabolic and Endocrine Diseases), FAPERGS (PPSUS/2013), and the Hospital de Clínicas de Porto Alegre Research and Event Promotion Fund (FIPE/HCPA).
