Abstract
Background:
All-suture anchors (ASAs) significantly reduced gap formation and increased strength compared with traditional fixation in a transosseous cadaveric quadriceps tendon repair model. However, the biomechanical performance of ASAs for unicortical quadriceps tendon repair has not been evaluated.
Hypothesis:
ASAs would reduce gap formation and exhibit comparable stiffness, yield load, and ultimate load to knotless hard-body suture anchors (SAs) for unicortical quadriceps tendon repair.
Study Design:
Controlled laboratory study.
Methods:
Eight matched pairs of male cadaveric knees were used to compare ASA and SA control repairs. Distal quadriceps tendons were prepared with 2 Krackow sutures using 1.3-mm suture tape. Sutures were fixed unicortically with 2 anchors using 2.6-mm ASAs or 4.75-mm SAs. Knees were actuated from 5° to 90° of flexion via the quadriceps tendon for 10 native preconditioning cycles and 250 cycles after repair at 0.1 Hz, with a peak force of 150 N per cycle. Repairs were loaded to failure at 50 mm/min. Gap formation (mm) during cyclic loading and stiffness (N/mm), yield load (N), and ultimate load (N) during load to failure were statistically compared between groups using paired t tests.
Results:
The ASA repair had significantly less gap formation at cycle 250 compared with the control repair (Δ = 4.2 mm [95% CI, 2.9 to 5.3 mm]; P < .001) and survived greater cycles before reaching a critical gap threshold of 5 mm (200 vs 50 cycles). No significant differences in stiffness or ultimate load were found between repairs. The yield load was significantly greater in the control group than the ASA group (Δ = 55 N [95% CI = 27-82 N]; P = .002).
Conclusion:
Unicortical quadriceps tendon repair with ASAs reduced gap formation by 43% compared with hard-body SAs. No significant differences in stiffness or ultimate load were observed, although a significantly greater yield load was found for the SA controls.
Clinical Relevance:
ASAs are a biomechanically viable alternative to hard-body SAs for unicortical quadriceps tendon repair, potentially resulting in less gap formation.
Keywords
Gap formation is a significant concern after quadriceps tendon repair, as it can lead to poor healing, functional deficits, and an increased risk of rerupture. 2 Transosseous patellar bone tunnels (TO) and suture anchor (SA) fixation are the most common approaches to quadriceps repair to restore knee extensor function, with comparable biomechanical and clinical outcomes.2,10,14 Consequently, there is no consensus on the optimal surgical technique for quadriceps tendon repair. Regardless, failure rates range from 0% to 15% for both TO and SA repairs and typically require substantial rehabilitation time. 16
Alternative fixation techniques have been developed to improve repair strength and limit gap formation. One such approach, using knotless SA fixation with suture tape, demonstrated improved mechanics and reduced gap formation compared with traditional TO and knotted SA repairs. 6 Adjustable techniques that enable intraoperative tensioning and retensioning may improve repair quality, as the use of adjustable-loop metal cortical fixation buttons for TO repair resulted in significantly less gap formation compared with a traditional SA repair. 4 When adjustable loops were utilized with knotless all-suture anchors (ASAs) in a similar TO technique, a reduction in gap formation and an increase in biomechanical strength were observed compared with traditional TO repairs, while also reducing the risk of potential hardware irritation from metal buttons. 15 Although adjustable ASA buttons demonstrated improved biomechanics in a TO approach, the biomechanical integrity of unicortical SA fixation with adjustable ASAs has not been explored.
This study aimed to compare the biomechanical integrity of quadriceps tendon unicortical fixation with ASAs to traditional hard-body SAs in a time-zero cadaveric biomechanical model. The authors hypothesized that ASAs would reduce gap formation and exhibit stiffness, yield load, and ultimate load comparable to those of hard-body SAs for unicortical quadriceps tendon repair.
Methods
The methods used to evaluate unicortical quadriceps tendon fixation were replicated from a recently published study evaluating transosseous ASA quadriceps tendon fixation and are summarized herein. 15 Eight matched pairs (n = 8 per group) of fresh-frozen human male cadaveric knees (mean age, 47.1 years; range, 40-53 years) procured from a registered tissue bank (Science Care) were assessed for bone deformity or previous injury. Institutional review board approval was not required for cadaveric research by the authors’ institution. The consistency of the bone quality of each patella was quantitatively measured at the anterosuperior aspect using a microindentation hardness probe (OsteoProbe; Active Life Scientific). Specimens were thawed to room temperature for 24 hours for preparation, frozen at −20°C, and rethawed for preparation and testing. Samples were stored at 4°C for intermittent periods of nonuse and acclimated to room temperature for testing.
All soft tissue proximal to the femoral epicondyle was dissected except for the quadriceps tendon on the patella. The tendon was secured 4 cm proximally to the patellar insertion with a whipstitched nylon strap for later quadriceps loading. Hanging weights (range, 1.5-3.5 kg) were added to the tibia and attached through a hole drilled 14 cm distally to the tibial plateau to compensate for the remainder of the missing lower extremity. A mean force of 150 ± 10 N was generated to reflect the quadriceps tendon force required to actuate the full leg into extension.1,9,11
Unicortical Repair Techniques
Unicortical repair techniques were randomized within each matched pair. Quadriceps tendon pathology was simulated by creating a full-thickness defect with a sharp scalpel, temporarily sparing 2 mm of tendon bilaterally to help stabilize the tendon for anatomic tendon reduction before severing. A traction of 10 N was applied through the nylon strap throughout the repair. The distal quadriceps tendon was sutured with 2 bilateral Krackow stitches using 1.3-mm suture tape (SutureTape; Arthrex), each with 4 passes, for both repairs (Figure 1).

Final constructs for the (A) suture anchor control and (B) all-suture anchor unicortical quadriceps tendon repair techniques. Krackow sutures were converted through the 2.6-mm all-suture anchor using preloaded shuttling sutures, before final knot tying at the distal tendon.
For the control repair, the distal Krackow sutures were secured with two 4.75-mm hard SAs (BioComposite SwiveLock; Arthrex) through predrilled and tapped 4-mm holes, such that the holes were equidistant from the center of the tendon insertion (Figure 1A). For the ASA repair, two 2.6-mm drill holes were made at the superior pole of the patella using a guide (Arthrex), with spacing similar to that of the control technique. The 2.6-mm ASAs (Knee FiberTak Button; Arthrex) were then inserted into these holes and set at the superior patellar cortex. The ASAs had 2 preloaded shuttle sutures used to convert the distal Krackow sutures through the anchor body and to reduce the tendon in a tension-slide manner. After achieving reduction, the tensioning sutures were tied through the distal tendon with a surgeon's knot and 5 alternating half-hitches (Figure 1B).
Biomechanical Testing
The biomechanical testing methods were identical to those of Wittstein et al 15 for cyclic and load-to-failure testing. For cyclic testing, specimens were mounted onto a custom fixture at the base of a servohydraulic materials testing system (Model 8871; Instron), with the transepicondylar axis parallel to the base and the tibia unconstrained in a neutral position. The proximal quadriceps tendon was oriented along its anatomic axis and attached to a 5-kN load cell with a pulley system for cyclic flexion-extension actuation. For load-to-failure testing, the patella and quadriceps tendon were isolated from the full knee and distracted until failure under uniaxial tension using a custom patellar and soft-tissue grip on an electromechanical materials testing system (Model E3000; Instron) with a 5-kN load cell.
Briefly, native specimens were preconditioned to 5° to 90° of flexion for 10 cycles at 0.1 Hz. After repair, passive fiducial markers with adhesive were applied to the tendon and patella for optical tracking (ARAMIS 3D 12M; Zeiss), with a minimum of 7 markers on each tissue. Next, the knee underwent 250 loading cycles from 5° to 90° of flexion at 0.1 Hz, reaching peak cyclic forces of 150 ± 10 N. After cycling, the quadriceps tendon was preloaded at 20 N for 30 seconds before loading to failure at 50 mm/min. A gauge length of 4 cm was used, defined as the distance between the superior pole of the patella and the grip point on the tendon.
Cyclic loading outcomes included tendon-bone gap formation (mm) measured at 90° of flexion at cycles 1, 10, 50, 100, 150, 200, and 250. Gap formation was measured as the mean difference in displacement between the distal tendon and proximal patella. A critical gap threshold of 5 mm was established to represent clinical healing impairment.7,12 Load-to-failure outcomes included stiffness in the linear elastic region (N/mm), yield load (0.2% offset from the linear elastic region; N), and ultimate load (N). Observed failure modes were recorded for all specimens.
Statistical Analysis
Paired t tests were used for comparisons of bone quality, gap formation, stiffness, yield load, and ultimate load. All data were normally distributed, as confirmed with the Shapiro-Wilk test. All analyses were performed in SigmaPlot (Version 14.0; Systat Software). The level of significance was set at α = .05. The sample size was consistent with Wittstein et al, 15 which showed significant differences and adequate power (>80%) for total cyclic gap formation (mean Δ = 6.3 mm; SD = 2.0 mm) when comparing ASA with transosseous quadriceps tendon repair.
Results
There was no significant difference in bone quality scores between matched pair knee patellae (P = .065); therefore, all were included in the paired statistical analysis.
Gap Formation
Unicortical ASA fixation resulted in significantly less gap formation (P < .01) than the control at all measured cycles (Figure 2, Table 1). The mean control unicortical fixation specimens surpassed the 5-mm defined critical threshold for gap formation within the first 50 cycles, whereas the mean unicortical ASA specimens approached the critical threshold at cycle 200. Gap formation at cycle 250 was (mean ± SD) 5.2 ± 0.9 mm for the ASA and 9.4 ± 1.4 mm for the control (Δ = 4.2 mm; 95% CI = 2.9 to 5.3 mm).
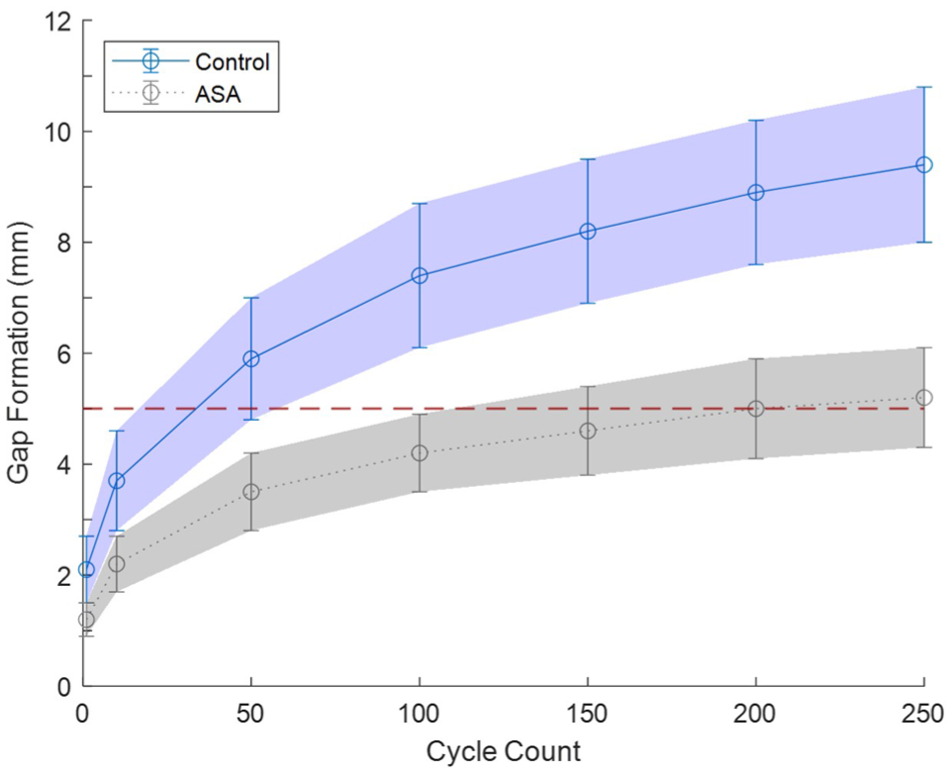
Tendon-bone gap formation through loaded knee flexion cycles using an ASA or control unicortical quadriceps tendon repair technique. A clinically important gap threshold of 5 mm (red line) is indicated to reflect a level of impaired healing.7,12 Data are presented as mean ± standard deviation. ASA, all-suture anchor.
Tendon-Bone Gap Formation Through Loaded Knee Flexion Cycles a

Data are presented as mean ± standard deviation. Bold P values indicate statistical significance (P < .05). ASA, all-suture anchor.
Load-to-Failure
There were no significant differences in stiffness or ultimate load between groups; however, the yield load was significantly lower in the unicortical ASA group compared with the control (mean Δ = 55 N; P = .002) (Table 2, Figure 3). The primary failure mode for the unicortical ASA group was anchor pullout (6/8), whereas suture slippage from the anchor was the primary failure mode in the control group (7/8). Additional failure occurred by suture cutout at the quadriceps tendon in 2 samples of the ASA group and by suture rupture at the anchor in 1 sample of the control group.
Load-to-Failure Analysis for Quadriceps Tendon Repair Using the Control or ASA Unicortical Technique a

Data are presented as mean ± standard deviation, unless otherwise indicated. The bold P value indicates statistical significance (P < .05). ASA, all-suture anchor.

Unicortical quadriceps tendon repair (A) stiffness, (B) yield load (0.2% offset), and (C) ultimate load box plots for control and ASA techniques. ASA, all-suture anchor.
Discussion
The primary finding of this study evaluating quadriceps tendon repair with unicortical fixation was that ASAs reduced gap formation compared with traditional hard-body SAs. No significant differences in stiffness or ultimate load were observed between the ASA and hard-body SA fixation, although a significantly lower yield load was observed in the ASA group.
The outcomes collected at time zero in this study provide insight into how unicortical quadriceps tendon repairs may influence postoperative healing during rehabilitation. To stabilize the healing enthesis, gapping or elongation during cyclic loading should be minimized, with a threshold of 5 mm previously established.7,12 In the present study, the SA control group reached this threshold by cycle 50, whereas the ASA group extended the number of loading cycles to 200. At 250 cycles, the ASA technique significantly reduced overall gapping by 4.2 mm compared with the SA technique, indicating superior maintenance of the tendon-bone interface under physiologically-relevant loading. Importantly, the time-zero test model created worst-case conditions in which other static and dynamic stabilizers were absent, and healing was not considered; thus, the values presented here are greater than would be expected clinically.
Load-to-failure testing also revealed how the unicortical quadriceps repair constructs respond to forces within and beyond their elastic limits. The linear stiffness in the elastic region showed no significant differences between the SA control (71 ± 5.9 N/mm) and ASA (69.3 ± 13.2 N/mm) repairs. Stiffness is a measure of the force response under tendon lengthening. An overly stiff tendon can predispose the tissue to tearing. In contrast, an overly compliant (i.e., low stiffness) tendon can place the muscle outside of its optimal function zone and negatively impact muscle strength recovery. 8 Stäubli et al 13 found that 10-mm strips of the native quadriceps tendon-patellar bone complex had an elastic modulus of 304 N/mm2, or a stiffness of 475 N/mm. As expected, neither repair fully restores native tendon function before biological healing. The yield load, which describes the transition from elastic to plastic (i.e., unrecoverable) lengthening, was higher in the SA control (312 ± 68 N) than in the ASA (257 ± 52 N) in the load-to-failure step, with both exceeding the 150 N required for active range of motion. However, cyclic loading at 150 N peak force demonstrated consistently greater unrecoverable elongation in the SA control compared to the ASA, with both groups reaching steady state behavior before the load-to-failure step. Thus, the yield load finding is likely an artifact of eliminating initial lengthening at the suture-tendon interface during cyclic loading. Lastly, the ultimate load did not significantly differ between the SA control (615 ± 92 N) and the ASA (562 ± 164 N). Collectively, the unicortical repairs provide a factor of safety ≥3.75 for ultimate load relative to the 150 N required for active range of motion.
While this study is the first to evaluate the biomechanical integrity of unicortical quadriceps tendon fixation with ASAs, our results are comparable to those of previous biomechanical studies. Kindya et al 6 reported a gap formation of 4.9 mm for a knotless SA technique and 7.4 mm for a conventional knotted SA technique at 250 cycles with 100 N peak force. In contrast, Gould et al 4 reported a plastic gap of 5.6 ± 1.7 mm at 200 cycles with a peak force of 250 N for a knotless SA technique. In the present study, the gap formation at 250 cycles with a peak force of 150 N was 9.4 ± 1.4 mm for the knotless SA and 5.2 ± 0.9 mm for the ASA. The greater gap formation in the SA in the present study is likely a consequence of using a smaller suture tape (1.3 mm) compared with previous studies (2 mm). The 1.3-mm suture tape used in the present study was selected to comply with the ASA requirements. For ultimate load, Kindya et al 6 reported 579 ± 129 N for the knotless SA and 399 ± 87 N for the knotted SA, the former of which is comparable to the ultimate loads found for both the SA and ASA groups in our study.
The biomechanical results of the current study suggest that unicortical ASA fixation may be a suitable alternative when selecting the appropriate procedure for patients. Although ASAs demonstrated superior resistance to gap formation compared with hard-body SAs for unicortical fixation, the gap formation and ultimate load were less favorable than with transosseous ASA fixation. Using identical methods to ours, Wittstein et al 15 reported a gap of 250 cycles of 3.6 ± 1.5 mm for transosseous ASA fixation versus 5.2 ± 0.9 mm for unicortical ASA fixation in the present study. Similarly, they found an ultimate load of 712 ± 113 N for transosseous ASA fixation, compared with 562 ± 165 N for unicortical ASA fixation, in the present study. Collectively, the results suggest that ASA fixation—whether unicortical or transosseous—provides clear biomechanical advantages over traditional techniques. However, higher-demand patients may benefit more from transosseous versus unicortical ASA fixation due to greater resistance to gap formation and greater repair strength.
ASA techniques, such as the one presented herein, enable intraoperative tensioning and retensioning to remove slack after joint cycling, thereby optimizing reduction at the patellar tendon interface and preventing gap formation. Because gap formation increases the risk of rerupture, conservative rehabilitation is typically required with current transosseous and traditional SA repair techniques. Furthermore, ASAs cause less bone loss than traditional SAs due to their smaller size, potentially reducing the risk of patellar fractures associated with transosseous tunnels. 5 The results suggest that ASA quadriceps tendon repairs may enable an earlier postoperative return to mobilization by establishing and maintaining the patella-tendon interface. Future clinical studies are warranted to understand the influence of ASA technology on patient outcomes and rehabilitation.
Limitations
The time-zero biomechanical nature of the study does not account for the stabilizing effects of biological healing. The quadriceps tendon was sharply transected at the patellar level, which does not exactly match the clinical scenario. The test methods also did not include other static and dynamic soft-tissue stabilizers that may protect the repair. The results may not reflect performance across age and sex, but do reflect the demographic most impacted by quadriceps tendon rupture. 3
Conclusion
Unicortical quadriceps tendon repair with ASAs reduced gap formation by 43% compared with hard-body SAs. No significant differences in stiffness or ultimate load were observed, although a significantly greater yield load was found for the SA controls.
Footnotes
Final revision submitted December 5, 2025; accepted December 27, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: The study was funded by an Arthrex grant No. AIRR-0209. B.C.W. has received speaking fees from Arthrex; consulting fees from Arthrex and LifeNet; and research support from Arthrex. M.D.T. is an employee of Arthrex. J.R.W. has received speaking fees from Arthrex and Vericel; consulting fees from Arthrex and Geistlich; and has stock or stock options in Viewfi. R.M.F. has received speaking fees from AlloSource, Arthrex, JRF, and Ossur; consulting fees from Allo-Source, Arthrex, and JRF; research support from Arthrex and Smith & Nephew; and publishing royalties and financial or material support from Elsevier. A.N.K. is an employee of Arthrex. M.K.E. is an employee of Arthrex. B.L.S. is an employee of Arthrex. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
