Abstract
Background:
Outcomes after arthroscopic rotator cuff repair (RCR) are frequently measured through clinically significant outcomes (CSOs) such as the minimal clinically important difference, the substantial clinical benefit, and the Patient Acceptable Symptom State. Global achievement of CSOs is challenging to predict.
Purpose:
To determine if unsupervised machine learning can identify distinct patient subgroups based on CSO achievement after elective arthroscopic RCR.
Study Design:
Case-control study; Level of evidence, 3.
Methods:
A prospectively collected database was analyzed to identify patients who underwent elective arthroscopic RCR from 2015 to 2017. Tear dimensions were measured on magnetic resonance imaging utilizing a validated technique. CSO achievements on the American Shoulder and Elbow Surgeons, the Single Assessment Numeric Evaluation, and the Constant-Murley subjective score at 2-year follow-up were calculated. An unsupervised random forest algorithm was utilized to develop and internally validate patient subgroups with significantly different rates of CSO achievement. Patient subgroup membership, along with a total of 30 demographic and clinical variables, as well as preoperative patient-reported outcomes, were incorporated into a stepwise multivariable logistic regression to identify factors predictive of optimal CSO achievement.
Results:
A total of 346 patients (192 male; mean ± SD age, 57.2 ± 9.1 years; body mass index, 30.1 ± 5.4 kg/m2) were eligible for inclusion and followed for a mean of 3.8 years (range, 2.0-6.2 years) Of these, a total of 333 patients were partitioned by the random forest algorithm into 2 subgroups (stability, 0.16; connectivity: 180.8; Dunn: 0.16; silhouette: 0.05), with 176 patients in the optimal achievement subgroup and 157 patients in the suboptimal achievement subgroup. The 2 subgroups differed significantly (all P≤ .004) in the likelihood of achievement of all CSOs. Stepwise multivariable logistic regression identified an increase of 1 mm in tear size in the sagittal dimension beyond 1.9 cm to predict a 10% increase in the probability of suboptimal achievement. Additional risk factors for suboptimal CSO achievement included increasing number of tendons involved (odds ratio [OR], 14.07; 95% CI, 4.50-44.02; P < .001), subscapularis involvement (OR, 8.67; 95% CI, 2.45-30.71; P = .01), and increased preoperative CMS score (OR, 1.11; 95% CI, 1.04-1.18; P = .001). Protective factors included performance of a subpectoral biceps tenodesis compared with biceps tenotomy (OR, 0.22; 95% CI, 0.05-0.92; P = .03).
Conclusion:
Clinically meaningful subgroups were uncovered using an unsupervised machine learning algorithm in patients undergoing arthroscopic RCR. Tear size, number of tendons involved, and subscapularis involvement were significant and additive predictors of suboptimal CSO achievement at 2-year minimum follow-up. Treatment of concurrent biceps pathology with tenodesis conferred 78% increased likelihood of CSO achievement compared with tenotomy.
Keywords
Rotator cuff tears are highly prevalent, affecting between 20% and 80% of individuals >50 years old; with incidence increasing with age.20,25,30,34 Arthroscopic rotator cuff repair (RCR) is the current standard of care treatment for patients with acute full-thickness tears as well as chronic tendinopathy who fail nonoperative treatment,1,24 and it remains one of the most performed elective procedures in the United States. 33 There has been significant research into the diagnosis and treatment of rotator cuff tears over the past 30 years; however, despite substantial progress in the understanding of rotator cuff disease, challenges remain in optimizing outcomes for all patients. 14 Partial- or full-thickness retears are still relatively common, affecting as many as 1 in 5 patients at 12 to 24 months’ follow-up. 17
Numerous studies have been performed to identify factors associated with retears after RCR. However, while an emerging body of evidence points to superior strength and clinical outcomes in patients with intact repairs, 35 historically, not all patients with retears demonstrate functional deficits in the short to midterm.7,17,21 Therefore, patient-reported outcomes (PROs) may represent an alternative method of evaluating a patient's recovery after arthroscopic RCR in the absence of long-term follow-up or observation of glenohumeral degenerative changes. Specifically, PRO responses over time may help identify achievement of clinically significant outcomes (CSOs) at key milestones during the postoperative period. 29 It is important to identify characteristics associated with a decreased likelihood of symptomatic recovery, as these characteristics may serve as potential therapeutic targets to improve patient outcomes.
Previous studies examining predictors of CSOs after arthroscopic RCR have demonstrated inconsistent results that differ from models predicting radiographic evidence of retear.9,15,29 This may be attributed to limitations in traditional case-control approaches, which require a stratifying variable (eg, smoking status), the determination of which can at times be arbitrary. Moreover, parametric regression models are only valid given specific assumptions about the distribution of data and less flexibly adapt to population changes over time. To mitigate these limitations, there is a growing trend of utilizing nonparametric, data-driven approaches to improve our understanding of orthopaedic disease. 22 Unsupervised clustering is an approach for exploratory analysis that aims to identify patient subgroups not based on any single variable, but on the natural differences identified after aggregating all known characteristics of each patient. 23
Therefore, the purpose of this study was to determine predictors of achieving CSO thresholds after elective arthroscopic RCR by utilizing unsupervised machine learning. We hypothesize that an unsupervised random forest algorithm can identify distinct clinically meaningful subgroups in patients undergoing arthroscopic RCR and that patients with larger sagittal (anteroposterior) tear dimensions and greater number of tendons involved would exhibit poorer CSO achievement.
Methods
Study Design, Patient Selection, and Data Source
The present study was a retrospective case-cohort study of prospectively collected PROs aggregated by a prospectively maintained institutional registry. Following approval by our university's institutional review board, an electronic data collection service (Outcome Based Electronic Research Database; Universal Research Solutions) was queried for PRO scores for all patients undergoing arthroscopic RCR at preoperative and 2-year postoperative follow-up. Inclusion criteria were completion of PRO data at preoperative and postoperative follow-up time points. Exclusion criteria included workers’ compensation patients, patients without consecutive PRO data, patients who had concomitant placement of a subacromial balloon device, and patients who underwent biological augmentation at the time of surgery. Patient data were compiled across 4 fellowship-trained sports medicine orthopaedic surgeons (A.B.Y., B.J.C., N.N.V., B.F.) between January 26, 2015 and May 1, 2017. Baseline demographics as well as comorbid variables were entered into the outcome data collection service. A team of trained research fellows and an orthopaedic surgery clinical fellow (M.H., E.J.B., H.H.P., M.R.) performed tear size measurements on preoperative magnetic resonance imaging based on a protocol as described by MacDermid et al. 19 Tear dimensions in both the coronal (mediolateral direction) and the sagittal (anteroposterior direction) planes were measured and documented. Tears were defined as chronic if presentation was not associated with a traumatic inciting injury and patients underwent a trial of nonoperative treatment for ≥3 months.
Surgical Techniques and Rehabilitation
Surgical technique was dependent on surgeon decision making based on preoperative and intraoperative assessment of tear size, configuration, location, and overall repairability. All patients underwent repair using 1 of 4 techniques—transosseous equivalent/suture bridge, double row, single row, or some combination thereof with additional side-to-side (posterosuperior rotator cuff or superior border of subscapularis) or luggage tag repairs as needed. All patients also received a subacromial decompression. Treatment of biceps pathology was based on a combination of preoperative patient-centered discussion and intraoperative findings. If biceps pathology was detected during diagnostic arthroscopy, concomitant open or arthroscopic biceps tenodesis, or biceps tenotomy, was performed. Factors influencing the choice of biceps treatment included patient preference and arthroscopic findings such as degree of tendinosis and retraction.
Postoperatively, all patients followed a standardized arthroscopic RCR rehabilitation protocol. The initial 4 to 6 weeks involved sling immobilization of the shoulder with pendular movements only. At 4 weeks, patients began passive range of motion (ROM) exercises with the help of a physical therapist. By 8 weeks, they progressed to active-assisted ROM, which included wall slides and isometric exercises. Finally, between 10 and 12 weeks after surgery, they were permitted to begin strengthening and active-ROM exercises as tolerated. Gentle active ROM and exercises for the hand, wrist, and elbow were encouraged throughout the postoperative period.
Patient-Reported Outcomes
Patients who were enrolled in the prospective registry completed shoulder-specific functional PROs, including the American Shoulder and Elbow Surgeons (ASES) form, the Single Assessment Numeric Evaluation (SANE), and the Constant-Murley score (CMS) shoulder component. We utilized the threshold values for CSO achievement previously established by Cvetanovich et al. 4 Briefly, to indicate achievement of the Patient Acceptable Symptom State (PASS), 12 patients responded “yes” or “no” to the following question: “Taking into account all activities you have done during your daily life, your level of pain, and your functional impairment, do you consider that your current state is satisfactory?” The minimal clinically important difference (MCID) and substantial clinical benefit (SCB) were determined by responses to the following anchor question: “Since your surgery, has there been any change in the overall function of your shoulder?” Patients could select 1 of the 15 possible responses to this question ranging from “no change” to “a great deal better/worse,” as shown in Supplemental Material Figure A1 (available separately), to quantify the degree of their perceived postoperative change. Respondents were categorized as experiencing no improvement, minimal change (“somewhat worse” to “somewhat better”), or substantial improvement (“a good deal better” to “a very great deal better”).
Unsupervised Clustering
Missing variables were imputed based on previously described standards and techniques. 18 After imputation of missing variables, patients were partitioned into clinically distinct subgroups by clustering with an unsupervised random forest algorithm. Clustering is a machine learning technique that produces optimized nonoverlapping regions in the data space based on a specified distance measure. User input into the model in this instance is minimal, without supplied features and a specified outcome (hence, “unsupervised”). Distinct from supervised machine learning (SML), this technique is often utilized for exploratory analysis, dimensionality reduction, and outlier removal, especially when there are manifold outcomes of interest in the data, such as in the present scenario. An ideal clustering assignment yields distinct subgroups, each associated with a different likelihood of CSO achievement.
As previously described, we utilized a random forest algorithm to first perform the distance calculations between every patient in the cohort, organized in a proximity matrix.23,27 The optimal number of clusters and cluster assignments were then determined using an agglomerative hierarchical clustering method with the Ward minimum variance criterion. 10 Algorithm performances were then evaluated through internal validation metrics, which assess the quality of clustering based on the partitions produced and the patients within each. We used the internal validation metrics of connectivity and silhouette coefficient, along with the internal stability metrics of average distance (AD) and figure of merit (FOM) to determine the best candidate clustering algorithm.
Briefly, connectivity is a measure of the degree to which nearest neighbors in the feature space are clustered together; it can take a value between zero and infinity and should be minimized. 16 The silhouette coefficient is a proportion of the distance between objects in a cluster to the distance between neighboring clusters and should be maximized. 16 AD measures the changes in AD between observations within the same cluster and FOM measures the variance within each cluster, respectively, after iterative elimination of features. 2
Visual assessment of the subgroups generated by unsupervised clustering was also performed. To reduce the >60 features associated with each patient into a 3-dimensional coordinate system that could be plotted, we performed principal component analysis (PCA). 26 Patients were then plotted in the resulting coordinate system consisting of the 3 principal components with their subgroup labels.
Statistical Analysis
Univariate comparisons between optimal and suboptimal subgroups were performed using Welch t tests for continuous variables and chi-square analyses for categorical variables. Finally, subgroup membership and >30 demographic and comorbidity variables were entered into a multivariable regression. The regression was fitted using a stepwise model maximizing the Aikake information criterion, which aims to identify a group of variables that together best predicts subgroup membership, although each individual predictor may not be significantly associated with subgroup membership. Compared with variable importance analysis internal to the random forest algorithm, multivariate logistic regression could produce estimands in the form of odds ratios (ORs), which quantify the contribution of specific risk factors to suboptimal subgroup membership. All statistical tests were 2-tailed, and the statistical difference was established with an alpha <.05. All modeling and statistical analysis was performed through the R Language for Programming Version 4.2.2 in RStudio software Version 1.1.143 (R Foundation for Statistical Computing).
Results
Study Cohort Characteristics and Subgroup Membership
A total of 346 patients (192 male; mean ± SD age, 57.2 ± 9.1 years; body mass index [BMI], 30.1 ± 5.4 kg/m2) were eligible for inclusion and followed for a mean of 3.8 years (range, 2.0-6.2 years). The random forest algorithm generated 2 main subgroups totaling 333, which were named “optimal” and “suboptimal” based on perceived CSO achievement (Table 1). The algorithm arrived at the ideal subgroup number of 2 according to the summary of internal validation metrics, which demonstrated negligible improvements in these metrics with partitioning of the study cohort to >2 subgroups.
Internal Validation of Ideal Cluster Number
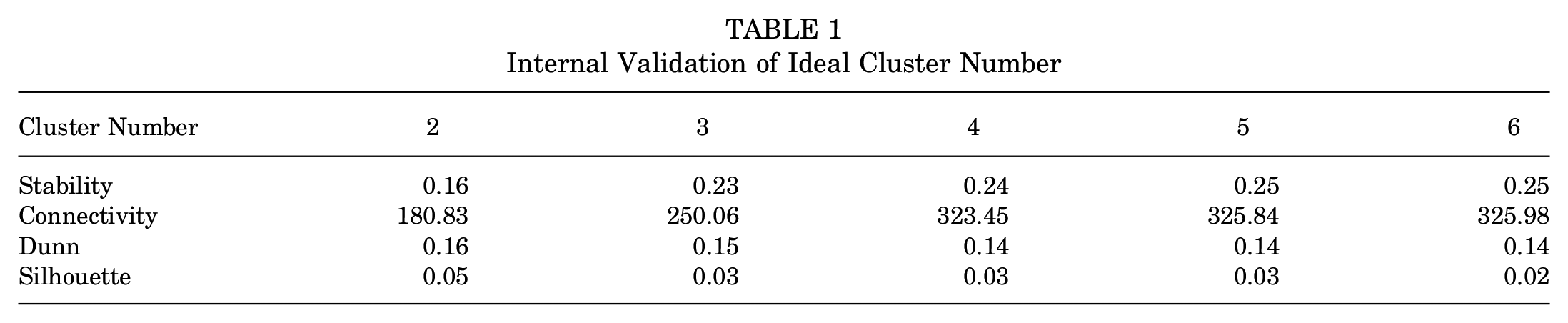
A total of 176 (50.9%) patients comprised the optimal subgroup, and 157 (45.4%) comprised the suboptimal subgroup, while 13 (3.8%) patients were excluded as outliers not belonging to either subgroup. The existence of 2 main subgroups with distinct patient features was clearly visualized when subgroup memberships were plotted following PCA (Figure 1A). The variables found by the random forest algorithm to drive subgroup membership included the tear's anteroposterior (sagittal) dimension, BMI, tear thickness, and preoperative CMS score (Figure 1B).

(A) Visualization of the 2 random forest partitioned patient subgroups in 3-dimensional feature space; the axis is generated following principal component (PC) analysis to reduce the dimensionality of the data set for ease of visualization. (B) Global variable importance of feature contributions to the performance of the random forest clustering algorithm. BMI, body mass index; CMS, Constant-Murley score; MCID, minimal clinically important difference; PASS, Patient Acceptable Symptom State; SANE, Single Assessment Numeric Evaluation; SCB, substantial clinical benefit.
Subgroup Demographics
Subgroup demographics are shown in Table 2. In the optimal subgroup, patients were 59% male with a mean age of 56 ± 10 years. In the suboptimal subgroup, patients were 52% male (P = .25) and had a mean age of 59 ± 8 years (P = .002). The prevalence of common comorbidities was balanced between groups, but patients in the suboptimal achievement subgroup were more likely to have hypertension (39.5% vs 20.5%; P < .001). Regarding baseline medication use, there were no differences in the use of hypertension medications, psychotropic medications, opioids, or nonsteroidal anti-inflammatory drugs (P > .05 for all).
Comparison of Patient Demographics Stratified by Cluster Membership a

Values are presented as mean ± SD or n (%) unless otherwise indicated. Statistically significant P values are bolded. ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; BMI, body mass index; NSAID, nonsteroidal anti-inflammatory drug.
Subgroup Injury and Operative Characteristics
Clinical characteristics by subgroup are demonstrated in Table 3. Patients with optimal outcomes were more likely to have isolated supraspinatus tears (87% vs 27%; P < .001), and the optimal subgroup had a mean of 1 tendon involved versus 2 tendons in the suboptimal group (P < .001). Patients in the suboptimal group were more likely to have subscapularis involvement (24% vs 9%; P < .001). Correspondingly, the suboptimal group had larger mean tear size: patients in this group had a mean sagittal (anteroposterior) dimension of 40 ± 16 mm and coronal (mediolateral) dimension of 39 ± 15 mm (both P < .001), while the optimal group had a mean sagittal dimension of 19 ± 9 mm and coronal dimension of 25 ± 10 mm (both P < .001). There were no differences in nonoperative treatments attempted between groups before surgery (see Supplemental Material).
Comparison of Injury and Operative Characteristics Stratified by Cluster Membership a

Values are presented as mean ± SD or n (%) unless otherwise indicated. Statistically significant P values are bolded. PEEK, polyether ether ketone.
Regarding surgical characteristics, the suboptimal group used a mean of 1 anchor more than the optimal group (3.6 ± 1.3 vs 2.7 ± 1.1; P < .001). The suboptimal group had a more heterogeneous mix of anchor materials used, while 81% of the optimal group used polyether ether ketone (PEEK) anchors (P = .03). In the suboptimal group, 70% of patients had anchors placed via a suture-bridge technique and 22% with a double-row repair, while in the optimal group 42% of patients had anchors placed via a suture-bridge technique and 46% had a double-row repair (P < .001). In the suboptimal group, more concurrent biceps procedures were performed, with 40% of patients receiving tenodesis and 10% receiving tenotomy; while 39% and 2% of the patients in the optimal subgroup underwent tenodesis and tenotomy, respectively (P = .01).
Subgroup Outcomes
Overall, the optimal group demonstrated greater proportions of CSO achievement compared with the suboptimal group (Table 4). A significantly higher percentage of patients in the optimal subgroup were able to achieve the MCID, SCB, and PASS on the ASES, SANE, and CMS at 2-year follow-up (P = .001-.004).
Comparison of CSOs Stratified by Cluster Membership a

Values are presented as n (%) unless otherwise indicated. Statistically significant P values are bolded. ASES, American Shoulder and Elbow Surgeons; CMS, Constant-Murley score; CSO, clinically significant outcome; MCID, minimal clinically important difference; PASS, Patient Acceptable Symptom State; SANE, Single Assessment Numeric Evaluation; SCB, substantial clinical benefit.
Predictors of Suboptimal Subgroup Membership
A stepwise multivariate logistic regression model was generated to identify preoperative and intraoperative predictors of membership in the suboptimal subgroup and quantify their influences, while adjusting for confounders (Table 5). Factors most associated with suboptimal subgroup membership were a greater number of involved rotator cuff tendons (OR, 14.07; 95% CI, 4.50-44.02; P < .001), concurrent biceps tenotomy (OR, 9.52; 95% CI, 2.16-41.96; P = .002), subscapularis involvement (OR, 8.67; 95% CI, 2.45-30.71; P = .01), preoperative CMS (OR, 1.11; 95% CI, 1.04-1.18; P = .001), and a 1-mm increase in tear size in the anteroposterior dimension compared to the optimal group (OR, 1.10; 95% CI, 1.05-1.15; P < .001). In other words, each millimeter increase in tear size beyond 19mm (median value of the optimal subgroup) incurs a 10% risk of suboptimal achievement as the overall size approaches 39.8mm (median value of the suboptimal subgroup). Factors associated with a lower likelihood of suboptimal subgroup membership included concurrent suprapectoral biceps tenodesis (OR, 0.22; 95% CI, 0.05-0.92; P = .03), suggesting a 78% decreased likelihood compared to patients who underwent tenotomy. An interaction term between subscapularis involvement and the number of tendons involved was highly significant, suggesting that if multiple tendons were involved, a subscapularis tear had a smaller effect on suboptimal subgroup membership. By contrast, if fewer tendons were involved, a subscapularis tear had a greater association with suboptimal subgroup membership.
Predictors of Suboptimal Cluster Membership on Multivariable Stepwise Regression Model a

CMS, Constant-Murley score; SANE, Single Assessment Numeric Evaluation. Statistically significant P values are bolded.
Interaction term between number of tendons involved and subscapularis involvement. This interaction term demonstrated that the effects of subscapularis involvement decreased with the number of total tendons involved and vice versa.
Discussion
The principal findings of this study are as follows: first, an unsupervised random forest algorithm effectively generated 2 statistically different subgroups in patients undergoing arthroscopic RCR. Second, a comparative examination of the resulting subgroups demonstrated significant differences in achievement of CSO. Finally, a multivariable stepwise regression identified number of tendons involved, the performance of biceps tenotomy versus tenodesis, subscapularis involvement, preoperative CMS score, and anteroposterior (sagittal) tear dimension, to drive subgroup membership. These findings represent clinically relevant points that can help drive preoperative counseling and manage expectations in patients at high likelihood of suboptimal outcomes.
The unsupervised random forest algorithm successfully generated 2 clinically meaningful subgroups. This is evident from both internal validation of the group differences as well as visualization of the subgroups after dimensionality reduction via PCA. We observed significantly higher rates of CSO achievement across all PRO measures in the optimal subgroup compared with the suboptimal subgroup. Based on variable importance analysis, tear size in the sagittal dimension as well as BMI were the 2 highest-weighted variables for partitioning subgroup membership. In other words, inclusion of those 2 variables (sagittal tear dimension and BMI) helped the algorithm most effectively assign final subgroup membership to each patient. Interestingly, while side-by-side comparisons between the subgroups demonstrated significant differences in tear dimension in both the sagittal and the coronal dimensions, there were no statistically significant differences in BMI between the 2 groups, and nor was BMI a significant predictor of suboptimal achievement on regression analysis. This is consistent with several studies in the literature that revealed no differences in clinical and functional outcomes after arthroscopic RCR in obese patients.8,32
Several studies have characterized the patient risk factors that prevent the achievement of CSO after RCR.4,31 Cvetanovich et al 4 demonstrated that preoperative functionality scores, active tobacco use, single-row repairs, obesity, and workers’ compensation status all served as negative predictive factors for the achievement of CSOs. Tramer and colleagues 31 subsequently showed that patients with larger tear sizes, a history of shoulder surgery, and concomitant distal clavicular excision were at an increased risk of failing to achieve CSO. They found active smoking to be an additional risk factor as well. We identified that the number of torn rotator cuff tendons, subscapularis tendon tears, tear sizes, and preoperative PRO data scores (CMS) were additive predictors for suboptimal CSO achievement.
While previous studies have shown that larger-sized tears are a risk factor for not achieving CSO, we additionally determined that this risk factor is most significant in the sagittal plane, which suggests that the anteroposterior configuration of the tear is an important consideration in predicting long-term PROs. This is further supported by the works of Kwon et al, 13 who identified anteroposterior tear dimension as a significant contributor in their establishment of the Rotator Cuff Healing Index, with 2.5 cm as the critical threshold beyond which tendon healing was significantly compromised. As previously noted, the larger size of an anteroposterior tear may point to multitendon involvement, including both the superoposterior cuff and possibly the superior subscapularis, which have significant functional consequences with loss of elevation and external rotation. Additionally, superoposterior fatty degeneration has long been highlighted as a poor predictor of repairability in the long term, especially with chronic presentation or delays in treatment. 3
An increased number of involved rotator cuff tendons was itself recognized as an important risk factor for suboptimal achievement. Indeed, an increased number of rotator cuff tendons involved likely predisposes to a more difficult and complex repair as well as greater subsequent disability. An especially salient observation is the fact that while subscapularis involvement alone was a predictor of suboptimal outcomes, as the number of tendons involved increased, the negative effect of subscapularis involvement was diminished. This suggests that the significant disability associated with massive superoposterior rotator cuff tears may to a degree alter the patient's perception of the functional consequences of subscapularis injury. This hypothesis may warrant further evaluation with future investigations.
Preoperative scores are a risk factor for not reaching CSO, with previous studies demonstrating that lower preoperative functional scores predict greater potential for achieving MCID and SCB, and higher preoperative functionality scores predict higher chances of attaining PASS. 4 Interestingly, the present study found that the treatment of a concomitant biceps tendon pathology with tenodesis appeared to increase the likelihood of achieving CSO when compared with tenotomy. However, this finding is likely confounded by substantial selection bias given surgeon preference for tenotomy in lower demand patients compared with tenodesis in healthy, active patients.
Using an unsupervised clustering algorithm is a novel approach in predicting patient outcomes after shoulder surgery. Previous methods involved utilizing either a case-control approach using a predetermined stratifier 28 (eg, smoker vs nonsmoker) or the regression approach, in which a regression model was constructed using either traditional parametric methods or machine learning methods to predict a binary outcome of interest (ie, achievement of the MCID on the ASES score).6,11,18 While both methods can effectively identify risk factors for poor outcomes, the former approach is dependent on accurate but arbitrary selection of the stratifying variable through clinical observation, whereas the latter requires a predefined binomial or multinomial outcome and can become cumbersome to deploy. In the example of our analysis, 9 models would have to be developed (3 CSOs on 3 PROs). Furthermore, both approaches require a degree of human input, either to define a stratifying variable or an outcome. Clustering using an unsupervised machine learning algorithm represents a truly data-driven method that provides a global assessment of patient outcomes subject to minimal bias from human interference. However, it is worth noting that this approach is not meant to supplant existing analytics, but rather to be used in conjunction for both hypothesis generation and exploratory analysis. For example, differences between generated subgroups can be used to drive future level 1 studies, or supervised models can be used to predict subgroup membership. A final upside of the unsupervised approach is that it is less reliant on the robust sample sizes required of an SML prediction model because a distance matrix can be theoretically calculated for any number of cases in a cohort. 5
Limitations
This study is not without its limitations. The results of our study and conclusions are limited to single institution data, and caution should be taken when attempting to generalize these findings to other populations; furthermore, addition of clinical outcomes to PRO data could potentially reinforce or refute the established subgroup identities. Given the retrospective nature of the data collected, it is difficult to assess whether the risk factors identified are causal or correlational. The unsupervised algorithm prioritizes clustering patients based on variables that most distinctly differentiate between the outcomes of interest (eg, achievement of CSOs), but does not necessarily demonstrate causation of suboptimal achievement. Many of the differences in the comparison of intraoperative variables are clearly the result of this confounding, such as anchor configuration and the number of repairs under marginal convergence. It is clear that patients did not have suboptimal outcomes because of repair performance under marginal convergence but rather because their initial injury configuration (eg, “L”- or “U”-shaped tears with significant extension of the tear medially) warranted such a repair; indeed, while arthroscopic assessment of tear configuration was not specifically documented in our series, it certainly requires attention in future investigations. Other relevant variables such as Goutallier grade or concomitant distal clavicular excision were similarly not examined in our series and deserve further study. Additionally, the complexity of distance calculations used by the unsupervised algorithm increases exponentially with the number of features included in the model; however, the random forest algorithm has an internal feature importance function to guide interpretability of the initial subgroups. Fortunately, the risk factors identified from the stepwise logistic regression are less subjective to this causality dilemma, as tear size and number of tendons involved are clearly associated with the initiating injury. Finally, the retrospective nature of this study likely introduced a degree of selection bias that could potentially drive the differences between tenodesis and tenotomy, because the latter is generally reserved for lower demand patients whose baseline activity and function may portend suboptimal outcomes.
Conclusion
Clinically meaningful subgroups were uncovered using a machine learning clustering approach in patients undergoing arthroscopic RCR. Tear size, number of tendons involved, and subscapularis involvement were highly significant and additive predictors of suboptimal CSO achievement at 2-year minimum follow-up. Treatment of concurrent biceps pathology with tenodesis conferred 74% increased likelihood of CSO achievement versus tenotomy.
Supplemental Material
sj-docx-1-ojs-10.1177_23259671251335977 – Supplemental material for Defining Clinically Meaningful Subgroups in Patients Undergoing Arthroscopic Rotator Cuff Repair Using Unsupervised Machine Learning
Supplemental material, sj-docx-1-ojs-10.1177_23259671251335977 for Defining Clinically Meaningful Subgroups in Patients Undergoing Arthroscopic Rotator Cuff Repair Using Unsupervised Machine Learning by Yining Lu, Elyse J. Berlinberg, Kareme Alder, Ethan Chervonski, Harsh H. Patel, Morgan Rice, Adam B. Yanke, Brian J. Cole, Nikhil N. Verma, Mario Hevesi and Brian Forsythe in Orthopaedic Journal of Sports Medicine
Footnotes
Final revision submitted December 4, 2024; accepted January 2, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: E.J.B. has stock or stock options in Pfizer, Inc, and Amgen, Inc. H.H.P. has received hospitality payments from Abbott Laboratories and Medtronic. A.B.Y. has received hospitality payments and consulting fees from Stryker and nonconsulting fees from Arthrex; is a paid consultant for AlloSource, JRF Ortho, Stryker, and Arthrex; has received research support from Arthrex, and Organogenesis; has stock or stock options in Icarus Medical, Patient IQ, and Sparta Biomedical; is an unpaid consultant for Patient IQ and Sparta Biomedical; and is a paid presenter or speaker for Stryker. B.J.C. has received consulting fees from Aesculap Biologics, Arthrex, Bioventus, Enovis, Vericel, OSSIO LTD, Pacira Pharmaceuticals Inc, Endo Pharmaceuticals, and ACUMED LLC; nonconsulting fees from Terumo BCT; IP royalties from Arthrex, and Elsevier Publishing; hospitality payments from GE Healthcare; honoraria from Vericel; research support from Aesculap/B.Braun, Arthrex, and National Institutes of Health (NIAMS and NICHD); and other financial or material support from JRF Ortho. He holds stock or stock options in Bandgrip Inc and Ossio. N.N.V. has received hospitality payments from Abbott Laboratories, Spinal Simplicity, and Relievant Medsystems; royalties from Arthrex, Smith & Nephew, Stryker, and Graymont Professional Products; consulting fees from Arthrex and Stryker; and research support from Breg, Ossur, Smith & Nephew, Stryker, and Arthrex. M.H. has received education payments from Arthrex, Foundation Medical, Medwest Associates, and Smith & Nephew; honoraria from Encore Medical; hospitality payments from Orthalign and Stryker; consulting fees from Enovis, Moximed, and Vericel; a grant from Medical Device Business Services; and publishing royalties and financial or material support from Elsevier. B.F. has received debt forgiveness from Medwest Associates; consulting fees from Stryker and Smith & Nephew; research support from Arthrex, Smith & Nephew, and Stryker; and publishing royalties and financial or material support from Elsevier. He holds stock or stock options in iBrainTech, Sparta Biopharma, and Zuno Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by Rush University Medical Center (No. 20041404-IRB01).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
