Abstract
Background:
Although previous studies have investigated the risk factors for rotator cuff syndrome (RCS), there remains controversy due to uncontrolled and uncertain confounding factors in their analyses.
Purpose:
To perform Mendelian randomization (MR) analysis using single-nucleotide polymorphisms to investigate the causal relationship between RCS and 4 risk factors: type 2 diabetes mellitus (T2DM), high blood pressure (HBP), body mass index (BMI), and low high-density lipoprotein cholesterol (HDL-C).
Study Design:
Descriptive epidemiology study.
Methods:
Genome-wide association study (GWAS) data for T2DM (ebi-a-GCST006867), BMI (ieu-b-40), HBP (finn-b-I9_HYPTENS), HDL-C (ieu-b-109), and RCS (ukb-b-50) were obtained from the IEU Open GWAS Project. The dataset of each risk factor was combined with the dataset of RCS, generating 4 datasets. Potential confounders and single-nucleotide polymorphisms related to RCS were excluded from these datasets. The causal relationships between the exposure factors and RCS were analyzed using 5 regression models: MR-Egger, weighted median estimate (WME), inverse-variance weighting (IVW), simple mode, and weighted mode. Heterogeneity in the causal effects was assessed using MR-Egger regression and IVW analyses. Sensitivity analyses were performed to determine the stability of the results.
Results:
The MR-Egger regression intercepts for T2DM, BMI, HBP, and HDL-C showed no horizontal pleiotropic effects. The results of the Cochran Q test showed P values of .075 and .080 for BMI in the MR-Egger regression and IVW models, respectively, indicating the absence of heterogeneity between BMI and RCS. The results of the weighted median estimate and IVW regression analyses showed a significant causal association between BMI and RCS, with odds ratios of 1.002 (95% CI, 1-1.004; P = .038) and 1.003 (95% CI, 1.001-1.005; P = .0003), respectively. No significant associations were found for T2DM, HDL-C, or HBP.
Conclusion:
In the present study, BMI was positively associated with the risk of developing RCS, while T2DM, HBP, and low HDL-C were not associated with RCS development.
Keywords
Rotator cuff syndrome (RCS) is a common cause of shoulder pain, significantly affecting millions of patients worldwide. 29 There are approximately 40,000 medical visits for shoulder pain in the United States, with health care costs estimated 19 at US$500 million in 2011. The prevalence of RCS is between 5% and 44% in patients <10 years and >60% in patients >20 years. The prevalence of chronic shoulder pain in adults can reach 80%.
RCS is a spectrum of diseases associated with injuries or degenerative conditions affecting the rotator cuff. These pathologies include subacromial impingement syndrome, subacromial bursitis, rotator cuff tendinitis, and partial- or full-thickness rotator cuff tears. 35 Subacromial impingement syndrome is the most common cause of shoulder pain.39,19 Intrinsic factors can also lead to tendon deformities and accelerate tearing. Potential risk factors for RCS include vascular changes, age, sex, genetics, and muscle tension. Environmental factors, including overhead throwing motion, are also associated with an increased risk of developing RCS. 12 Smoking correlates with rotator cuff degeneration, symptomatic rotator cuff tears, and the increased size of these tears.7,17 Other comorbidities may also affect the development and progression of RCS. Metabolic syndrome is involved in the pathophysiology of RCS, and observational studies have shown that low high-density lipoprotein cholesterol (HDL-C), high blood pressure (HBP), hyperglycemia, and high body mass index (BMI) are associated with an increased risk of developing rotator cuff injuries.37,16,23,13 Further, thyroid disorders, type 1 diabetes, and inflammatory arthritis are potential risk factors for RCS.24,1,25,10,38
In epidemiological studies, the estimation of causal effects of exposure on outcome is limited by confounding factors and reverse causality. The Mendelian randomization (MR) method has been widely adopted in causal inference studies using genome-wide association study (GWAS) data to overcome these limitations. The MR method randomizes populations into new groups based on the random assortment and recombination of alleles during gametogenesis. In theory, this method reduces the influence of confounders, thus making causal inferences more accurate than other methods. The 2-sample MR method uses genetic variations as instrumental variables to measure the causal effects of exposure on outcomes independently. The implementation of MR analysis relies on 3 key assumptions: (1) the instrumental variable is significantly associated with the exposure; (2) the instrumental variable is independent of any confounding factors between the exposure and the outcome; and (3) the instrumental variable cannot directly affect the outcome and can only indirectly influence the outcome through the exposure.
In the present study, we performed a 2-sample MR analysis using instrumental variables (single-nucleotide polymorphisms [SNPs]) and GWAS summary data to investigate the causal relationship between RCS and 4 exposure factors: HBP, type 2 diabetes mellitus (T2DM), HDL-C, and BMI (databases of thyroid disorders and type 1 diabetes are small and thus unsuitable for 2-sample MR analysis). A directed-acyclic graph was constructed using the instrumental variable, the exposure factors, and the outcome to elucidate the aforementioned assumptions for MR analysis and outline the study design (Figure 1).11,33,18,3

A directed acyclic graph using the instrumental variable (SNPs), the exposure factors (BMI, T2DM, HBP, and HDL-C), and the outcome (RCS). BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; RCS, rotator cuff syndrome; SNPs, single-nucleotide polymorphisms; T2DM, type 2 diabetes mellitus.
Methods
Datasets
GWAS data for T2DM, BMI, HBP, HDL-C, and RCS were obtained from the IEU Open GWAS Project website on August 1, 2023 (source URLs in Appendix Table A1). The populations in the exposure and outcome datasets were European, with no restriction on sex. Ethics committee approval was not needed for this study.
Assumptions for Using SNPs as Instrumental Variables
Three assumptions need to be satisfied when using SNPs as instrumental variables. First, the instrumental variable highly correlates with exposure 9 (F statistic >10). Second, there is no genetic pleiotropy (ie, the effects of the instrumental variable on the outcome are mediated solely by the exposure). In this study, the intercept term of the MR-Egger regression model was tested for zero (P > .05) to confirm the absence of genetic pleiotropy.28,6 Third, instrumental variables are independent of unmeasured confounding factors. It can be assumed that SNPs are independent of socioeconomic and cultural factors because SNPs in MR methods follow the random assortment of alleles from parents to offspring. 5
Criteria for Selecting SNPs From GWAS Summary Data for Exposure Factors
SNPs with a P value of >5 × 10−8 were chosen. A linkage disequilibrium coefficient (r2) of 0.001 and a linkage disequilibrium region width of 10,000 kb were determined to ensure that the selected SNPs were independent, eliminating the impact of genetic pleiotropy on the results.8,21 Data on SNPs were retrieved from the GWAS on RCS, with r2 values of >0.8. SNPs with high linkage disequilibrium were used as proxies for missing SNPs, and SNPs without suitable proxies were removed.20,5 The selection criteria for proxy SNPs primarily include those with r2 threshold = 0.8 and minor allele frequency threshold = 0.3. Additionally, SNPs significantly (P < .00001) associated with potential confounders of RCS—including age >50 years and arm impedance—were identified and manually removed using PhenoScanner (http://www.phenoscanner.medschl.cam.ac.uk).
Estimation of Causal Effects
Causal relationships between the 4 exposure factors (T2DM, BMI, HBP, HDL-C) and RCS were assessed using 5 regression models: MR-Egger, weighted median estimate (WME), inverse-variance weighting (IVW), simple mode, and weighted mode. The IVW method does not require individual-level data and can directly estimate causal effects using summary data. The MR-Egger regression model assesses the correlation of each SNP with RCS and its correlation with the exposure factors, then fits a linear function. The WME method estimates the causal effect of each SNP on the exposure-outcome relationship. Sensitivity analyses—including heterogeneity and pleiotropy tests—were conducted to assess the stability of the results. Heterogeneity was detected using the Cochran Q test. 4 The IVW random-effects model was applied in the absence of heterogeneity, and the IVW random-effects model combined with the WME method was applied in the presence of heterogeneity. Sensitivity analysis was performed using the leave-one-out method, in which the causal effect was re-estimated after sequentially excluding a single variant at a time to evaluate the effect of each variant on the analysis. All methods were performed using the TwoSampleMR package in R (Version 4.3.1; The R Project for Statistical Computing), with the significance level set at P < .05.
Results
Selection of SNPs
The GWAS data for the 2-sample MR analysis are shown in Table 1. After screening, 118, 507, 62, and 362 SNPs were identified in the T2DM, BMI, HBP, and HDL-C datasets, respectively. Correlation analysis with the outcome, confounder identification and removal using PhenoScanner, and allele alignment were performed, allowing the selection of 44, 204, 26, and 128 SNPs, respectively (Table 2).
Genome-wide Association Studies Included in the 2-Sample Mendelian Randomization Analysis a

The populations in these datasets were European, and both sexes were included. BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; RCS, rotator cuff syndrome; T2DM, type 2 diabetes mellitus.
SNPs Associated with the 4 Exposure Factors a

BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; SNPs, single-nucleotide polymorphisms; T2DM, type 2 diabetes mellitus.
Heterogeneity, Pleiotropy, and Funnel Plots
Funnel plots showed that the selected SNPs of BMI were distributed symmetrically, indicating no significant biases (Figure 2). The P values of the Cochran Q test for BMI using the MR-Egger and IVW regression analyses were .075 and .080, respectively, indicating no heterogeneity. The MR-Egger regression intercept terms for T2DM, BMI, HBP, and HDL-C were 9.762 × 10−5 (P = .925), –2.627 × 10−5 (P = .630), –6.635 × 10−5 (P = .682), and 5.233 × 10−6 (P = .789), respectively, indicating that none of the selected SNPs had horizontal pleiotropic effects on the outcome and suggesting the stability of the findings (Table 3). Conversely, the P values for T2DM, HDL-C, and HBP were not significant, indicating the absence of pleiotropy or heterogeneity and the absence of a significant correlation between these factors and RCS.

Funnel plots. Each point represents a single-nucleotide polymorphism (SNP). The overall distribution was symmetrical, with a pyramid shape, indicating stability in the results and suggesting that SNP selection was not significantly biased. This was more evident for BMI and HDL-C. SEIV refers to the standard error in estimating instrumental variables; βIV refers to the estimated effect of instrumental variables. BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; IVW, inverse-variance weighting; MR, Mendelian randomization; T2DM, type 2 diabetes mellitus.
Heterogeneity and Sensitivity Test Results a
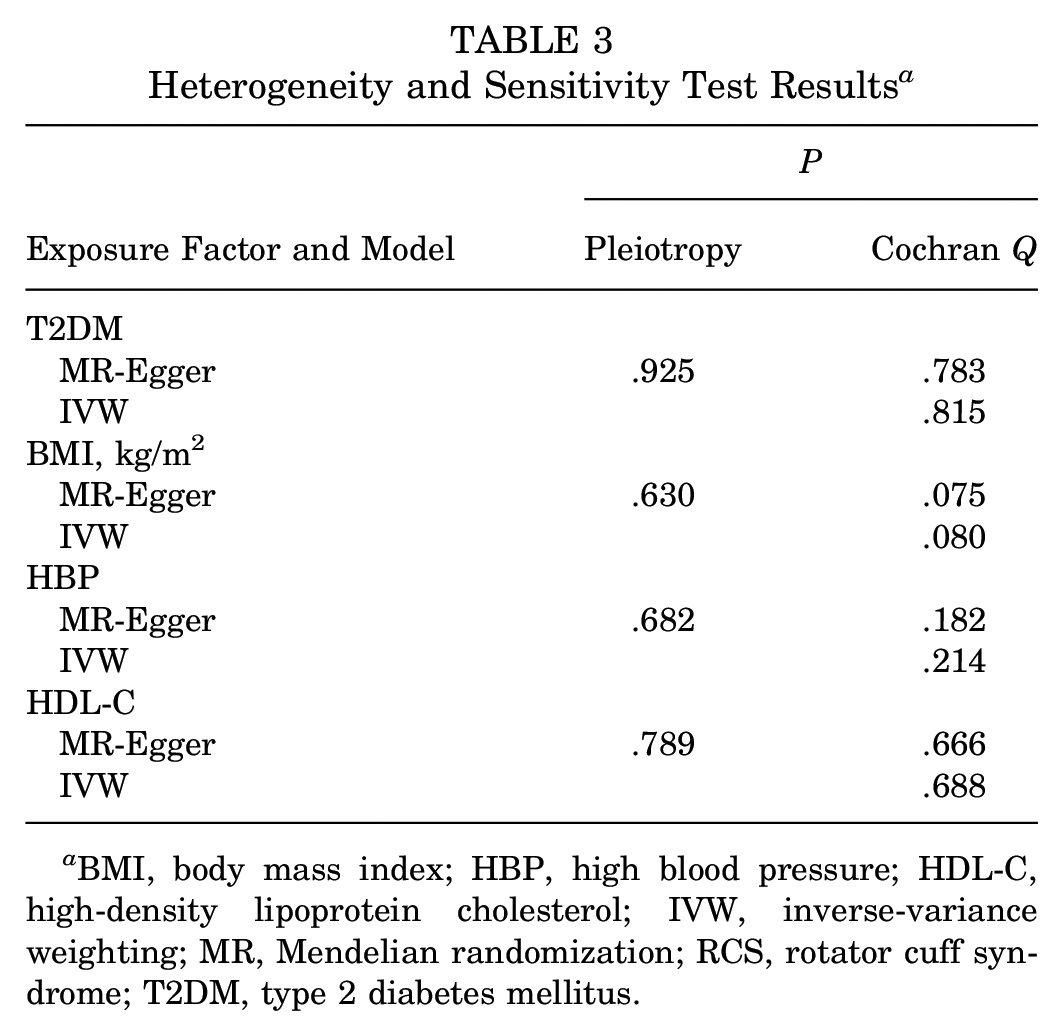
BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; IVW, inverse-variance weighting; MR, Mendelian randomization; RCS, rotator cuff syndrome; T2DM, type 2 diabetes mellitus.
Leave-One-Out Analysis
The results showed that the effect estimates for the remaining SNPs were located to the right of the null distribution, indicating no causal relationship, and were similar to the overall effect estimate (Figure 3). This result suggests that removing individual SNPs did not strongly affect the results. Hence, the MR findings were robust and not heavily influenced by any SNP.

Forest plots for leave-one-out analysis. All single-nucleotide polymorphisms >0 indicate the absence of bias in the experimental results, except for HBP, which approached zero. BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; T2DM, type 2 diabetes mellitus.
Causal Relationships
The regression analyses used to assess the causal relationship between BMI and the risk of RCS are presented in Table 4 and Figures 4 and 5. The odds ratios (ORs) for BMI in the MR-Egger, WME, IVW, simple-mode, and weighted-mode methods were 1.005 (95% CI, 0.999-1.013; P = .245), 1.002 (95% CI, 1-1.004; P = .038), 1.003 (95% CI, 1.001-1.005; P = .0003), 1.005 (95% CI, 0.998-1.013; P = .169), and 1.001 (95% CI, 0.995-1.007; P = .744), respectively. The ORs using the other methods were ≤1. The results of the IVW analysis for BMI (P = .0003 and β = 0.003) demonstrated that BMI was significantly positively associated with RCS. We focused on the results of the IVW method because no heterogeneity or pleiotropy was detected. This model showed that for every 1-standard deviation increase in BMI, the estimated risk of developing RCS increased by 1.003-fold (Figure 4).
Results of the 5 Regression Models a

Bold P values indicate statistical significance (P < .05). BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; IVW, inverse-variance weighting; MR, Mendelian randomization; OR, odds ratio; RCS, rotator cuff syndrome; T2DM, type 2 diabetes mellitus; WME, weighted-median estimate.

Scatter plots. The x-axis represents the effect of SNPs on the exposure factor, and the y-axis represents the effect of SNPs on the outcome (rotator cuff syndrome). A positive slope indicates that the exposure factor negatively affects the outcome. BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; IVW, inverse-variance weighting; MR, Mendelian randomization; SNPs, single-nucleotide polymorphisms; T2DM, type 2 diabetes mellitus.
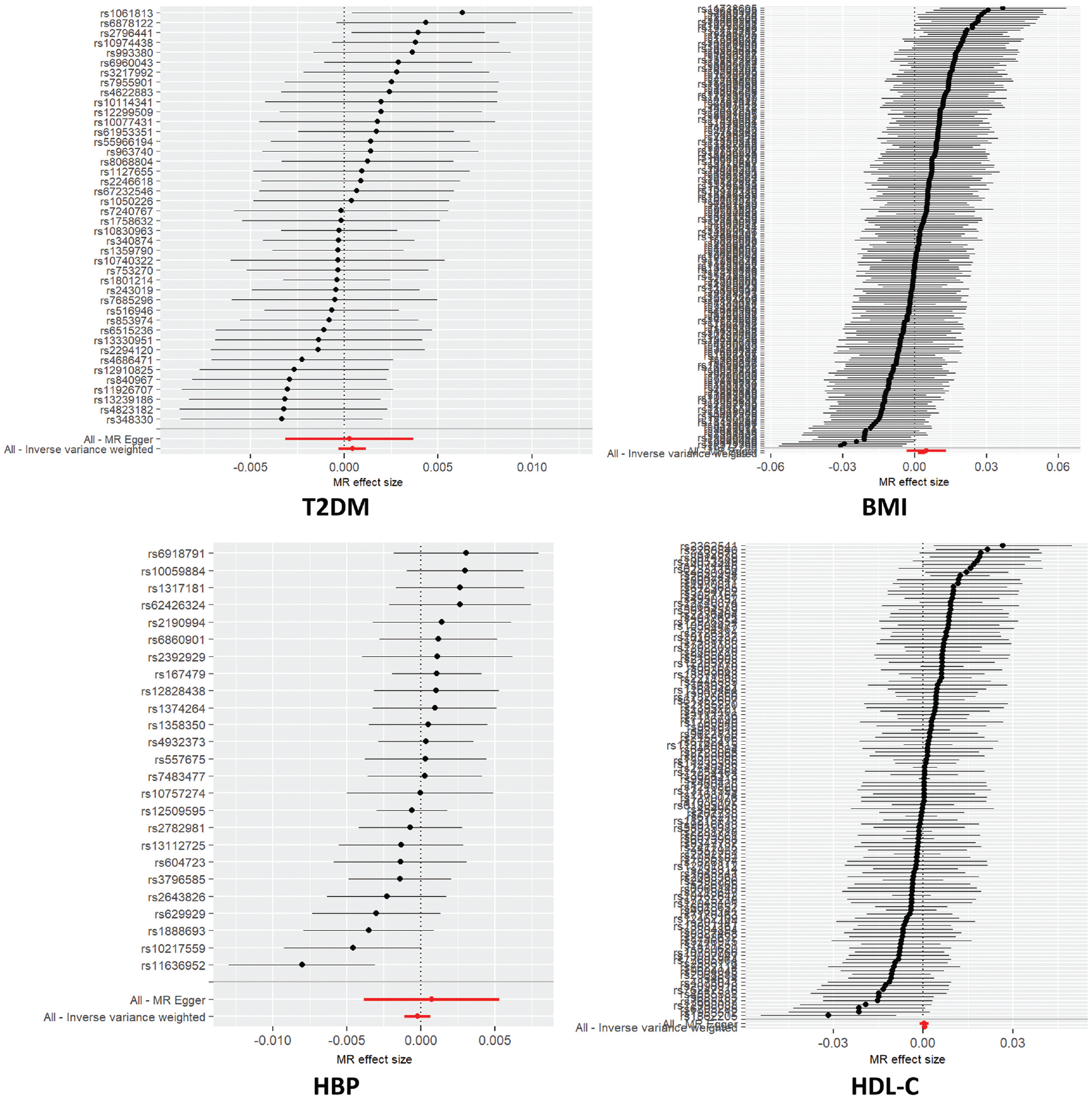
Forest plots. The horizontal axis of the forest plot represents the effect estimate, which indicates the extent to which the exposure factor affects the outcome factor. The vertical axis lists SNPs (single nucleotide polymorphisms). The long red lines represent the 95% confidence interval, in which all four exposure factors include 0, excluding the possibility of no association, and the short horizontal lines represent the 90% confidence interval, in which only BMI does not include 0 and the midpoint is greater than 0, indicating a strong positive correlation tendency. BMI, body mass index; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; T2DM, type 2 diabetes mellitus.
Discussion
The results of this study supported a causal relationship between BMI and RCS development, with ORs of 1.002 (95% CI, 1-1.004; P = .038) using WME regression analysis and 1.003 (95% CI, 1.001-1.005; P = .0003) using IVW regression analysis. The IVW model showed that for every 1-standard deviation increase in BMI, the estimated risk of developing RCS increased by 1.003-fold
Diabetes, HBP, and HDL-C have been found to contribute to rotator cuff tendon quality deterioration, accelerating tearing. A systematic review and meta-analysis identified 22 factors associated with symptomatic rotator cuff tears and analyzed 5 factors. 23 The pooled analysis demonstrated that age >50 years, diabetes, and excessive physical activity were associated with an increased risk of rotator cuff tendon lesions. Park et al 27 evaluated 634 participants diagnosed with supraspinatus tears on MRI and found that metabolic factors such as diabetes, BMI, low HDL-C, and metabolic syndrome were independent factors significantly associated with posterior superior rotator cuff tears. These results demonstrate the importance of intrinsic factors in the development of shoulder injuries and lay the foundation for further investigations of causality using MR analysis.
BMI is a known risk factor for RCS. For instance, Gumina et al 16 measured BMI and body fat percentage stratified by age and sex in 381 consecutive patients undergoing arthroscopic rotator cuff repair. The results of their multivariate linear regression analysis showed that these 2 factors were associated with the occurrence and severity of rotator cuff tears. Similarly, Wendelboe et al 37 found that high BMI was a risk factor for rotator cuff tendinopathy and related diseases in 311 patients aged between 53 and 77 years, consistent with other studies.36,26 In a 2023 study, Siddiqui et al 30 investigated the impact of increased BMI on tendons using strain elastography and finite element analysis and reported that obese individuals were more prone to tendon injuries due to increased tendon thickness, which causes bulging under higher loads. Further, BMI can influence the retear rate and rotator cuff healing after surgery. In this respect, Ateschrang et al 2 found that BMI >30 kg/m2 was associated with higher retear rates regardless of whether patients underwent arthroscopic or open surgery, consistent with the results of our MR analysis indicating a positive correlation between BMI and the risk of RCS.
In our study, T2DM was not associated with an increased risk of RCS. However, the association between these 2 factors is controversial. Titchener et al 34 found that diabetes was a risk factor for rotator cuff disease in their study of 5000 patients. Siu et al 31 evaluated diabetic and nondiabetic patients with rotator cuff tears and found that diabetes was associated with increased interleukin (IL)-1β expression in the subacromial fluid. Moreover, IL-1β expression was positively associated with shoulder stiffness and functional decline. In 2022, Yoshikawa et al 40 studied the effects of diabetes-induced glycation and oxidative stress on nontraumatic rotator cuff tears in 20 patients >50 years and found that diabetes was associated with (1) the upregulation of NADPH oxidase (NOX)-1, NOX-4, IL-6, the receptor for advanced glycation end products (RAGEs), type III collagen, matrix metalloproteinase (MMP)-2, tissue inhibitor of metalloproteinases (TIMP)-1, and TIMP-2; (2) decreased protein expression of type I collagen; (3) a significant increase in the number of reactive oxygen species (ROS)–positive cells and apoptotic cells; and (4) elevated levels of advanced glycation end products (AGEs) and their cognate receptors. These results indicate that diabetes-induced hyperglycemia increases the expression of AGEs and cognate receptors in the shoulder, increasing NOX expression, ROS production, and cell apoptosis.
Observational studies suggest that diabetes is not related to rotator cuff tears. For instance, Zhao et al 41 reviewed 18 articles from 14 countries, including a total of 9809 patients (3164 in the study group and 6645 in the control group) and found that diabetes was not significantly associated with an increased risk of rotator cuff tears, which was consistent with our results. Moreover, diabetes has been associated with a higher risk of undergoing rotator cut repair surgery but not with rotator cuff revision surgery. 32 Retrospective studies have suggested that diabetes was a risk factor for rotator cuff disease but did not distinguish between type 1 and type 2 diabetes. Notwithstanding, the results of an MR analysis suggest that type 1 diabetes is a risk factor for rotator cuff tendinitis. 14 Moreover, the effects of T2DM on rotator cuff disease are unknown.
We found no direct causal relationship between HBP and RCS, which differs from the results of retrospective studies. Zhao et al 41 demonstrated that hypertension was associated with rotator cuff tear. Giri et al 13 found a potential correlation between hypertension and rotator cuff tears. Gumina et al 15 found no association between hypertension and the risk of small rotator cuff tears. However, hypertension was a significant risk factor for the occurrence of large tears; for instance, hypertension increased the risk of large and massive tears by 2-fold and 4-fold, respectively. 15 Nonetheless, the role of hypertension as a risk factor is debatable, and underlying mechanisms are unknown.
Abnormal blood lipids involve an increase in low-density lipoprotein cholesterol and a decrease in HDL-C. Lai et al 22 found that dyslipidemia was associated with worse outcomes in patients receiving treatment for rotator cuff tears; high triglycerides and low HDL-C had the greatest impact. Park et al 27 showed that low HDL-C was significantly associated with atraumatic posterosuperior rotator cuff tears. However, additional clinical and mechanistic studies are needed to analyze the association between HDL-C and rotator cuff tears.
Limitations
This study has limitations. First, the populations in the analyzed datasets were of European ancestry, potentially leading to bias. Second, the effects of other potential risk factors for RCS—including type 1 diabetes, metabolic diseases, and osteoarthritis—were not analyzed. Third, this study did not evaluate underlying mechanisms because of time and resource constraints.
Conclusion
BMI was positively associated with the risk of developing RCS, and T2DM, HBP, and low HDL-C were not causally associated with RCS but might have indirect effects. The 2-sample MR method was found to be efficient in making causal inferences. We used stable genetic variations as instrumental variables and conducted multiple screenings to reduce the influence of confounding factors. In addition, we selected multiple instrumental variables to increase accuracy.
Footnotes
Appendix
Sources for the T2DM, BMI, HBP, HDL-C, and RCS Datasets a

| Dataset | GWAS Data Source (IEU Open GWAS Project website) |
|---|---|
| T2DM | https://gwas.mrcieu.ac.uk/datasets/ebi-a-GCST006867 |
| BMI | https://gwas.mrcieu.ac.uk/datasets/ieu-b-40 |
| HBP | https://gwas.mrcieu.ac.uk/datasets/finn-b-I9_HYPTENS |
| HDL-C | https://gwas.mrcieu.ac.uk/datasets/ieu-b-109 |
| RCS | https://gwas.mrcieu.ac.uk/datasets/ukb-b-50 |
BMI, body mass index; GWAS, genome-wide association study; HBP, high blood pressure; HDL-C, high-density lipoprotein cholesterol; IEU, MRC [Medical Research Council] Integrative Epidemiology Unit at the University of Bristol; RCS, rotator cuff syndrome; T2DM, type 2 diabetes mellitus.
Final revision submitted April 9, 2024; accepted April 17, 2024.
The authors have declared that there are no conflicts of interest in the authorship and publication of this contribution. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
