Abstract
Objective
To analyse the efficacy of low-dose rosuvastatin for treating hypo high-density lipoprotein (HDL) cholesterolaemia in patients with type 2 diabetes and dyslipidaemia.
Methods
Patients with HDL-cholesterol (C) <40 mg/dl and triglycerides (TG) <400 mg/dl who were receiving treatment with lipid-lowering drugs other than rosuvastatin (or previously untreated with lipid-lowering drugs) and with low-density lipoprotein [LDL]-C ≥120 mg/dl were included. Patients were treated with 2.5 or 5 mg rosuvastatin orally, once daily, to achieve the target LDL-C level specified in Japanese guidelines. Changes in total cholesterol, HDL-C, TG, LDL-C, LDL-C/HDL-C and non-HDL-C at 3 and 6 months were prospectively analysed. Safety was evaluated by examining changes in hepatorenal function, glucose metabolism and creatine kinase.
Results
Out of 49 patients, all lipid parameters other than TG were significantly improved at 3 and 6 months. At 3 months, 83.3% of patients had achieved the target LDL-C level. Among nonlipid parameters, no changes were observed except for estimated glomerular filtration rate, which was improved by + 5.2% and + 9.6% at 3 and 6 months, respectively.
Conclusions
Low-dose rosuvastatin was effective in improving hypo-HDL cholesterolaemia and may have renoprotective effects.
Keywords
Introduction
Smoking, dyslipidaemia, hypertension, obesity, diabetes and lifestyle are known risk factors for cardiovascular diseases such as coronary heart disease. Type 2 diabetes – which accounts for the majority of diabetes cases and is usually accompanied by obesity – is often associated with dyslipidaemia, including hypo high-density lipoprotein (HDL) cholesterolaemia, hyper low-density lipoprotein (LDL) cholesterolaemia and hypertriglyceridaemia.1,2 Many patients with type 2 diabetes, therefore, are at heightened risk of cardiovascular disease because of concomitant dyslipidaemia, which is also associated with insulin resistance. Consequently, these patients require active intervention for correction of dyslipidaemia in addition to treatment for diabetes. Specifically, low HDL-cholesterol (C) and high LDL-C levels have been reported to be independent risk factors for developing cardiovascular disease,3–5 therefore, patients with diabetes and both of these risk factors require careful and strict management of LDL-C and HDL-C levels, in addition to adequate control of blood glucose. 6
Statins have been reported to improve hyper-LDL cholesterolaemia and hypertriglyceridaemia, to increase HDL-C, and to reduce the risk for cardiovascular disease in patients with type 2 diabetes and dyslipidaemia. 7 Meta-analyses using the PubMed and LIVALO® (pitavastatin) effectiveness and safety (LIVES) Study databases have also shown that various statins can increase HDL-C.8,9 A statin-induced increase in HDL-C is usually accompanied by a decrease in LDL-C and may be primarily explained by a cholesterol ester transfer protein-mediated mechanism. 8 In addition, there may be different mechanisms involved in the effects of different statins. For example, atorvastatin has been shown to increase HDL-C in a dose-independent manner, whereas rosuvastatin was more effective in increasing HDL-C at lower doses.8,10 Clinical studies that exclusively include patients with diabetes and hypo-HDL cholesterolaemia may reveal the mechanisms involved in statin-mediated HDL-C increase, however, no such studies are currently reported. There are also currently no reports on the differences in clinical characteristics between patients who respond to statin therapy and those who do not, nor are there studies on the prediction of statin response.
The present study was conducted to evaluate the efficacy of rosuvastatin (which is a potent hydrophilic statin routinely used in Japan, that decreases LDL-C and also markedly increases HDL-C 10 ) in patients with type 2 diabetes and hypo-HDL cholesterolaemia. Treatment with rosuvastatin aimed to achieve the target LDL-C levels specified in the Japan Atherosclerosis Society guidelines for the diagnosis and prevention of atherosclerotic cardiovascular diseases in Japan–2012 version. 11 In addition, lipid improvement, safety and factors that affected changes in HDL-C at 3 and 6 months were evaluated.
Patients and methods
Study population
Patients diagnosed by an investigator (T.K., M.M., S.K., T.M., M.U., S.T., M.H., T.I., A.O. or Y.T.) as having type 2 diabetes, who also had levels of HDL-C <40 mg/dl and triglyceride (TG) <400 mg/dl, and were under regular treatment at the following institutes in Yokohama, Japan: St Marianna University School of Medicine Yokohama City Seibu Hospital; National Hospital Organization Yokohama Medical Centre; Saiseikai Yokohamashi Tobu Hospital; St Marianna University School of Medicine, between December 2011 and April 2013, were enrolled into this prospective intervention study. Regardless of LDL-C value, patients with HDL-C <40 mg/dl and TG <400 mg/dl who were under treatment with lipid-lowering drugs other than rosvastatin were included. Patients with HDL-C <40 mg/dl and TG <400 mg/dl who were previously untreated with lipid-lowering drugs, with LDL-C ≥120 mg/dl, were also included. Patients meeting any of the following criteria were excluded from the study: familial hypercholesterolaemia; history of hypersensitivity to statins; active hepatic disease; renal dysfunction; serum creatine kinase (CK) >1000 IU/l; receiving cyclosporine therapy; gravidity or potential gravidity; evidence or family history of hypothyroidism or hereditary muscular disorder (muscular dystrophy, etc.); history of drug-induced muscle disorder; drug misuse or alcoholism; patients who, in the opinion of the investigator, were not appropriate for the study. Patients who were already receiving other statin therapies did not undergo any washout or transition phase before they started on rosuvastatin.
The study protocol was approved by each institutional ethical review board (authorization approval Nos: 1961, St Marianna University School of Medicine Yokohama City Seibu Hospital and St Marianna University School of Medicine; 20120004, National Hospital Organization Yokohama Medical Centre; 2011035, Saiseikai Yokohama City Tobu Hospital). Written informed consent was obtained from each patient.
Study design and evaluation
Patients were treated with 2.5 or 5 mg rosuvastatin orally, once daily, to achieve the target LDL-C levels of <120 mg/dl for primary prevention category III or <100 mg/dl for secondary prevention, in accordance with the Japan Atherosclerosis Society guidelines for the diagnosis and prevention of atherosclerotic cardiovascular diseases in Japan–2012 version. 11 Baseline blood lipid parameters were measured immediately prior to the start of rosuvastatin treatment. Changes in blood lipid parameters (total cholesterol, HDL-C and TG), proportion of patients who achieved the treatment goal, and factors that affected the change in HDL-C at 3 and 6 months following the start of treatment with rosuvastatin were analysed. In principle, changes in nonstudy drugs (including dose change), or in therapies that may affect lipid parameters, were not permitted during the study. Dosage increase and/or switching of antidiabetics were permitted for patients with glycosylated haemoglobin (HbA1c) levels >8.0%, if the attending physician judged it necessary.
The LDL-C level was calculated using the Friedewald formula; 12 the LDL-C/HDL-C ratio was calculated by dividing the calculated LDL-C level by HDL-C, and the non-HDL-C level was calculated by subtracting HDL-C from total cholesterol.
Safety was evaluated by analysing changes in HbA1c estimated glomerular filtration rate (eGFR), aspartate aminotransferase (AST), alanine aminotransferase (ALT), γ-glutamyl transferase (γ-GT), CK and blood urea nitrogen (BUN). HbA1c levels were expressed in units specified by the National Glycohemoglobin Standardization Program (NGSP), 13 which is the international standard (see below).
Measurement
Blood samples (10 ml) were taken from the antecubital vein at 08:00–09:00 h following an overnight fast. Samples were immediately centrifuged at 1 500
Blood glucose was measured using Sica Liquid Glu (Kanto Kagaku, Tokyo, Japan) according to the manufacturer’s instructions. HbA1c was determined by a latex cohesion method (Determiner HbA1c, Kyowa Medex, Tokyo, Japan), calibrated against a Japanese Clinical Laboratory Use Certified Reference Material (JCCRM), and designated as HbA1c (Japanese Diabetes Society [JDS]). HbA1c (NGSP) was calculated from HbA1c using the formula: HbA1c (NGSP) (%) = 1.02 × HbA1c (JDS) (%) + 0.25 (%). 13
Estimated GFR was calculated from serum creatinine (Cr) concentration using the revised equation: eGFR (ml/min/1.73 m2) = 194 × Cr−1.094 × Age−0.287 (× 0.739, if female). 14 AST, ALT, γ-GT, CK, Cr and BUN were determined using enzyme immunoassays (Iatro-LQ AST, ALT, γ-GT, CK Rate, CRE and UN Rate; Mitsubishi Kagaku Bio-Clinical Laboratories). Patients were assessed for chronic kidney disease using the definitions and staging system provided in the Japanese Clinical Practice Guidebook for Diagnosis and Treatment of Chronic Kidney Disease – 2012. 15
Statistical analyses
Data were presented as mean ± SD. Paired
Results
Patient characteristics
Demographic and baseline clinical characteristics of patients with type 2 diabetes and hypo high-density lipoprotein cholesterolaemia (
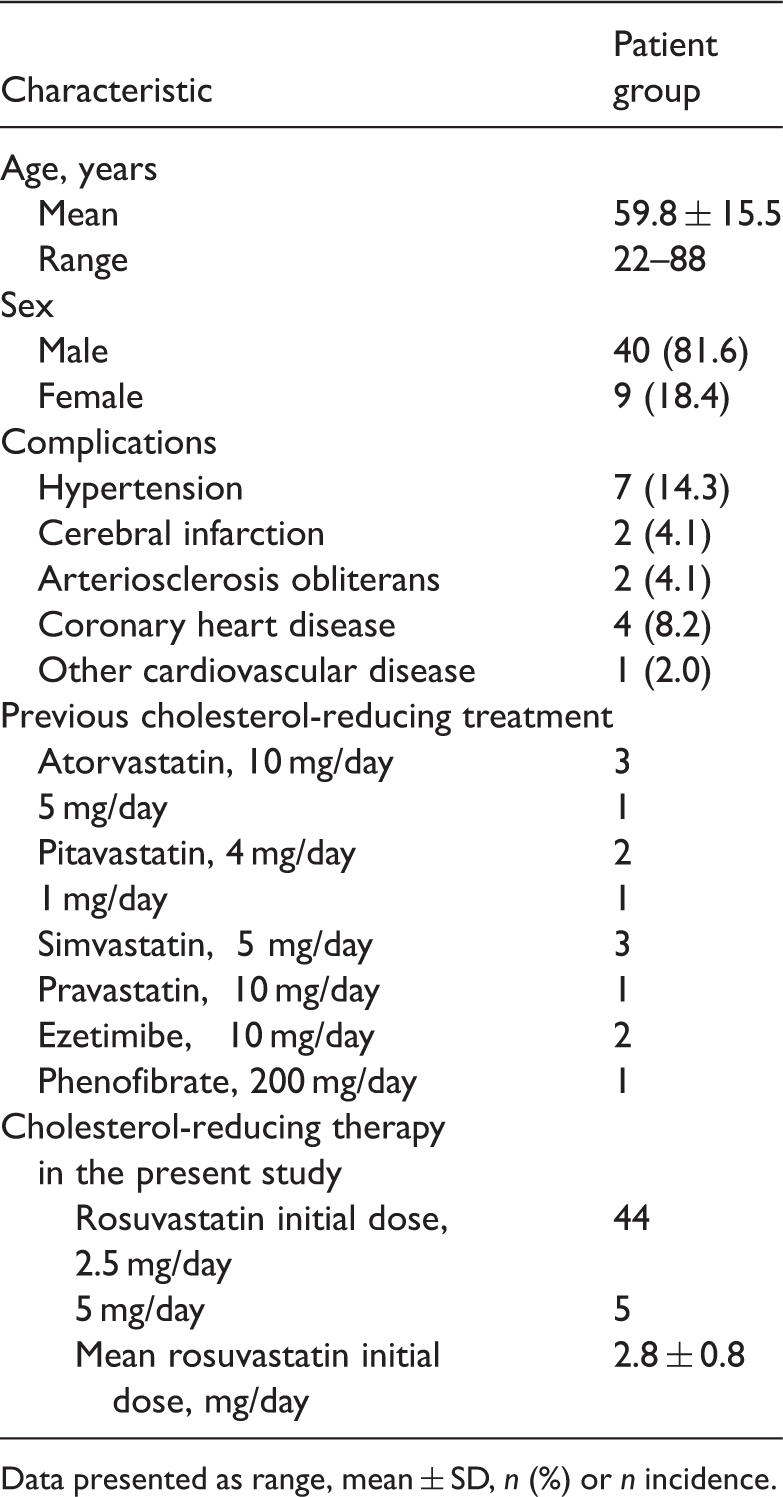
Data presented as range, mean ± SD,
Antidiabetic treatments used during the present study by patients with type 2 diabetes and hypo high-density lipoprotein cholesterolaemia (
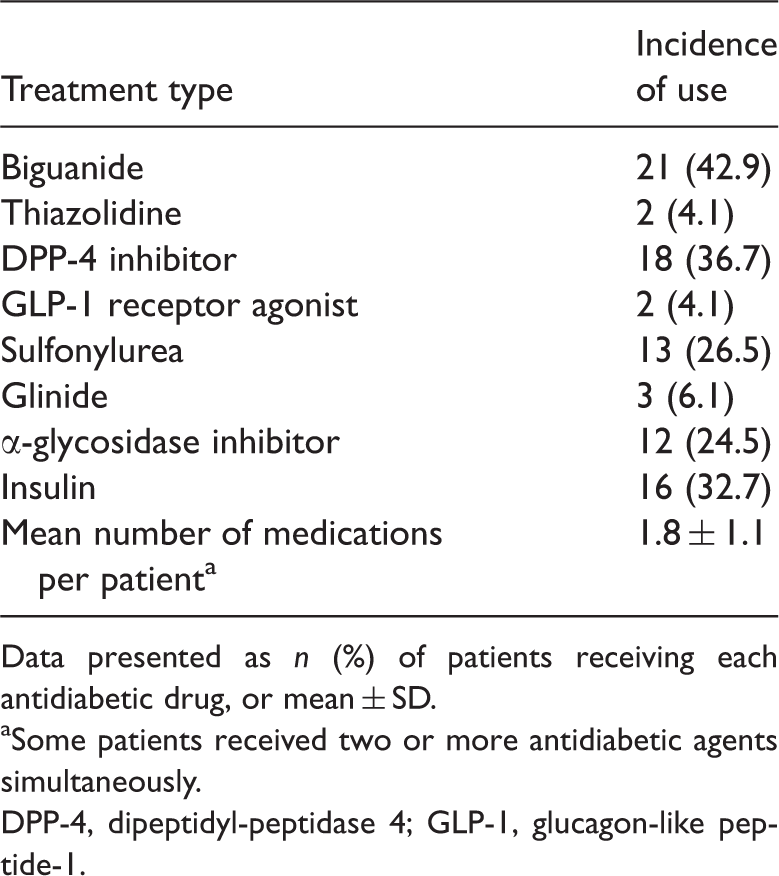
Data presented as
Some patients received two or more antidiabetic agents simultaneously.
DPP-4, dipeptidyl-peptidase 4; GLP-1, glucagon-like peptide-1.
Effect of rosuvastatin (2.5 mg or 5 mg, orally, per day) on lipoprotein and triglyceride levels in patients with type 2 diabetes and hypo high-density lipoprotein cholesterolaemia (

Data presented as mean ± SD or %.
Percentages calculated as the average of the change rate for each patient.
NS, no statistically significant difference (
TC, total cholesterol; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; TG, triglyceride.
Effect of rosuvastatin (2.5 mg or 5 mg, orally, per day) on glucose metabolism and renal/hepatic function in patients with type 2 diabetes and hypo high-density lipoprotein cholesterolaemia (

Data presented as mean ± SD.
NS, no statistically significant difference (
HbA1c, glycosylated haemoglobin; NGSP, National Glycohemoglobin Standardization Program; 13 eGFR, estimated glomerular filtration rate; AST, aspartate aminotransferase; ALT, alanine aminotransferase; γ-GT, γ-glutamyl transpeptidase; CK, creatine kinase; BUN, blood urea nitrogen.
Treatment effect
Statistically significant improvements were observed in total cholesterol, LDL-C, HDL-C, LDL-C/HDL-C ratio, and non-HDL-C at 3 and 6 months ( Achievement rate for low-density lipoprotein cholesterol (LDL-C) target levels set by the Japan Atherosclerosis Society guidelines for the diagnosis and prevention of atherosclerotic cardiovascular diseases in Japan–2012 version,
11
in patients with type 2 diabetes and hypo high-density lipoprotein cholesterolaemia (
Blood biochemistry analysis showed that eGFR significantly improved by + 5.2% at 3 months and + 9.6% at 6 months, compared with baseline values (
There was a statistically significant negative correlation between the change in HDL-C at 3 months and baseline TG levels (
Discussion
This is the first report describing a prospective intervention study in which the effects of a potent statin were studied exclusively in patients with type 2 diabetes, concomitant hypo-HDL-cholesterolaemia and hyper-LDL-cholesterolaemia. In the present study, the mean initial dose of rosuvastatin was low (2.8 ± 0.8 mg per day), because treatment aimed to achieve target LDL-C levels. Despite these low doses, improvements in HDL-C levels were observed at 3 months (+ 12.8%) and 6 months (+ 11.6%). LDL-C levels are considered to be controlled under 70 mg/dl, however, patients with lower HDL-C levels have a higher risk of coronary artery disease than those with higher HDL-C levels. 16 Intensive LDC-C-lowering therapy with rosuvastatin has been shown to result in significant regression of coronary plaque volume in patients with stable coronary artery disease. 17 In that same study, there was a significant, but weak, correlation between the percentage change in total atheroma volume and HDL-C, as well as in the LDL-C/HDL-C ratio. 17 These results indicate that lowering LDL-C and elevating HDL-C might be beneficial to prevent cardiovascular disease events.
Data in the present study suggest that rosuvastatin may increase HDL-C in patients with diabetes who are more likely to develop hypo-HDL cholesterolaemia. Other reports have shown that rosuvastatin can increase HDL-C and decrease LDL-C in a dose-dependent manner,18,19 suggesting that it may be more effective at higher doses than at the low dose used in the present study. A high LDL-C/HDL-C ratio has been reported as a risk factor for cardiovascular events 20 and therefore requires appropriate control. In the present study the LDL-C/HDL-C ratio decreased by 36.2% at 3 months and 32.2% at 6 months, but was not well controlled (mean ratios, 2.48 and 2.37, respectively), suggesting that a further rosuvastatin dose increase would be necessary to prevent cardiovascular events.
Potential factors that may affect the HDL-C-increasing effect of rosuvastatin in patients with type 2 diabetes were examined. HDL-C at 3 months was negatively correlated with baseline TG, suggesting that statins may increase HDL-C less effectively in patients with higher TG. It has long been known that patients with type 2 diabetes have hypertriglyceridaemia and hypo-HDL cholesterolaemia concurrently. In the present study, patients with TG <400 mg/dl were enrolled, with a primary aim of improving LDL-C levels. The relationship between TG and HDL-C in lipid metabolism, and the fact that hypo-HDL cholesterolaemia and hypertriglyceridaemia are independent risk factors for cardiovascular events, support the importance of active TG control.
Multiple regression analysis in the present study suggested that the increase in HDL-C, in response to treatment with rosuvastatin, was smaller in patients with higher TG, higher HbA1c and older age at baseline, compared with other patients. Insulin resistance in patients with type 2 diabetes is known to reduce the effect of insulin, which results in a decrease in lipoprotein lipase activity and an increase in very-low-density lipoprotein synthesis, despite high blood insulin concentrations. 21 These mechanisms may contribute to a less-marked increase in HDL-C in patients who have a higher HbA1c or poor glycaemic control at baseline.
In the present study, TG was shown to have a statistically significant correlation with HDL-C at 3 months in both simple correlation analysis and multiple regression analysis; however, no such correlation was shown at 6 months. This may be in part because fewer patients were followed up at 6 months than at 3 months (26 versus 44 patients, respectively). It may be necessary, nonetheless, to control HDL-C by selecting and adjusting dietary therapy, exercise therapy and pharmacotherapy (including statins) for individuals with the above-mentioned risk factors (increased age, high TG levels and high HbA1c levels).
Nephropathy is one of the most important complications of diabetes.
22
The previously reported renoprotective effects of rosuvastatin23,24 were supported in the present study by the improvements in eGFR observed at both 3 and 6 months, despite inclusion of fewer patients at the 6 month follow-up. Since patients with type 2 diabetes often have hypo-HDL cholesterolaemia and hypertriglyceridaemia concurrently, many patients have high LDL-C/HDL-C ratios despite having not very high total cholesterol levels. It is noteworthy that low-dose rosuvastatin not only improved the lipid parameters, but also improved eGFR in these patients, who are at heightented risk of cardiovascular events. To further examine this renoprotective effect, additional analyses were performed in an attempt to identify a factor or factors that may have contributed to the improvement of eGFR in response to treatment with rosuvastatin. A significant negative correlation was demonstrated between the change in eGFR and the change in LDL-C at 6 months (
While rosuvastatin had no effect on glucose metabolism parameters (such as fasting blood glucose or HbA1c) in the present study, large-scale meta-analyses have reported that the use of statins induced diabetes, although infrequently, and were associated with an increased number of new cases.26,27 It was concluded, however, that conventional statin therapy should not be changed, because the benefits for prevention of cardiovascular events overweigh the glucose metabolism-related risks.26,27 In Japan, no conclusion has been drawn regarding the effect of statins on glucose metabolism in the clinical setting,28,29 but HbA1c was not significantly affected after long-term (6-month) treatment with rosuvastatin in the present study. Given the benefits of statins for prevention of cardiovascular events, it may be crucial to continue statin therapy in patients with type 2 diabetes and dyslipidaemia, while monitoring glucose metabolism-related parameters, including HbA1c. Although all statins (including rosuvastatin) may induce various side-effects, 30 no adverse effects were observed following rosuvastatin therapy, throughout the study period, in the present analysis.
The results of the present study may be limited by the fact that there is no alternative study arm for comparison. In addition, the period of the study was relatively short at 6 months and the patient numbers were relatively low, especially at the 6-month timepoint.
In conclusion, this small-scale 6-month study showed that low-dose rosuvastatin markedly decreased LDL-C and improved low HDL-C levels by 11–12%, in patients with type 2 diabetes and hypo high-density lipoprotein cholesterolaemia. Rosuvastatin treatment appeared to be useful in improving hypo-HDL cholesterolaemia, in addition to other lipid parameters, in these patients. Although the effects of statins on glucose metabolism are yet to be established in Japan,28,29 low-dose rosuvastatin – a hydrophilic statin – had no effect on glucose metabolism in the present study. In addition, low-dose rosuvastatin improved eGFR, a measure of renal function.
Footnotes
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Acknowledgements
We wish to thank the patients, investigators, and their staff for participating in this study.
