Abstract
Background:
Multiligament knee injuries involving the medial side are common. When performing surgical reconstruction, use of the medial hamstrings (HS) as grafts remains controversial in this setting.
Purpose:
To determine the role of the medial HS in stabilizing the valgus knee for different types of medial-sided knee injury.
Study Design:
Controlled laboratory study.
Methods:
A biomechanical study on 10 cadaveric knees was performed. Valgus load (force moment of 10 N/m) was applied at 0°, 30°, and 60° of flexion, and the resultant rotation was recorded using an optoelectronic motion analysis system. Measurements were repeated for 4 different knee states: intact knee, superficial medial collateral ligament (sMCL) injury, deep medial collateral ligament (dMCL) injury, and posterior oblique ligament (POL) injury. For each state, 4 loading conditions (+ loaded; – unloaded) of the semitendinosus (ST) and gracilis (GRA) tendons were tested: ST+/GRA+, ST+/GRA–, ST–/GRA+, and ST–/GRA–.
Results:
At 0° of flexion, combined unloading of the ST and GRA (ST–/GRA–) increased valgus laxity on the intact knee compared with the ST+/GRA+ condition (P < .05). For all medial-sided injury states (isolated sMCL; combined sMCL and dMCL; and combined sMCL, dMCL, and POL damage), ST–/GRA– increased valgus laxity at 0° and 30° of flexion versus ST+/GRA+ (P < .05 for all). The absolute value of valgus laxity increased with the severity of medial-sided ligament injury. Isolated ST unloading increased valgus laxity for the intact knee and the MCL-injured knee (combined sMCL and dMCL) at 0° of flexion (P < .05 vs ST+/GRA+). Isolated unloading of the GRA had no effect on valgus knee stability.
Conclusion:
The medial HS tendons contributed to the stabilization of the knee in valgus, and this was even more important when the medial side was severely affected (POL damage). This stabilizing effect was greater between 0° and 30°, in which the POL is the main valgus stabilizer.
Clinical Relevance:
When deciding on graft selection for multiligament knee injury reconstruction, the surgeon should be aware of the effect of harvesting the medial HS tendon on valgus laxity.
In the context of knee trauma, the medial collateral ligament (MCL) is one of the most frequently injured ligamentous structures (7.9%).5,16 In cases of anterior cruciate ligament (ACL) injury, concurrent MCL injuries occur 20% to 38% of the time.4,8,9,17,20 Medial-side injury (MCL and/or posterior oblique ligament [POL]) is also found in 42% to 53% of cases of multiligament damage to the knee.18,21
Medial stabilization during valgus stress on the knee is provided predominantly by the medial ligamentous structures and musculotendinous structures. The ACL and posterior cruciate ligament (PCL) also contribute as secondary knee stabilizers to valgus stress. The main ligamentous structures of the medial side of the knee are composed of the MCL and POL. The MCL is the major stabilizer of the knee in valgus stresses from 30° of flexion through full flexion. 14 The POL also contributes to valgus stability for flexion degrees between 0° and 30°. 1 Their effects are complementary during the entire range of motion. Additionally, the musculotendinous structures, especially the sartorius and medial hamstrings (HS; consisting of the semitendinosus [ST] and gracilis [GRA]), contribute to the valgus stabilization of the knee.12,13,23
HS tendon autografts are routinely harvested for use during ligament reconstruction.2,19,26 The clinical effect of harvesting of medial HS tendons in the presence of a medial-sided injury is unknown. In a study by Svantesson et al, 24 HS tendon autograft assessed in the setting of associated ACL and MCL injuries had no influence on revision rate. Ateschrang et al 3 also used HS for ACL reconstruction alongside internal MCL bracing. However, in a literature review, Figueroa et al 8 had more cautious conclusions, proposing a medial-sided reconstruction with an allograft and an ACL reconstruction using either a patellar tendon (bone–patellar tendon–bone) or a single HS (4-strand ST graft) for younger patients or with a second allograft for patients with low functional demand.
Biomechanical tudies that analyzed the stabilizing effect of the medial HS in the context of concurrent ACL and MCL injury have demonstrated HS contribution to valgus stability.12,13 However, HS contribution in valgus stability has not been evaluated for more severe medial-plane injuries with POL involvement. Our objective in the current study was to determine the role of the medial HS in stabilizing the valgus knee for different levels of medial-sided knee injury. Our hypothesis was that it would play a significant role in stabilizing the knee in valgus and that this effect would be amplified upon sequential MCL and POL injury.
Methods
Study Design
Ten unilateral human cadaveric specimens were tested. The cadaveric specimens were obtained from the anatomy laboratory of the Jacques Lisfranc Faculty of Medicine in Saint-Étienne, France. Ethical approval for human cadaveric studies was not required by our institution. Exclusion criteria were any history of surgical, traumatic, arthritic, or rheumatic scarring or deformity of the lower extremity that could interfere with the assessment of motion. A complete clinical examination looking for neutral alignment (in frontal and sagittal plane), full range of motion, and knee ligament stability was performed to exclude specimens with pathologic abnormalities that could also influence results.
Specimen Preparation
The bodies were stored between –18°C and –25°C and thawed 48 hours before testing. No other manipulation that could interfere with our study had previously been performed on the bodies. The pelvis and femur were fixed on a static structure to avoid any interfering mobilization of the hip during motion analysis. A circumferential skin flap was removed from the knee. The sartorius, semimembranosus, GRA, ST, quadriceps, and biceps femoris tendons were located and individually overtied with a nonabsorbable suture reinforced with a nylon braided pull cord. The tendons were separately connected to a weight to apply force to each tendon to mimic muscle tone based on previous studies (sartorius, 8 N; semimembranosus, 24 N; GRA, 8 N; ST, 24 N; quadriceps, 200 N; and biceps femoris, 64 N).15,25 A custom-built pulley system was used to apply the forces in the myotome axis corresponding to each tendon throughout the knee flexion extension cycle. Each weight could be disconnected to simulate the absence of a tendon (Figure 1).

Pulley system that permits loading/unloading tendons (sartorius, semimembranosus, gracilis, semitendinosus, quadriceps, biceps femoris). (A) Perspective view. (B) Top view.
Motion Analysis Protocol
A motion analysis system (Motion Analysis) was used to evaluate knee movements. The motion analysis testing procedure was based on a previously published protocol. 22 For lower extremity modeling (definition of tibial and femoral anatomical axes), 4 optical markers per leg segment (F1 to F4 on the femur and T1 to T4 on the tibia) were firmly attached to the tibia and femur using custom-made 3.5-mm bicortical screws.
Five bony landmarks defined by the International Society of Biomechanics11,27 were located by a probe connected to an optical marker for modeling the lower extremity: medial and lateral epicondyle of the knee, culminating bony landmark of the medial and lateral malleoli, and, after an arthrotomy, the center of the tibial spine. The positions were recorded at 100 Hz by 8 synchronized high-definition cameras (Raptor-E) that were installed in the defined analysis area. Static and dynamic calibration of the cameras was performed previously. The markers were positioned in front of the cameras to avoid any interference with the soft tissues during the experiment.
Experimental Protocol
A valgus force of 50 N was applied using a load cell fixed at 0.2 m distal from the center of the knee to produce a valgus force moment of 10 N/m on the joint.12,13 Three cycles of valgus loading and flexion-extension of the knee were performed to remove viscoelastic effects of tissue. Then, with the motion analysis system, valgus laxity (in degrees) of the intact knee was recorded at 0°, 30°, and 60° of flexion while applying this force moment. Valgus laxity measurements were recorded on the ST and GRA tendons under 4 different HS loading conditions (+, loaded; –, unloaded): ST+/GRA+, ST+/GRA–, ST–/GRA+, and ST–/GRA–. Valgus laxity cycles were repeated 3 times for each condition, and the mean value of the 3 values was used for statistical analysis.
This process was repeated on each knee after successively sectioning the superficial MCL (sMCL), the deep MCL (dMCL), and then the POL (on their tibial insertion) to reproduce the most frequent pathophysiological sequence. 7 The ACL and PCL were always left intact during the experimental protocol (Figure 2).

Experimental protocol. Hamstrings (HS) loading conditions: ST+/GRA+, both semitendinosus and gracilis loaded; ST+/GRA–, isolated semitendinosus loading; ST–/GRA+, isolated gracilis loading; ST–/GRA– both semitendinosus and gracilis unloaded. dMCL, deep medial collateral ligament; POL, posterior oblique ligament; sMCL, superficial medial collateral ligament.
Statistical Analysis
Valgus laxity measurements were recorded as means and standard deviations for each knee ligament state (intact knee, sMCL sectioned, sMCL+dMCL sectioned, and sMCL+dMCL+POL sectioned) for the 4 HS loading conditions as well as for each flexion condition. Within-specimen comparison of the data extracted from the 10 knees was conducted with 3-factor repeated-measures analysis of variance, with the 3 independent factors being ligament state (healthy knee, sMCL sectioned, sMCL+dMCL sectioned, sMCL+dMCL+POL sectioned), HS loading condition (ST+/GRA+, ST+/GRA–, ST–/GRA+, ST–/GRA–), and knee flexion angle (0°, 30°, 60°). An initial preliminary analysis was performed to assess the correlation between ligament state and knee valgus. A second analysis focused on the correlation between the effect of HS loading condition and knee valgus as a function of ligament state and flexion angle. The significance level for each analysis was set at P < .05. Statistical analyses were performed using SPSS Version 28 (IBM).
Results
Effects of Medial Plane Lesions on Medial Stability During Valgus Stress
Knee valgus laxity increased significantly (P < .05 for all) after successive sectioning of the sMCL, dMCL, and POL at each knee flexion angle (0°, 30°, 60°) for a force moment of 10 N/m (Table 1).
Valgus Laxity According to Testing State at 0°, 30°, and 60° of Flexion

Data are presented in degrees as mean ± SD. Statistically significant increase in valgus laxity with each successive sectioning condition at each of the 3 knee flexion conditions (P < .05 for all). dMCL, deep medial collateral ligament; POL, posterior oblique ligament; sMCL, superficial medial collateral ligament.
At full extension, the increase in valgus laxity after sectioning all the medial ligamentous structures was 2.65° versus the intact knee (Table 1). The proportion of laxity due to sectioning of the MCL (sMCL+dMCL) was 42% (37% for sMCL and 5% for dMCL), corresponding to an increase in valgus laxity of 1.10° versus the intact knee (0.98° for sMCL and 0.12° for dMCL), as opposed to 58% for the POL (+1.55° compared with the sMCL+dMCL–sectioned knee).
At 30° of flexion, the increase in valgus laxity after sectioning all the medial ligamentous structures was 6.65° versus the intact knee (Table 1), 68% of which was due to sectioning of the MCL (33% for sMCL and 35% for dMCL), which corresponded to an increase of 4.53° versus the intact knee (2.21° for sMCL and 2.32° for dMCL). The proportion of laxity due to sectioning of the POL was 32% (+2.12° compared with the sMCL+dMCL–sectioned knee).
At 60° of flexion, the increase in valgus laxity was of 6.85° versus the intact knee (Table 1). The proportion of laxity due to sectioning of the MCL was 88% (43% for sMCL and 45% for dMCL), corresponding to an increase in valgus laxity of 6.06° versus the intact knee (+2.95° for sMCL and +3.11° for dMCL), and the proportion of laxity due to sectioning of the POL was 12% (+0.79° compared with the sMCL+dMCL–sectioned knee).
Effects of Medial HS Sectioning on Medial Stability During Valgus Stress
Intact Knee
At full extension, combined unloading of ST and GRA (ST–/GRA–) increased valgus laxity by 0.44° (P < .05), isolated ST loading (ST+/GRA–) by 0.20° (P = .25) and isolated GRA loading (ST–/GRA+) by 0.40° (P < .05) in comparison with both tendons loaded (ST+/GRA+). At 30° of flexion, ST–/GRA– increased valgus laxity by 0.19° (P = .14), ST+/GRA– by 0.10° (P = .44), and ST–/GRA+ by 0.19°(P = .18) in comparison with ST+/GRA+. At 60° of flexion, ST–/GRA– showed negligible increases in valgus laxity by 0.07° (P = .72), ST+/GRA– of 0.01 (P = .94), and ST–/GRA+ 0.04 (P = .78) in comparison with ST+/GRA+ (Figure 3).

Knee valgus laxity (degrees) in function of semitendinous (ST) and gracilis (GRA) loading at 0°, 30° and 60° of flexion for an intact knee. ST+/GRA+, both semitendinosus and gracilis loaded; ST+/GRA–, isolated semitendinosus loading; ST–/GRA+, isolated gracilis loading; ST–/GRA– both semitendinosus and gracilis unloaded.
SMCL Sectioning
At full extension, ST–/GRA– increased valgus laxity by 0.43°(P < .05), ST+/GRA– by 0.20° (P < .05), and ST–/GRA+ by 0.30°(P = .16) in comparison with ST+/GRA+. At 30° of flexion, ST–/GRA– increased valgus laxity by 0.52° (P < .05), ST+/GRA– by 0.29 (P = .09), and ST–/GRA+ by 0.37 (P < .05) in comparison with ST+/GRA+. At 60° of flexion, ST–/GRA– led to an increased valgus laxity by 0.36°(P = .06), ST+/GRA– by 0.21° (P = .14), and ST–/GRA+ by 0.25° (P = .1) in comparison with ST+/GRA+ (Figure 4).

Knee valgus laxity (degrees) in function of semitendinous (ST) and gracilis (GRA) loading at 0°, 30°, and 60° after superficial medial collateral ligament sectioning. ST+/GRA+, both semitendinosus and gracilis loaded; ST+/GRA–, isolated semitendinosus loading; ST–/GRA+, isolated gracilis loading; ST–/GRA– both semitendinosus and gracilis unloaded.
SMCL+dMCL Sectioning
At full extension, ST–/GRA– increased valgus laxity by 0.59°(P < .05), ST+/GRA– by 0.26° (P = .35), and ST–/GRA+ by 0.37°(P < .05) in comparison with ST+/GRA+. At 30° of flexion, ST–/GRA– increased valgus laxity by 0.65°(P < .05), ST+/GRA– by 0.4° (P = .08), and ST–/GRA+ by 0.43° (P = .07) in comparison with ST+/GRA+. At 60° of flexion, ST–/GRA– led to an increased valgus laxity by 0.54° (P < .05), ST+/GRA– by 0.28° (P = .14), and ST–/GRA+ by 0.36°(P = .12) in comparison with ST+/GRA+ (Figure 5).
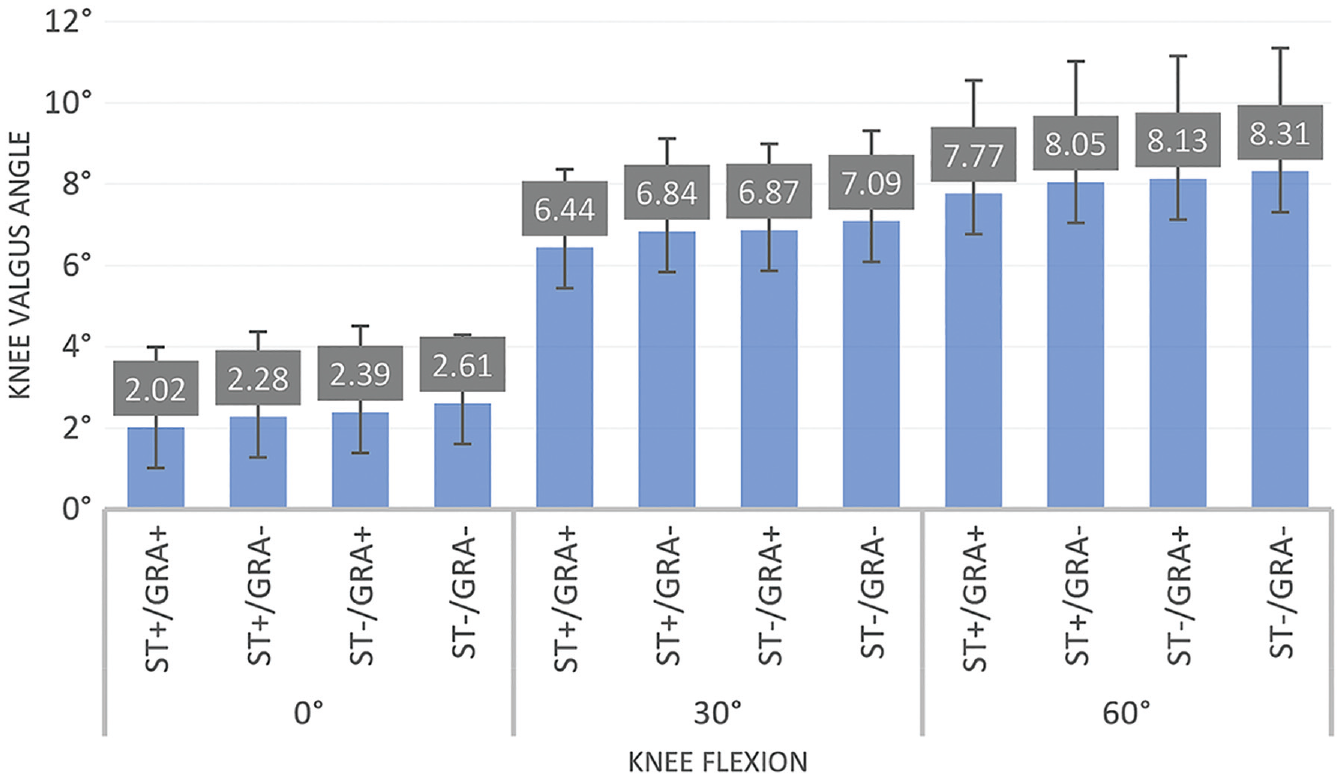
Knee valgus laxity (degrees) in function of semitendinous (ST) and gracilis (GRA) loading at 0°, 30° and 60° of flexion after sMCL+dMCL sectioning. dMCL, deep medial collateral ligament; sMCL, superficial medial collateral ligament. ST+/GRA+, both semitendinosus and gracilis loaded; ST+/GRA–, isolated semitendinosus loading; ST–/GRA+, isolated gracilis loading; ST–/GRA– both semitendinosus and gracilis unloaded.
SMCL+dMCL+ POL Sectioning
At full extension, ST–/GRA– increased valgus laxity by 1.76° (P < .05), ST+/GRA– by 0.42° (P = .38), and ST–/GRA+ by 0.85°(P = .15) in comparison with ST+/GRA+. At 30° of flexion, ST–/GRA– increased valgus laxity by 1.66° (P < .05), ST+/GRA– by 0.25° (P = .55), and ST–/GRA+ by 0.68° (P = .06) in comparison with ST+/GRA+. At 60° of flexion, ST–/GRA– increased valgus laxity by 0.64°(P = .09), ST+/GRA– by 0.48° (P = .2), and ST–/GRA+ by 0.49°(P = .14) in comparison with ST+/GRA+ (Figure 6).

Knee valgus laxity (degrees) in function of semitendinous (ST) and gracilis (GRA) loading at 0°, 30°, and 60° of flexion after sMCL+dMCL+POL sectioning. dMCL, deep medial collateral ligament; POL, posterior oblique ligament; sMCL, superficial medial collateral ligament. ST+/GRA+, both semitendinosus and gracilis loaded; ST+/GRA–, isolated semitendinosus loading; ST–/GRA+, isolated gracilis loading; ST–/GRA– both semitendinosus and gracilis unloaded.
Discussion
Our study showed that the medial HS tendons have a stabilization function in valgus stress on the knee, amplified by the severity of a combined medial plane ligament injury, thereby confirming our hypothesis. This stabilizing effect is most critical in the early range of knee flexion (between 0° and 30°).
Analysis according to ligament status shows us that concomitant unloading of the GRA and ST leads to an increase in valgus laxity. This laxity appears more important when the medial-sided injury is severe. Absolute values of valgus laxity increase proportionally with the severity of medial side damage (in extension, GRA and ST unloading leads to a knee valgus laxity increase of 1.76° for a combined MCL/POL section vs 0.43° for an isolated sMCL section and 0.59° for a dMCL section).
For isolated unloading of either medial-side HS tendon (GRA or ST), the stabilizing effect of the ST appears to be greater than that of GRA, regardless of the ligament condition. Isolated unloading of the ST leads to a statistically significant increase in valgus laxity in full extension on an intact knee and after sectioning of the sMCL and dMCL. In the case of a valgus laxity after sectioning of the MCL and POL, despite a large absolute value (0.85°), isolated ST unloading did not increase laxity in a statistically significant manner (P = .15). These results are clinically relevant in this setting when deciding whether even a single HS graft harvest can be recommended.
When we consider the degrees of knee flexion, the results showed us that the stabilizing effect of the HS is more important for degrees of flexion close to full extension, regardless of the ligament state of the knee. These findings could be explained by the direction of the HS being closer to the direction of the MCL when the knee is close to full extension.
Our results confirm that sectioning of the medial ligamentous structures leads to the expected statistically significant increase in valgus laxity of the knee over the entire range of knee flexion. This laxity increased after successive sectioning of sMCL, dMCL and POL. Between 30° and 60° of flexion, the relative role of the MCL prevailed over that of the POL (68% vs 32% at 30°: 88% vs 12% at 60°). In contrast, in full extension, the POL is predominant for valgus control (42% vs 58%). These results are in line with the literature.7,28 Griffith et al 10 did not find a significant result for medial stabilization during valgus stress with the POL, but this could be explained by a nonconsensus description of the POL in the literature, which may lead to a variation in protocol. These findings also show that the effects of the medial HS are predominant in joint ranges for which the POL is the main valgus stabilizer of the knee. Their valgus stabilizing action are therefore complementary between 0° and 30° of knee flexion. Between 30° and 60° of flexion, valgus knee stabilization is mainly provided by the sMCL and dMCL complex, and at the same time the effect of the medial HS becomes less important in valgus knee stability.
This study provides a rationale for graft selection in the management of multiligament knee injury (MLKI) with medial plane damage. The increasing involvement of the HS in valgus stability according to the severity of the medial plane lesions leads us to consider other options when deciding on the graft choice for MLKI reconstruction including severe medial side injuries.19,28
In case of associated MCL and POL damage in a context of MLKI, surgical reconstruction of the medial side is recommended.6,8 For grafts, the use of ipsilateral HS (ST and GRA) seems to have an impact on valgus stability of the knee in view of our results. Effects of HS tendon harvesting on medial stability should be considered when deciding the management of MLKI.
Limitations
This study has some limitations. This was a biomechanical study, and extrapolation to a clinical scenario must be made with caution. The absolute values of the increase in valgus laxity after unloading the HS remained low (most often <1°), except after sectioning of combined MCL and POL after concomitant unloading of the ST and the GRA. The literature does not provide an answer concerning the laxity threshold that causes clinical consequences; however, it would be logical to minimize this at every opportunity during knee reconstruction. In addition, the results described were collected on cadaveric subjects, which cannot reproduce the physiological varus effect of the loaded knee that could minimize the increase in valgus rotation shown by our study. Also, the static nature of our study does not allow us to consider the adaptations of the physiological muscle tone likely to further decrease the laxity induced by HS harvesting.
Other limitations included the fact that our study focused only on medial-sided injuries; however, in clinical practice, the pattern of medial-sided MLKIs is much more diverse and must be analyzed with care for proposing the best management. We also did not analyze rotational stability or anteroposterior translation, which can be affected by medial-sided injuries. The possibility of an MCL repair was investigated in this study. This option could be an alternative to approach the conditions of a healthy knee. A complementary biomechanical study would be necessary to assess the contribution of the medial HS in case of MCL suture. We also did not consider healing of medial plane damage or regeneration of the HS after harvest, which could change clinical outcomes. Finally, the study was conducted on knees with an intact ACL and PCL. To be extensive, we should also have taken measurements on a knee after ACL and PCL reconstruction to be closer to each MLKI lesion pattern.
Conclusion
The medial HS tendon contributes to the stabilization of the knee in valgus, and this is even more important when the medial side is severely affected (POL damage). This stabilizing effect is greater in the early range of knee flexion (between 0° and 30°) in which POL is the main valgus stabilizer of the knee. When deciding on graft selection for MLKI reconstruction, the surgeon must be aware of the effect of harvesting medial HS tendons on valgus laxity particularly in case of POL injury.
Footnotes
Acknowledgements
The authors acknowledge Jean-François Bellus and Florian Bergandi.
Final revision submitted May 11, 2023; accepted May 22, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.K. has received consulting fees from Stryker and speaking fees from Implantcast. R.P. has received consulting fees from Lepine. B.S.-C. has received consulting fees and royalties from Arthrex. T.N. has received consulting fees from Lepine. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
