Abstract
Background:
Articular cartilage repair remains challenging despite the availability of techniques, including autologous chondrocyte implantation (ACI) for repairing large cartilage defects. Platelet-rich plasma (PRP) therapy, a novel therapy focused on chondrocyte regeneration, needs to be investigated regarding its potential to improve the outcomes of ACI.
Purpose:
To examine the effect of PRP therapy on the outcomes of cartilage repair using the ACI procedure in a rabbit model of knee joint cartilage damage.
Study Design:
Controlled laboratory study.
Methods:
A total of 30 knees in 15 Japanese White rabbits (joint cartilage damage model) were divided into nontreatment (n = 7), PRP (n = 8), ACI (n = 7), and combined ACI and PRP (n = 8) groups. At 4 weeks and 12 weeks postoperatively, histological and visual examination of the surgical site was performed, and the regenerated cartilage and calcified bone areas were measured by imaging the specimens.
Results:
Pretransplantation evaluation in the cultured cartilage showed the histological properties of hyaline cartilage. At 4 weeks postoperatively, the regenerated cartilage area at the surgical site showed a larger safranin O–positive area in the ACI group (2.73 ± 4.46 mm2) than in the combined ACI and PRP group (1.71 ± 2.04 mm2). Calcified bone formation in the ACI group was relatively lower than that in the other groups. Cartilage repair failure occurred in all groups at 12 weeks postoperatively.
Conclusion:
The authors found no positive effects of PRP on the outcomes of ACI in a rabbit model. There was a smaller safranin O–positive region with the addition of PRP to ACI compared with ACI alone. In the subchondral bone, bone formation might have been promoted by PRP.
Clinical Relevance:
Administering PRP at the time of ACI may not have a positive effect and may have deleterious effects on cartilage engraftment and regeneration.
Articular cartilage covers the epiphysis of diarthrodial joints and contributes to lubrication and shock absorption. Histologically, articular cartilage is composed of hyaline cartilage and an abundant extracellular matrix. 20 However, the regenerative capacity of articular cartilage after injury is limited because of the lack of nerves, blood vessels, and lymphatic vessels and its poor cellular components. 20
Articular cartilage damage in the knee joint is common among athletes and the general public. More people engage in sports activities now than previously, which has increased the incidence of articular cartilage injuries. 40 As articular cartilage is more vulnerable to shear rather than compression stress, 31,45 traumatic cartilage damage caused by a twist or torque to the knee joint can occur at any age.
To prevent the progression of osteoarthritis, undergoing surgical cartilage repair as soon as possible for articular cartilage defects would be ideal. Surgical repair of articular cartilage damage includes microfracture, 38 osteochondral autograft transplantation (OAT), 16 osteochondral allograft (OCA) transplantation, 12 and cell-based transplants such as autologous chondrocyte implantation (ACI), 5,33 all of which have presented satisfactory therapeutic results. 1,21,32 Large articular cartilage lesions are best treated with ACI 5,33 or OCA transplantation. 12
In recent years, cell therapies, namely the intra-articular injection of platelet-rich plasma (PRP) or stem cell preparations, have been developed and used as alternative treatments for articular cartilage repair when consecutive treatments fail to show improvement. 29 While PRP injections have been suggested to be more effective than hyaluronic acid injections for pain relief, 17 no significant difference has been observed between these 2 injections regarding the knee functional scores. 47 Similar to the effect of nonoperative therapies, the main effect of PRP therapy is pain relief via anti-inflammatory action rather than via cartilage regeneration. 43 Although a study indicated that PRP is effective for cartilage regeneration, 29 another study showed no effect on full-thickness cartilage defects or severe knee osteoarthritis. 36 As PRP therapy promotes chondrocyte regeneration from the remnant tissue, it may be ineffective for cases of full-thickness chondral defect with no normal remnant cartilage tissue.
In collaborative research with Ochi et al, 33 we previously developed a cultured cartilage product that, in a rabbit model, led to a statistically significant difference between cell transplantation and scaffold transplantation (8 knees in each group; unpublished data). This product, J-TEC's Autologous Cultured Cartilage (JACC), has been clinically used in Japan for ACI. The current study was conducted to examine the effect of adding PRP to the ACI procedure in anticipation of promoting engraftment of transplanted cultured cartilage. This is the first experimental study on PRP administration to JACC.
Methods
Experimental Animals
The protocol for this study received ethics committee approval. We used 15 Japanese White rabbits that were 19 weeks of age and weighed 3.1 to 3.7 kg (Kitayama Labes Co Ltd). They were bred 1 by 1 in a 350 × 500 × 355-mm 3 cage in an environment with a temperature of 23°C ± 3°C and humidity of 55% ± 15% and were fed and watered freely. The experiments were performed after 11 days of acclimation. A total of 30 knees in 15 rabbits were randomly divided into 4 groups to create articular cartilage damage models: nontreatment (n = 7), PRP (n = 8), ACI (n = 7), and combined ACI and PRP (n = 8). The rabbits in each group were divided and sacrificed at 4 weeks (14 knees in 7 rabbits; 3 or 4 knees from each group) and 12 weeks (16 knees in 8 rabbits; 4 knees from each group). Different surgeries were assigned to each of the knees from the same animal. A weight loss ≥20% within 3 days, confirmed via daily weight measurement, was set as the humane endpoint; no individual rabbit showed this level of weight loss.
In the nontreatment group, a knee chondral defect was created and not treated; in the PRP-only group, the PRP solution was administrated alone; in the ACI group, the chondral defect was treated only with ACI; and in the combined ACI and PRP group, PRP solution was administered to the knee chondral defect after treatment with ACI.
Preparation of PRP
An allogenic PRP solution was prepared from a 50-mL blood sample collected from Japanese White rabbits using a Vets set (BS Medical Co Ltd). A 5-mL blood sample with 0.5 mL of ACD-A solution (BD Vacutainer; Becton Dickinson) was centrifuged at 520g at 20°C for 6 minutes. The collected blood plasma component was separated from the centrifuged sample and recentrifuged at 400g at 20°C for 4 minutes. After removing the supernatant, we collected approximately 0.5 mL of the remaining PRP. Then, a total of 5 mL of PRP solution was prepared from the 50-mL blood sample, divided into 100-µL portions, and stored at –80°C. The frozen sample was thawed at room temperature at the time of surgery, and 50 µL of undiluted PRP was applied to the defect with and without ACI.
Preparation of Cultured Cartilage for ACI
Allogenic atelocollagen-embedded cultured cartilage for ACI was prepared in the same way as JACC. Articular cartilage tissue was collected from allogenic Japanese White rabbits aged <4 weeks. Chondrocytes were separated from the collected cartilage tissue using trypsin and collagenase, and a cell suspension of chondrocytes and an atelocollagen implant (Koken Co Ltd) were mixed to produce a 25 mm–diameter cultured cartilage (final cell concentration, 4 × 105 cells/cm3), as described by Tohyama et al. 42 The cartilage was cultured for 16 to 17 days in a medium containing 10% fetal bovine serum, antibiotics, and ascorbic acid derivatives. After washing to remove the fetal bovine serum, the cultured cartilage was immersed in phenol red–free Dulbecco-Valkt Minimum Essential Medium until immediately before use for transplantation. To evaluate the quality of the cultured cartilage, we stained the articular cartilage tissue samples with safranin O/fast green (S-O) and performed immunohistological analysis using an anti–type 2 collagen antibody (Anti-hCL(II), clone: F-57; Kyowa Pharma Chemical Co).
Chondral Defect Creation and ACI Procedure
After placing rats under general anesthesia via intramuscular injection with 1.0 mL/kg of a triple mixed anesthetic (0.5 mL of dexmedetomidine hydrochloride [1.0 mg/mL; Domitor; Orion Corp], 0.4 mL of midazolam [5.0 mg/mL; Dormicum; Maruishi Pharmaceutical Co Ltd], and 0.1 mL of butorphanol tartrate [5.0 mg/mL; Bettlefar; Meiji Seika Pharma Co Ltd]), we opened each knee joint using a subvastus approach and laterally retracted the patella. A 3 mm–deep chondral defect 5 mm in diameter was created at the femoral trochlea center using a surgical bur. In the nontreatment group, the knee incision was closed immediately after the chondral defect was created (Figure 1A). In the PRP group, the knee incision was closed after administering 50 µL of PRP to the chondral defect (Figure 1B).
ACI was performed with reference to the study by Katsube et al. 23 Briefly, the prepared allogenic cultured cartilage sheet was hollowed out using a 5 mm–diameter disposable biopsy punch (Kai Corp) and transplanted into the chondral defect. The dried collagen membrane (Chondro-Gide; Geistlich Pharma AG) was hollowed out using the same-sized biopsy punch, placed to cover the transplanted cultured cartilage, and sutured to the peripheral cartilage rim of the defect at 6 locations using No. 6-0 nonabsorbable nylon sutures. In the ACI group, the knee incision was closed after the ACI procedure for the chondral defect (Figure 1, C-1 and C-2). In the combined ACI and PRP group, ACI was followed by the administration of 50 µL of PRP into the space between the cultured cartilage sheet and the collagen membrane before the knee incision was closed (Figure 1D). The routine for postoperative rabbits was unlimited cage activity.
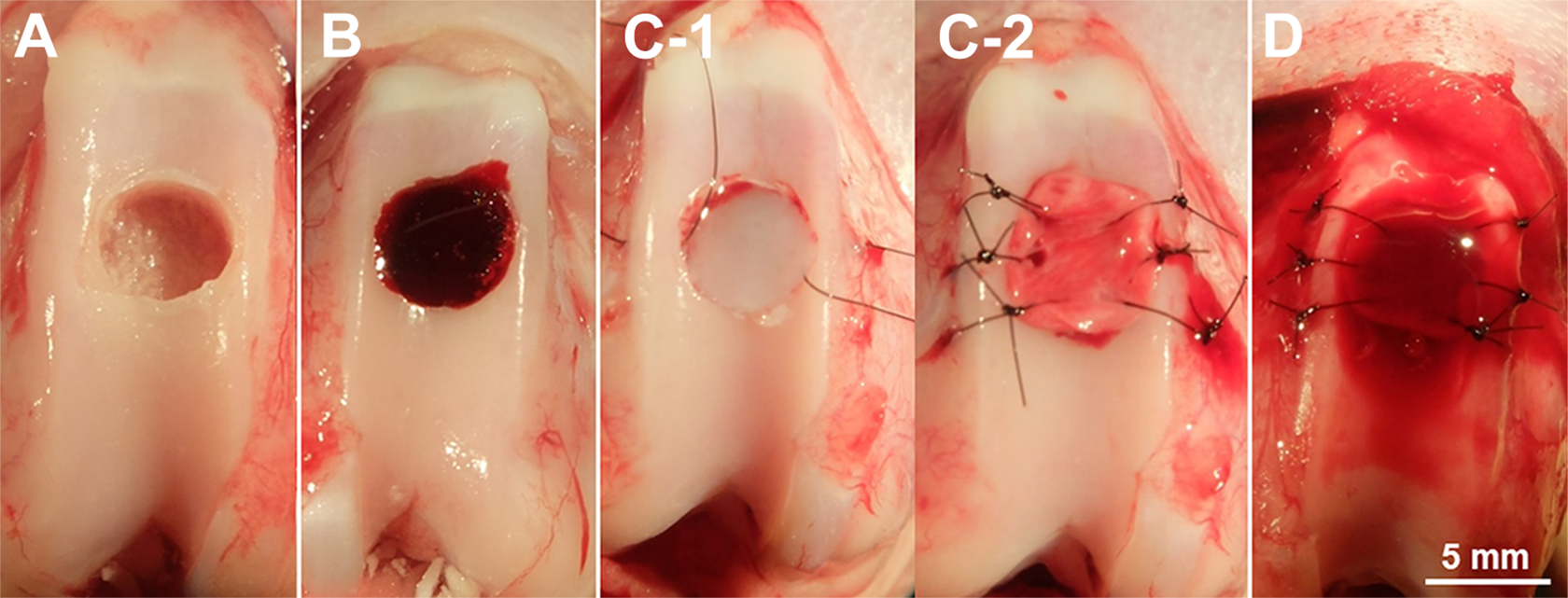
Surgical procedure for administering platelet-rich plasma (PRP) with autologous chondrocyte implantation (ACI). (A) In the nontreatment group, a chondral defect 3 mm deep and 5 mm in diameter was created at the center of the femoral trochlea. (B) In the PRP group, 50 µL of PRP was administered to the chondral defect. (C-1) In the ACI group, a prepared cultured cartilage sheet was hollowed out using a disposable biopsy punch 5 mm in diameter and transplanted into the chondral defect, and (C-2) collagen membrane (Chondro-Gide) was placed to cover the transplanted cultured cartilage and was sutured at the chondral defect. (D) In the combined ACI and PRP group, ACI was followed by the administration of 50 µL of PRP into the space between the cultured cartilage sheet and the collagen membrane.
Histological Evaluation
At 4 weeks and 12 weeks postoperatively, the osteochondral portion of the transplanted area was cut out and fixed using a neutral buffer solution containing 4% paraformaldehyde (Fujifilm Wako Pure Chemical Corp). After decalcification, paraffin embedding was prepared to create tissue slices, which were stained with hematoxylin and eosin (H-E) and S-O to evaluate cartilage regeneration at the implantation site.
Image Analysis
The regenerated cartilage and calcified bone area at 4 and 12 weeks postsurgery were analyzed using ImageJ (US National Institutes of Health). The scale was converted to pixels using ImageJ. The captured images of S-O staining and H-E staining specimens were color separated. After setting the threshold value, we measured the regenerated cartilage area by extracting the S-O–positive region in the grafted site. The calcified bone area was measured by extracting the calcified bone region after color separation of images of H-E–stained tissue slices. These analyses were purely qualitative assessment as a preliminary investigation into the feasibility of the combined technique. No statistical analysis of the measured area was performed.
Results
Pretransplantation Evaluation of Cartilage Matrix in the Cultured Cartilage
The cartilage matrix in the cultured cartilage was evaluated using histological or immunohistochemical staining before transplantation. Chondrocytes in the cultured cartilage showed a 2-layer structure in the atelocollagen gel, and those in the surface layer were stratified into 6 to 7 layers (thickness, approximately 100 µm), which were strongly stained by S-O (Figure 2A). From the middle to deep layer of the cultured cartilage, chondrocytes formed colonies and produced a cartilage matrix around the chondrocytes. Immunohistochemical staining confirmed the expression of type 2 collagen around the chondrocyte layer (Figure 2B). The cultured cartilage before transplantation produced sufficient cartilage matrix and had histological properties of hyaline cartilage.

Cartilage matrix evaluation of cultured cartilage. Cartilage disks 25 mm in diameter (final cell concentration, 4 × 105 cells/cm3) were cultured for 16 to 17 days. After culture, (A) safranin O/fast green staining and (B) immunohistochemical analysis with an anti–type 2 collagen antibody were performed. (A) The cultured cartilage surface was strongly stained by safranin O. From the middle to deep layer of the cultured cartilage, chondrocytes formed colonies and produced a cartilage matrix around the chondrocytes. (B) The type 2 collagen expression was confirmed around the chondrocyte layer. The cultured cartilage before transplantation produced sufficient cartilage matrix and had hyaline cartilage properties.
Macroscopic Observations
Figure 3 shows macroscopic images of the femoral trochlear groove at 4 weeks after surgery. In the nontreatment group (n = 3) (Figure 3A), there was 1 case each in which a slightly transparent white tissue covered the defect, a central depression was seen in the repaired defect, and red tissue was present at the edge of the defect. In the PRP group (n = 4) (Figure 3B), one of the cases showed the whole defect repaired with a slightly transparent white tissue. In the other 3 cases, the presence of a red tissue was observed in poorly repaired parts located in the upper edge, around the whole edge, and at the center of the defect in each image. In the ACI group (n = 3) (Figure 3C), there was 1 case each in which the transplantation site was completely filled with white tissue, the transplantation site was covered by a blood clot, and the center of the defect was only covered by red tissue. In the combined ACI and PRP group (n = 4) (Figure 3D), the transplantation site was repaired with a white tissue (n = 1), the blood clot covered the transplant site (n = 2), and the red tissue partially covered the transplant site (n = 1).

Macroscopic observations at 4 weeks after surgery. (A) In the best specimens of the nontreatment group, the defect was covered with a slightly transparent white tissue. (B) In the platelet-rich plasma (PRP) group, the observations were similar to those in the nontreatment group. (C and D) In the best specimens of the autologous chondrocyte implantation (ACI) group and the combined ACI and PRP group, respectively, the defect was covered with a white, glossy, smooth tissue on one of the transplanted knees.
At 12 weeks after surgery, 2 cases in the nontreatment group showed that the defect was well covered with a soft tissue of the same color as that of the surrounding normal cartilage tissue. Of the remaining 2 cases, one had a tissue of different color from the surrounding cartilage tissue at the center of the defect, and the other had the edge of the repaired defect covered with a tissue whiter than the normal cartilage (Figure 4A). In the PRP group (n = 3) (Figure 4B), 1 case could not be evaluated because of infection in the knee. In the other 2 cases, the defect was covered with a tissue of the same color as the surrounding normal cartilage tissue. The last case showed depression at the center of the defect, although the defect edge was covered with a tissue similar to the surrounding cartilage tissue. In the ACI group (n = 4) (Figure 4C), 1 case showed that the transplant site was repaired with a tissue colored as the surrounding normal cartilage. The next case showed that the center of the transplanted area was partially covered with a white tissue, and another case showed the transplant site was partially repaired with a tissue of similar color to that of the surrounding cartilage. The last case showed depression at the center of the transplant site. In the combined ACI and PRP group (n = 4) (Figure 4D), there was 1 case each in which the transplantation site was repaired with a white tissue, was partially covered with a tissue colored as the surrounding cartilage, was partially covered with a red tissue, and had a depression.

Macroscopic observations at 12 weeks after surgery. (A and B) Images of 2 tissue samples from the nontreatment group and the platelet-rich plasma (PRP) group showing a well-covered chondral defect with a tissue colored similar to the surrounding normal cartilage tissue. One knee in the PRP group was infected. This sample was excluded from the evaluation. (C) One case of the autologous chondrocyte implantation (ACI) group has the grafted area covered with a white tissue. (D) The sample of a case of the combined ACI and PRP group showing a grafted area covered with a white tissue.
Histological Evaluation
H-E and S-O staining were performed to examine the regenerated cartilage and bone formation at 4 weeks after surgery (Figure 5). In the nontreatment group (n = 3), 1 case showed a strong S-O–positive region in the defect up to the same height as that of the surrounding cartilage layer, bone formation in the lower layer, and tissue containing S-O–negative blood cells covering the defect surface. In the cases with depression at the defect site, S-O was faintly positive, and fibrocartilage was observed. Conversely, cases with a red tissue at the edge of the defect revealed the presence of blood cells in the defect, as well as an S-O–positive area and bone formation in the deep layer just distal to the surrounding cartilage (Figure 5, A and E). In the PRP group (n = 4), 3 cases showed weak S-O–positive and fibrocartilage formations in the defect. One case showed remarkable bone formation in the depth of the defect, and another case had many blood cells in the defect. Interestingly, the cartilage matrix and proteoglycan in the cartilage tissue around the defect disappeared in 2 cases (Figure 5, B and F). In the ACI group, a strong S-O–positive and hyaline cartilage formation was observed in 1 case, and a slightly S-O positive region was observed in 2 cases (Figure 5, C and G). In the combined ACI and PRP group (n = 4), weak and partial S-O–positive regions were observed in 2 cases. Many blood cells were present at the transplant sites of 3 cases. Peripheral cartilage proteoglycan disappearance, which was confirmed in the PRP group, was confirmed in 1 case (Figure 5, D and H).

Histological evaluation at 4 weeks after surgery. (A and E) Images showing the strong safranin O/fast green (S-O)–positive region indicating the defect or fibrocartilage in the tissues of the nontreatment group. (B and F) Images showing a weak S-O–positive region or fibrocartilage formation in the defect in the PRP group. We obtained remarkable bone formation in the depth of the defect with many blood cells in it. (C and G) Images of the specimens from the autologous chondrocyte implantation (ACI) group showing a strong S-O–positive region with hyaline cartilage formation and a slightly S-O–positive region. (D and H) Images of specimens from the ACI and platelet-rich plasma combination group showing weak and partial S-O–positive regions. Many blood cells are present at the transplant sites. The arrowhead indicates the boundary between normal cartilage and the defect or transplant site.
Histological evaluations were also performed at 12 weeks after surgery (Figure 6). In the nontreatment group (n = 4), 3 cases showed a thin cartilage layer and bone formation at the defect periphery. Of these cases, only 1 had an S-O–positive region at the center of the defect. Another case, indicating the worst result within the group, showed irregular cartilage tissue regeneration without any S-O-positive region. (Figure 6, A and E). The regenerated cartilage layer was thinner in the PRP group than in the nontreatment group, and bone formation was observed up to the height of the surrounding normal cartilage layer (Figure 6, B and F). In the ACI group (n = 4), 1 case showed partial S-O–positive cartilage regeneration from the superficial to the middle layer of the transplanted cartilage and osteogenesis right under the cartilage layer. Another case showed the presence of fibrous and cartilage tissues in the superficial and middle layers, respectively. The other 2 cases showed weak fibrous cartilage and bone formation at the transplant site (Figure 6, C and G). In the combined ACI and PRP group (n = 4), a strong S-O–positive regenerated cartilage was observed in the middle layer of the transplant site (n = 1), fibrous cartilage and partial cartilage were formed (n = 2), and transplanted sites were covered with soft tissues containing fibrous cartilage (n = 2) (Figure 6, D and H).
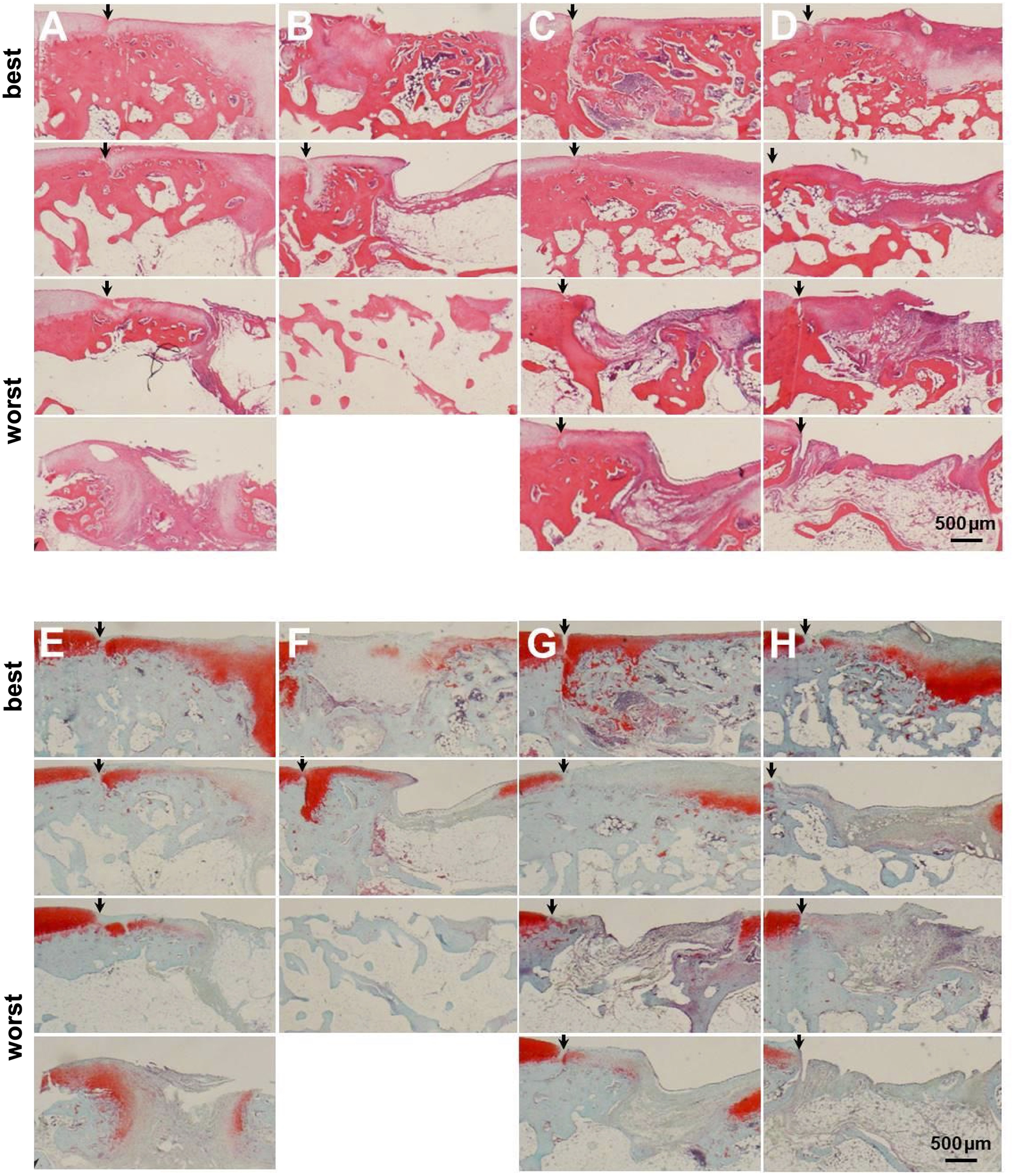
Histological evaluation at 12 weeks after surgery. (A and E) Images of tissue samples from the nontreatment group showing a thin cartilage layer and bone formation at the periphery of the defect. (B and F) Images from tissue samples of the platelet-rich plasma (PRP) group showing the regenerated cartilage layer, which is thinner than that of the nontreatment group, and bone formation can be observed reaching the height of the surrounding normal cartilage layer. (C and G) Images of the autologous chondrocyte implantation (ACI) group specimens, with the best case showing partial safranin O/fast green (S-O)–positive cartilage regeneration and osteogenesis right under the cartilage layer. (D and H) Images of the combined ACI and PRP group specimens, with the best case having strong S-O–positive regenerated cartilage that is observed in the middle layer of the transplant site. The arrowhead indicates the boundary between normal cartilage and the defect or transplant site.
Image Analysis of Regenerated Cartilage
The regenerated cartilage area (S-O–positive region) at the transplantation site was measured from the S-O–stained histological image. At 4 weeks after surgery, 1 case from the ACI group showed cartilage regeneration with a 7.88-mm2 area. Cartilage regeneration was observed in the ACI (n = 3) and combined ACI and PRP (n = 4) groups, with average areas of 2.73 ± 4.46 and 1.71 ± 2.04 mm2, respectively. Conversely, the average areas of cartilage regeneration in the untreated (n = 3) and PRP (n = 4) groups were 0.78 ± 1.13 and 1.02 ± 1.13 mm2, respectively (Figure 7).
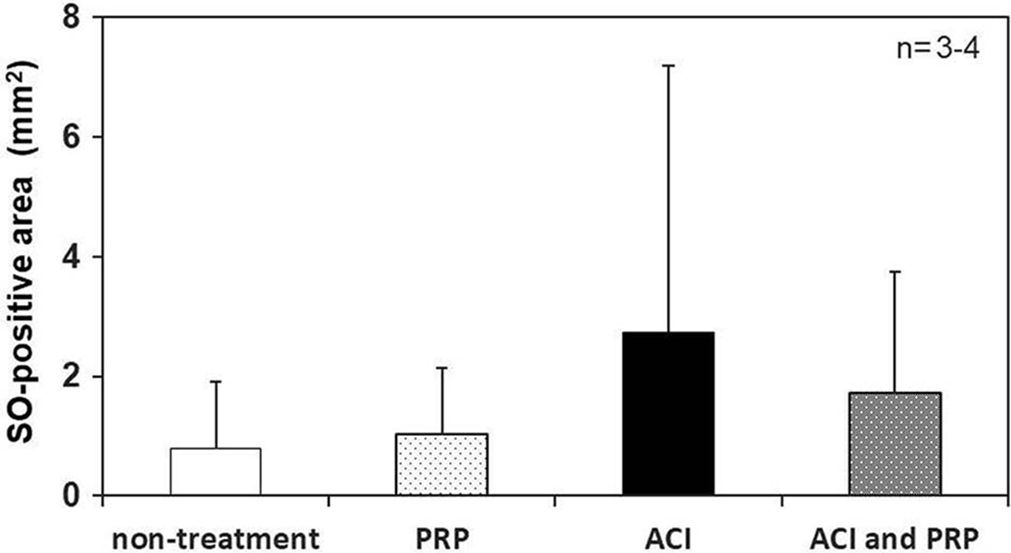
Image analysis of tissue section specimens: area measurement of the regenerated cartilage. ACI, autologous chondrocyte implantation; PRP, platelet-rich plasma; SO, safranin O/fast green.
Image Analysis of the Calcified Bone
The calcified bone area at the transplantation site was also measured from the H-E–stained histological image. At 4 weeks after surgery, the PRP group (n = 4) showed the most calcified bone formation, with an average area of 1.76 ± 0.73 mm2. The second highest bone calcification was observed in the combined ACI and PRP group (n = 4), with an average area of 1.29 ± 0.26 mm2. In the nontreatment (n = 3) and ACI (n = 3) groups, the average areas of calcified bones were 0.99 ± 1.11 and 0.80 ± 0.50 mm2, respectively (Figure 8).
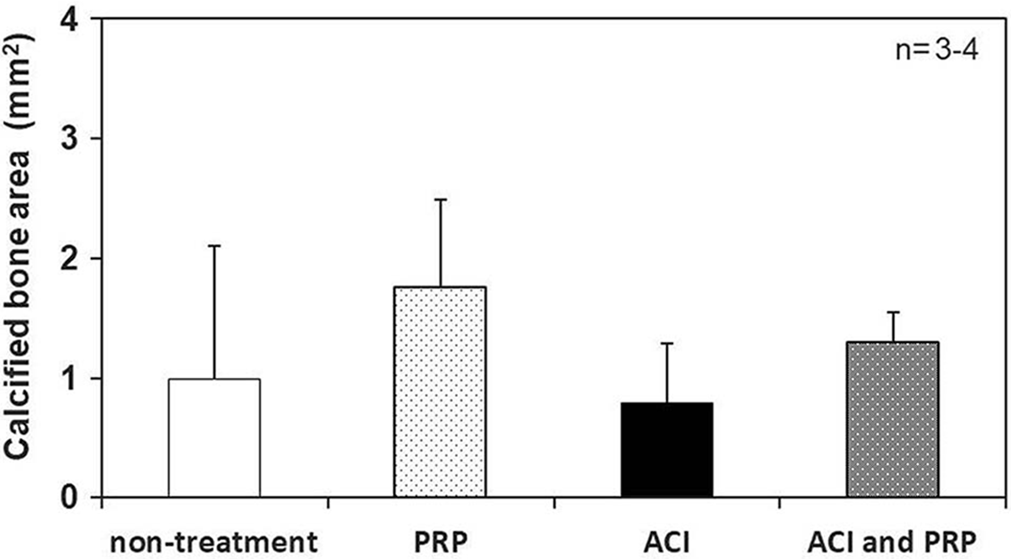
Image analysis of the tissue section specimens: area measurement of the calcified bone. ACI, autologous chondrocyte implantation; PRP, platelet-rich plasma.
Discussion
To our knowledge, this study is the first report to verify the effect of PRP on JACC in an animal model. JACC, which was developed by Ochi et al, 33 was approved by the Japanese Ministry of Health, Labour, and Welfare in 2012 as the first orthopaedic cell therapy in Japan. It has been included in the Japanese health insurance coverage since 2013. In 2019, Chondro-Gide, an artificial collagen membrane, was introduced as an alternative to autologous periosteum that simplified the transplantation procedure. Excellent clinical results of ACI using JACC for large chondral defects have been reported. 21,22 Several investigators have verified the effect of PRP on osteochondral lesions using animal models, 3,6,14,26,27 but only a few have investigated the effects of PRP on ACI. 4 While several clinical studies have investigated the effects of PRP on osteochondral lesions, PRP was administered after microfracture in most of those studies, 11,37 and in only 1 study, PRP was administered after ACI. 15
In this study, no positive effect of PRP on cartilage regeneration of ACI was shown, even for treatment with PRP alone. In the macroscopic observation performed at 4 weeks after surgery, the color tone was most similar to that of the normal cartilage tissue in 1 case in the ACI group. Image analysis of the regenerated cartilage area showed a greater S-O–positive area in the ACI (2.73 ± 4.46 mm2) than in the combined ACI and PRP (1.71 ± 2.04 mm2) group. Brehm et al 4 examined the effect of PRP therapy on the repair of 6 mm–wide osteochondral defects in goats’ knee joints, which were transplanted with scaffold-free autologous bioengineering cartilage constructs, by comparing them with PRP-only, periosteal flap–only, periosteal flap with PRP, and periosteal flap with human fibrin glue treatments. While the periosteal flap was effective, the effect of PRP was negative. 4 Giannini et al 15 divided 81 cases of focal osteochondral lesions of talar dome into an open-field ACI group, an arthroscopic ACI group, and a group with arthroscopic bone marrow–derived cell transplantation with platelet-rich fibrin gel. All 3 groups showed improvement, although not significant. However, their report had results consistent with our findings, showing that PRP itself had no additive effect on cartilage defect repair.
Histological examination performed at 4 weeks after surgery showed that there might be a possible difference in the subchondral bone formation between the groups with and without PRP administration. It can be explained that PRP had no positive effect on ACI cartilage regeneration and had a negative effect on the initial engraftment of the cultured cartilage and the maturation process of the regenerated cartilage. We showed that the calcified bone formation area at the surgical site of the cartilage defect at 4 weeks after surgery was much larger in the groups with PRP alone (the PRP and combined ACI and PRP groups: 1.76 ± 0.73 and 1.29 ± 0.26 mm2, respectively) than in those without PRP (the nontreatment and ACI groups: 0.99 ± 1.11 and 0.80 ± 0.50 mm2, respectively). The calcified bone formation was more advanced at 12 than at 4 weeks after transplantation in all groups (2.15 ± 0.86, 2.68 ± 1.94, 3.00 ± 0.97, and 2.50 ± 0.44 mm2 in the nontreatment, PRP, ACI, and combined ACI and PRP groups, respectively; data not shown). This result suggested that administering PRP alone or in combination with ACI might induce calcified bone formation in the subchondral bone for cartilage defects. Administering PRP may not have a positive effect and may have deleterious effects on cartilage engraftment and regeneration.
Previous studies have reported that an abnormal biological environment in the chronic osteoarthritis model promotes cartilage degeneration; moreover, overexpression of the nerve growth factor, transforming growth factor-β, and vascular endothelial growth factor by osteoblasts from the subchondral bone could inhibit subchondral bone remodeling. 39 Therefore, PRP containing a physiologically active substance may promote remodeling of the subchondral bone, as a positive effect. 10 In our study, ACI was used for the traumatic osteochondral defect rather than direct injection into the joint. Its repair process was initiated by transplanting cultured chondrocytes with the scaffold that has accumulated cartilage-like tissue with the cartilaginous extracellular matrix. Proliferation of chondrocytes in the cultured cartilage column, production, and accumulation of the cartilaginous matrix lead to cartilage regeneration and maturation, followed by subchondral bone formation and remodeling. Our results suggested that PRP might act on subchondral bone remodeling after cultured cartilage transplantation and before cartilage maturation. Thus, it may interfere with ACI engraftment to promote bone formation.
In this study, we adopted an osteochondral defect model with a 5-mm diameter and a 3-mm depth that did not heal spontaneously in the femoral trochlea of Japanese White rabbits. 9 The macroscopic observation at 4 weeks postoperatively revealed that the ACI-treated osteochondral defects were well filled without depression, regardless of PRP treatment. All the defects without ACI were repaired with fibrocartilage, except for 1 case in the defect group with a strongly positive S-O region. One model of the ACI group showed that the chondral defect was repaired with macroscopic normal cartilage tissue with normal hyaline cartilage on histology that stained strongly positive for S-O at 4 weeks after surgery. Although the staining intensities were different, the S-O–positive region was present in all models of the ACI group. Our results suggested that cultured cartilage transplantation could yield cartilage regeneration at 4 weeks after transplantation.
None of the groups achieved cartilage repair at 12 weeks postoperatively. Similarly, Katsube et al 23 used rabbits and reported cartilage repair failure at 24 weeks, possibly attributable to the species-specific difference in the metabolic activity of rabbits and the density of stem cells around the defect 30 —this was considered as a limitation of this experimental model. Larger animals, such as horses 13 and goats, 25 should be used to assess long-term results.
Our study had some limitations. First, the sample size was too small to show the effectiveness of ACI. This was an initial study on PRP with JACC, and further studies can be conducted with larger sample sizes. Second, the timing of PRP administration was not investigated; PRP was administered at the time of transplantation. PRP administration after engraftment of the subchondral bone and the cultured cartilage sheets may demonstrate a positive effect on the regenerated cartilage maturation.
As a third limitation, different preparation methods of PRP were not compared because of their inconsistencies among published studies. We selected the double-centrifugation protocol, as in many other animal studies using PRP. However, as preparation methods of PRP may vary in spin speed and duration, it is difficult to compare the properties of each PRP among studies. Fourth, the effects of cellular components contained in PRP, including the leukocyte content, platelet concentration rate, and platelet cell morphology, were not investigated. Cellular components of PRP differ depending on its preparation method and influence on the concentration of growth factors and cytokines contained therein. 24 Disappearance of proteoglycans in the surrounding normal cartilage, as was observed in 1 case of the PRP and the combined ACI and PRP groups, may suggest the effect of proteases secreted from cellular components in PRP, causing hydrolysis of peptide bonds. 24 The usefulness of pure PRP, which completely removes leukocytes to eliminate their influence on the catabolic effect in tissues and activation of inflammation, has been reported. 44 Future studies should examine the effects of leukocytes contained in PRP.
A fifth limitation was that various PRP forms were not examined; only liquid PRP was used in this study. Titan et al 41 suggested a way to mix PRP with hyaluronic acid scaffold for easy retention at the administration site. PRF, a “second-generation PRP,” is expected to release stable growth factors over 2 to 4 weeks at the administration site because of its gel form. 18 Maruyama et al 27 investigated the difference in cartilage repair of OAT with PRP versus PRF at 12 weeks after surgery, which showed that fibrocartilage was formed in OAT with PRP whereas hyaline cartilage was formed in OAT with PRF. This study could not rule out the possibility of administered PRP diffusing into the cultured cartilage sheet and the knee joint over time. However, we showed that bone formation in the subchondral bone was promoted in the PRP and combined ACI and PRP groups when compared with the other groups without PRP, demonstrating a certain effect of PRP on the osteochondral defect site.
Sixth, the difference in the number of PRP injections could not be examined, as PRP was injected only once at the surgery time. Chouhan et al 8 compared multiple and single PRP injections in a guinea pig model of early knee osteoarthritis, which showed short-term anti-inflammatory effects on synovitis in both groups but long-term anti-inflammatory effects only in the multiple-injections group. Even short-term chondroprotective effects were observed only with multiple injections. As single PRP administration to rabbit joints has only a transient effect, further effects could be expected by administering multiple injections. However, we showed no positive effects on cartilage repair with a single PRP injection. Thus, it is unclear whether multiple injections would have a more positive effect.
As a final limitation, the effects of infusing PRP on synovial joints, joint capsules, ligaments, and other intra-articular tissues and the anti-inflammatory effect of PRP were not evaluated. Many in vitro studies have reported the positive effects of PRP on intra-articular tissues (ie, mesenchymal stem cell induction, cell proliferative ability, anti-inflammatory, and analgesia). 2,28 Recently, PRP was reported to act particularly on synovial-derived cells to increase lubricin production, which reduces the friction coefficient on the surface of articular cartilage, preventing wear. 35 In clinical practice, positive effects of PRP, such as anti-inflammatory and analgesic effects, have been reported. 7 However, this study focused only on the effects of ACI on engraftment and maturation and did not show a positive effect of PRP on ACI. Some in vitro studies have demonstrated that injecting PRP into chondrocytes promotes chondrocyte proliferation. 19,34,46 However, in vivo and in vitro studies differ in the administration of PRP into the knee joint and can affect the target and other sites. Indeed, in this study, PRP, which leaked from the cartilage defect, was suspected to act on other intra-articular tissues. Such in vivo studies with multiple mechanisms of action may have different results from those of in vitro studies.
Conclusion
After examining the effect of PRP on ACI using an animal model, we observed no positive effect. There were greater safranin O–positive regions in ACI without the PRP effect. Bone formation in the subchondral bone was promoted by PRP.
Footnotes
Acknowledgment
The authors thank Mrs. Yuko Yanai for support with manuscript editing.
Final revision submitted November 22, 2021; accepted November 30, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: This study was carried out based on a joint research contract between Japan Tissue Engineering Co Ltd, J-TEC, and Kameda Medical Center, and J-TEC paid for the materials and animals used in the study. This work was supported by a grant for collaborative research from Japan Tissue Engineering Co Ltd. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Japan Tissue Engineering Ethics Committee (approval No. AK2019-002).
