Abstract
Background:
Autologous chondrocyte implantation (ACI) and matrix-induced autologous chondrocyte implantation (MACI) are performed to treat focal chondral defects (FCDs); both are 2-step procedures involving a biopsy, followed by transplantation. There is little published research evaluating ACI/MACI in patients who undergo a biopsy alone.
Purpose:
To determine (1) the value of ACI/MACI cartilage biopsies and concomitant procedures in patients with FCDs of the knee and (2) the conversion rate to cartilage transplantation as well as the rate of reoperation.
Study Design:
Case series; Level of evidence, 4.
Methods:
A retrospective review was performed of 46 patients (63% female) who underwent a MACI (or ACI) biopsy between January 2013 and January 2018. Preoperative data, intraoperative data, and postoperative outcomes were assessed at a minimum of 2 years after the biopsy. The conversion rate from a biopsy to transplantation and the reoperation rate were calculated and analyzed.
Results:
Among the 46 patients included, 17 (37.0%) underwent subsequent surgery, with only 12 undergoing cartilage restoration surgery, for an overall transplantation rate of 26.1%. Of these 12 patients, 9 underwent MACI/ACI, 2 underwent osteochondral allograft transplantation (OCA), and 1 underwent particulated juvenile articular cartilage implantation at 7.2 ± 7.5 months after the biopsy. The reoperation rate was 16.7% (1 patient after MACI/ACI and 1 patient after OCA) at 13.5 ± 2.3 months after transplantation.
Conclusion:
Arthroscopic surgery with debridement, chondroplasty, loose body removal, meniscectomy/meniscal repair, and other treatment approaches of knee compartment abnormalities at the time of a biopsy appeared to be sufficient in improving function and reducing pain in patients with knee FCDs.
Keywords
Focal chondral defects (FCDs) are estimated to affect 56 per 100,000 person-years. 3 Because of the relative metabolic inactivity and avascular status of damaged articular cartilage, there is limited or no healing capacity. Without treatment, these lesions may progress to generalized osteoarthritis over time. 4 While many surgical procedures have been developed to treat focal chondral lesions, autologous chondrocyte implantation (ACI) and matrix-induced autologous chondrocyte implantation (MACI) are among 2 of the newer methods. Briefly, these procedures utilize a 2-step surgical process in which the initial step is a biopsy that is then followed by the second step of transplantation. The purpose of a biopsy is to diagnose the FCD arthroscopically and to harvest a full-thickness cartilage specimen. This specimen is taken from a nonweightbearing region of the knee joint, and chondrocytes are cultured for implantation into the FCD after debridement. 4 While the original first-generation ACI technique utilizes a sutured periosteal cover, the second-generation ACI technique utilizes a collagen scaffold. This eventually led to the third-generation procedure, commonly referred to as MACI, that utilizes a 3-dimensional scaffold. 4
Both ACI and MACI have yielded positive results in treating FCDs. However, there have been mixed results when comparing them with microfracture or debridement. The heterogeneity of ACI/MACI techniques being compared may have contributed to these mixed results, as there has been a significant evolution and increasing ease with subsequent generations of ACI/MACI techniques. Similarly, there have been advances in microfracture favoring nanodrilling to a deeper penetration that has shown promise in animal models and encouraging short-term results. 5,10,11,15 Some studies have shown that ACI/MACI had significantly better clinical outcomes compared with microfracture, 1,13 while other studies have shown no significant difference between the procedures. 6,7,14 Further, at a short-term follow-up of 2 years, ACI/MACI appears to have similar outcomes compared with other knee cartilage procedures but has shown improved outcomes at a long-term follow-up of 10 years in comparison. 12 However, given the lack of high-level evidence and overall mixed results, the increased cost and burden on patients associated with ACI/MACI 2 introduce the question of whether this should be the new standard for treating FCDs.
The purpose of this study was to determine the value of ACI/MACI cartilage biopsies and concomitant procedures in patients with FCDs of the knee and determine the conversion rate to cartilage transplantation as well as the rate of reoperation.
Methods
The protocol for this study was determined to be exempt from institutional review board approval. The study cohort was identified using Current Procedural Terminology (CPT) codes (27412, 27415, 29877, and 29881, respectively) for patients who underwent ACI/MACI, osteochondral allograft transplantation (OCA), debridement/chondroplasty of the knee joint, and meniscal debridement between 2013 and 2018 by 2 board-certified fellowship-trained orthopaedic surgeons (A.F.C. and R.M.F.). All of these CPT codes were necessary for screening patients, as there is no CPT code used solely for ACI/MACI biopsies. The concomitant procedures were used to identify patients of interest via a retrospective chart review.
Patients were included if they underwent a knee cartilage restoration biopsy and a minimum of 2 years had elapsed since undergoing the procedure. Indications for a biopsy included a surface-based, full-thickness grade 4 chondral lesion that was symptomatic (based on preoperative symptoms), without underlying bone marrow edema (based on preoperative magnetic resonance imaging), contained within healthy surrounding cartilage, and larger than 1 × 1 cm in size. A biopsy was not routinely performed for lesions that were smaller than 1 × 1 cm, uncontained, asymptomatic, and/or with bony marrow edema present. Patient demographic data, physical examination findings, operative findings, and advanced imaging results were collected via a retrospective chart review of patient electronic medical records.
Our primary outcome measures were whether patients who had undergone a biopsy eventually underwent cartilage transplantation, specifically ACI/MACI, and whether those who underwent cartilage transplantation then underwent revision surgery. Our secondary outcome measure was the time elapsed between surgical procedures. These were obtained through a chart review.
Statistical Analysis
Descriptive statistics were used to report the number of patients who underwent transplantation versus a biopsy, concomitant procedures, and the time elapsed between procedures. Because a small proportion of patients required surgery after a biopsy, the Fisher exact test was used to determine whether the location or size of the defect was associated with undergoing cartilage transplantation (and specifically, MACI/ACI).
Results
Among 744 patient charts reviewed, a total of 46 patients (63% female) underwent an ACI/MACI biopsy during the study period and were included. The mean age of the patients included in the study was 30.0 ± 10.5 years (range, 14-49 years), and the mean body mass index was 25.2 ± 5.1 kg/m2 (range, 17-42 kg/m2). Overall, 29 patients (63.0%) had small cartilage lesions (<6 mm), while 17 patients (37.0%) had large cartilage lesions (≥6 mm). The locations or sources of the abnormality for the primary biopsy procedure are listed in Table 1. The location of the defect was not significantly associated with patients undergoing cartilage transplantation (P = .4051) or specifically ACI/MACI (P = .2591). Similarly, the size of the defect was not significantly associated with patients undergoing cartilage transplantation (P = .7375) or specifically ACI/MACI (P = .7068).
Location or Source of Abnormality for Primary Biopsy Procedure (N = 46)

The overall conversion rate to cartilage transplantation after an ACI/MACI biopsy was 26.1% (12/46), and the reoperation rate of patients after transplantation was 16.7% (2/12). Several concomitant procedures were performed during the primary biopsy procedure, with the most common being chondroplasty of the patella, which half of the patients underwent. A summary of the concomitant procedures performed during the primary biopsy procedure is provided in Table 2.
Concomitant Procedures Performed During Primary Biopsy Procedure (n = 46) a

a Dashes represent not applicable.
Among the 46 patients included in the study, 17 (37.0%) underwent a secondary procedure at a mean of 8.2 ± 7.8 months after the primary biopsy. Of these patients, 12 (70.6%) underwent cartilage transplantation at a mean of 7.2 ± 7.5 months (range, 1.62-31.37 months) after the biopsy; 9 patients (75.0%) underwent ACI/MACI, 2 patients (16.7%) underwent OCA, and 1 patient (8.3%) underwent particulated juvenile articular cartilage implantation. Clinical indications for proceeding with a transplantation procedure were if symptoms were localized to the area of the defect combined with arthroscopic confirmation of the appropriateness of the defect for transplantation. The decision to proceed with OCA rather than ACI/MACI was made if the lesion had progressed and/or new magnetic resonance imaging showed subchondral bone marrow edema progression. More specifically, the rate of patients who underwent the definitive treatment option, ACI/MACI transplantation, was only 19.6% (9/46). A breakdown of the conversion to cartilage transplantation by location can be found in Table 3.
Conversion to Cartilage Transplantation by Location a
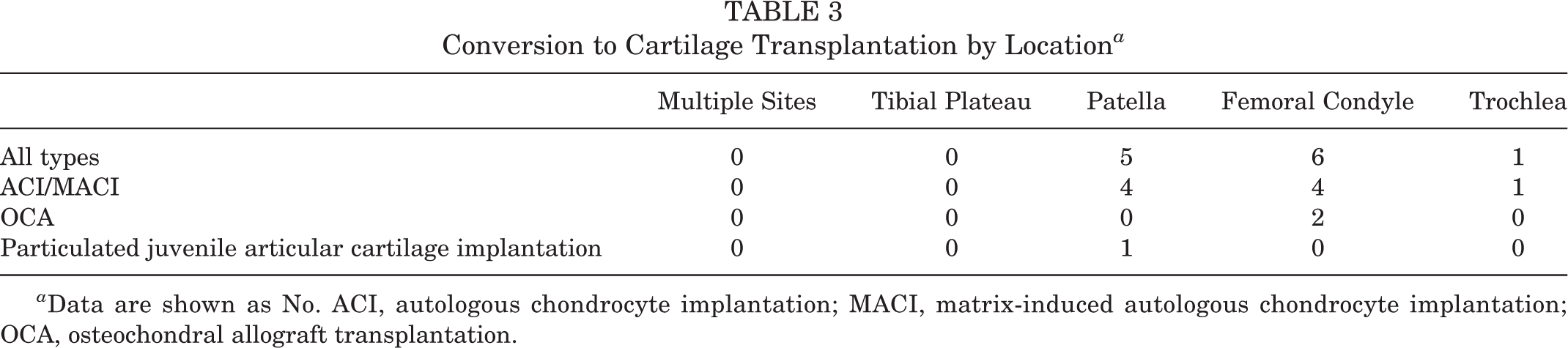
a Data are shown as No. ACI, autologous chondrocyte implantation; MACI, matrix-induced autologous chondrocyte implantation; OCA, osteochondral allograft transplantation.
Of the 12 patients who underwent cartilage transplantation, 7 (58.3%) underwent ≥1 concomitant procedures, including realignment osteotomy (n = 4), platelet-rich plasma augmentation (n = 1), partial meniscectomy (n = 1), meniscal repair (n = 1), medial patellofemoral ligament reconstruction (n = 1), or hardware removal (n = 2 femoral EndoButton (Arthrex) from anterior cruciate ligament reconstruction). Of the 5 patients who underwent secondary procedures but did not undergo cartilage transplantation, 1 underwent hardware removal from prior tibial tubercle osteotomy, 1 underwent high tibial osteotomy, 1 underwent tibial tubercle osteotomy, 1 underwent tibial tubercle osteotomy and lateral retinacular lengthening, and 1 underwent partial medial meniscectomy, removal of loose bodies, and chondroplasty of the patella and lateral tibial plateau. Table 4 shows a summary of the concomitant procedures performed during all secondary procedures after the biopsy.
Concomitant Procedures Performed During Secondary Procedures (n = 17) a

a The mean time between primary and secondary procedures was 8.2 ± 7.8 months. Dashes represent not applicable. ACI, autologous chondrocyte implantation; MACI, matrix-induced autologous chondrocyte implantation; OCA, osteochondral allograft transplantation.
Among the 12 patients who underwent cartilage transplantation, 2 patients (16.7%; 1 ACI/MACI and 1 OCA) underwent a reoperation at a mean of 13.5 ± 2.3 months after initial transplantation. One patient who underwent a secondary procedure but did not proceed with cartilage transplantation required a third operative procedure for the removal of tibial tubercle osteotomy screws. A summary of the concomitant procedures performed during all revision tertiary procedures can be found in Table 5. There were no complications reported in any patient.
Concomitant Procedures Performed During Revision Tertiary Procedures (n = 3) a

a The mean time elapsed from the previous procedure was 13.4 ± 1.9 months. Dashes represent not applicable. ACI, autologous chondrocyte implantation; MACI, matrix-induced autologous chondrocyte implantation; OCA, osteochondral allograft transplantation.
Discussion
The results of this study demonstrate that approximately 75% of patients who underwent an initial ACI/MACI biopsy procedure did not go on to undergo definitive cartilage restoration surgery. Of those who did go on to undergo cartilage restoration surgery, an additional operative procedure was required in one-sixth of cases. This speaks to the ability of arthroscopic surgery with debridement, chondroplasty, loose body removal, meniscectomy/meniscal repair, or other treatment options of knee abnormalities at the time of a biopsy to provide meaningful functional improvements. In fact, FCDs can often be an incidental finding in patients undergoing meniscal repair, partial meniscectomy, or loose body removal. This is an important point, as the initial procedure of a cartilage biopsy for ACI/MACI is not without its own risks. McCarthy et al 9 followed a cohort of patients who underwent cartilage harvest from the knee for ACI of chondral defects of the hip or ankle. Although most patients fared well, there was a group of patients who experienced significant decreases in patient-reported outcome scores, particularly those who had cartilage taken from the intercondylar notch. 9
No prior study has followed patients who only underwent the initial ACI/MACI biopsy procedure and compared their postoperative clinical outcomes with patients who underwent both stages including the transplantation procedure. With an estimated failure rate of 18.5% for first-generation ACI procedures at the 5-year time point since surgery, 8 there may be some merit in avoiding the second stage of ACI/MACI if patients experience significant pain relief from the first surgical stage. A comparison of clinical outcomes between these 2 cohorts can help orthopaedic surgeons better understand if postponing the transplantation procedure, or not performing it altogether, is a valid treatment option. This may help patients avoid unnecessary surgery, the complications associated with surgery, and increased health care costs as well as potentially delay the progression of osteoarthritis for as long as possible.
It is worth mentioning that while it is relatively established that ACI/MACI provides improved results, especially in the long term, the 2-stage procedure is associated with higher costs and raises questions as to what the preferred method for treating FCDs should be. However, when comparing it with other cartilage restoration procedures, the increased costs of ACI/MACI were offset by the higher costs of reoperations associated with other cartilage restoration procedures. 12 Further, in a study performed in the United Kingdom, this procedure was estimated to reduce the lifetime likelihood of knee replacement surgery by 50% as well as provide a better quality of life and substantial lifetime benefits compared with microfracture, deeming it cost-effective as defined by the upper willingness-to-pay threshold used in the United Kingdom. 2 Ultimately, it is imperative that surgeons use their clinical acumen to help determine at the time of arthroscopic surgery whether a patient’s symptoms are truly stemming from an FCD or if they can be addressed without a biopsy. Certainly, this series suggests that surgeons could be more selective in determining when to proceed with a biopsy for future ACI/MACI.
Further analysis of the outcomes relative to both stages of the ACI/MACI procedure may provide insight into increasing the longevity of these results. During the 2-stage ACI/MACI procedure, the second stage of cartilage transplantation can be performed within 5 years of the initial biopsy, as implants are viable for up to that time point. Our study is among the first to investigate the rate of conversion to final cartilage transplantation, providing valuable insight on the results of the initial biopsy as well as the immediate necessity and remaining benefits to be gained by proceeding with definitive treatment. Based on this study, patients undergoing a cartilage biopsy for future ACI/MACI transplantation progress to cartilage restoration surgery only 26.1% of the time at a minimum follow-up of 2 years. This suggests that arthroscopic surgery with debridement and/or chondroplasty of abnormalities in the knee compartment at the time of a biopsy may be a sufficient option to improve function and reduce pain in patients with knee FCDs. Alternatively, this could reflect that the FCDs at the time of a biopsy were not the primary cause of a patient’s symptoms but rather that meniscal lesions or symptomatic loose bodies were. Interestingly, neither the size nor the location of the lesion was significantly associated with patients ultimately proceeding with cartilage transplantation. While these results are preliminary and warrant additional research with greater sample sizes and longer follow-up times, they suggest that concomitant procedures performed alongside the ACI/MACI biopsy may provide longevity to the procedure and can often lead to improved outcomes without the need for a second surgical step. Given the shelf life of biopsy specimens, patients may opt for the second surgical step up to 5 years after an ACI/MACI biopsy. However, while there are no storage costs incurred by patients in the United States from their biopsy if they do not elect to go forward with the definitive second stage of the ACI/MACI procedure, those who do proceed with the second stage absorb these storage costs.
Limitations
One limitation of this study is that we do not know the number of patients who were lost to follow-up, as we collected data from the company that cultured the cartilage specimens (Vericel) to determine if the samples had been used. Although unlikely, it is possible that patients may have undergone another biopsy and transplantation outside of our clinic. In addition, the sample size for our study was small, and data were collected retrospectively, with incomplete data on all previous surgical procedures that patients had undergone before the biopsy, which should be considered when interpreting outcomes. A further limitation of this study is the follow-up time, as a longer follow-up period may provide better understanding of the trends of the ACI/MACI procedure and more accurately determine the longevity potential of the biopsy and concomitant procedures for treating symptoms. Last, there were no outcome measures available for this patient cohort, nor was there standardized imaging available at final follow-up to evaluate for the possible progression of osteoarthritis.
Conclusion
The findings presented in this study illustrate 2 important conclusions. The ACI/MACI biopsy and associated concomitant procedures such as chondroplasty, debridement, and loose body removal have both diagnostic and therapeutic benefits. As such, <40% of our patients who had undergone a biopsy ultimately underwent subsequent surgery in the ensuing 2 years after their initial procedure. Second, delaying the definitive treatment of transplantation can increase the longevity of the native knee. FCDs can be an indication of the progression of osteoarthritis, and the end stage of this disease is total knee arthroplasty. These results warrant further research to better elucidate the clinical outcomes of patients who undergo solely an ACI/MACI biopsy and determine which patients are more likely to require definitive cartilage restoration procedures in the near future.
Footnotes
Final revision submitted December 7, 2022; accepted January 19, 2023.
One or more of the authors has declared the following potential conflict of interest or source of funding: B.J.C. has received research support from Aesculap, Arthrex, and Regentis; education payments from Endo Pharmaceuticals and Medwest; consulting fees from Acumed, Aesculap, Arthrex, Bioventus, Endo Pharmaceuticals, Flexion Therapeutics, Geistlich Pharma, Regentis, Samumed, Smith & Nephew, Vericel, and Zimmer Biomet; speaking fees from Arthrex, LifeNet Health, Pacira Pharmaceuticals, and Terumo BCT; hospitality payments from GE Healthcare; honoraria from Vericel; and royalties from Arthrex and DJO; and has stock/stock options in BandGrip, Ossio, and Regentis. A.F.V. has received research support from Arthrex; consulting fees from Stryker; and hospitality payments from Arthrex, Vericel, and Steris. R.M.F. has received research support from Arthrex and Smith & Nephew; consulting fees from AlloSource, Arthrex, and JRF Ortho; and speaking fees from Ossur. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by the University of Colorado Anschutz Medical Campus (No. APP001-1).
