Abstract
Background:
Third-generation autologous chondrocyte implantation (ACI), also known as matrix-induced ACI (MACI), was approved for clinical practice in December 2016. Studies specifically investigating outcomes of MACI for complex patellofemoral chondral lesions are limited.
Purpose:
To report patient-reported outcome measures (PROMs), complications, and failure rates after MACI for patellofemoral chondral defects at a minimum follow-up of 2 years.
Study Design:
Case series; Level of evidence, 4.
Methods:
For this retrospective review of prospectively collected data, the authors identified patients who underwent treatment with MACI for focal chondral defects in the knee between August 2017 and September 2020. PROMs, including International Knee Documentation Committee (IKDC) score, Kujala score for patellofemoral disorders, and the Veterans RAND 12-item Health Survey (VR-12) score, were obtained preoperatively and a minimum of 2 years postoperatively. The percentage of patients who met the minimal clinically important difference (MCID) for each PROM was reported. Failure was defined as (1) graft failure on follow-up magnetic resonance imaging or second-look arthroscopy, (2) revision MACI or other chondral procedure, or (3) conversion to unicompartmental or total knee arthroplasty.
Results:
A total of 50 patients (34 female; mean age, 32.43 ± 7.33 years; mean follow-up, 2.71 ± 0.79 years) remained after application of the exclusion criteria. There was a significant increase in all PROMs from preoperatively to postoperatively, including the IKDC (43.39 vs 68.58; P < .001), Kujala (58.93 vs 77.07; P < .001), VR-12 Mental (53.12 vs 57.90; P = .002), and VR-12 Physical (40.40 vs 49.89; P < .001) scores, with 5 (10%) failures. The MCID was achieved by 77.8% of patients in IKDC score and 69.0% in the Kujala score. Kaplan-Meier survival analysis showed survival probabilities of 98.0%, 96.0%, and 85.7% at 1, 2, and 4 years, respectively. MACI for patellofemoral bipolar lesions (n = 11) showed significant improvement in IKDC (50.06 vs 74.07; P = .008) and Kujala (69.33 vs 84.33; P = .046) scores, and 2 (18.2%) failures. Kaplan-Meier survival analysis with log-rank test demonstrated no significant differences in survival distributions between unipolar and bipolar patellofemoral lesions (P = .387).
Conclusions:
Third-generation ACI (MACI) is a successful and effective treatment method for difficult-to-treat patellar, trochlear, and bipolar patellofemoral chondral defects.
Keywords
Focal cartilage defects of the knee are a common cause of pain and disability. 19 The patellofemoral joint is one of the most common locations for cartilage defects, 37 typically due to a traumatic patellar dislocation event or chronic repetitive overload. 29 Patellofemoral osteochondral lesions are challenging to treat because of the anatomy of the joint, where there is frequently excessive compression and shear stress as the knee is loaded through flexion and extension.10,39 Additionally, patellar morphology, patellar tilt, limb malalignment, or other patellar tracking issues can contribute to the complexity of managing patellofemoral cartilage defects. 35 As hyaline articular cartilage has limited, if any, intrinsic healing capabilities due to its avascular and hypocellular nature, surgical intervention is the primary treatment method for cartilage restoration.2,7,36 If left untreated, lesions may progress over time, leading to arthritis. 3 In the patellofemoral joint, several techniques have been used, typically dependent on the size of the lesion. For smaller lesions (<2 cm2), debridement, bone marrow stimulation, osteochondral autograft, and particulated cartilage are typical options. 6 Larger lesions are managed with osteochondral allograft (OCA) or autologous chondrocyte implantation (ACI). 6
Because the results of patellar OCAs in the literature have been mixed, with up to a 45% failure rate at 15 years postoperatively, 17 cell-based modalities are being used more frequently for larger patellar cartilage defects. ACI, a 2-stage cartilage repair procedure, first involves arthroscopy of the affected knee for a cartilage biopsy. After a minimum of 3 to 4 weeks for cell growth, the knee undergoes a second mini-open surgery to implant the expanded autologous chondrocytes into the cartilage defect. The study at hand involves third-generation ACI, or matrix-induced ACI (MACI), whereby a stable, porcine collagen membrane carries cultured chondrocytes and is fibrin glued onto the subchondral bone beneath the cartilage lesion. 5 As ACI produces functional hyaline-like repair tissue, good midterm and long-term results have been reported in the knee.1,11,23,33 However, studies specifically investigating the outcomes of MACI for patellar, trochlear, and bipolar patellofemoral chondral lesions are limited. Thus, the aim of this study was to report on the patient-reported outcome measures (PROMs), complications, and failure rates after MACI for patellar, trochlear, and bipolar patellofemoral chondral defects.
Methods
Patient Cohort
For this retrospective review of prospectively collected data, we utilized our institutional review board–approved database to identify 82 patients who underwent cartilage treatment with MACI on porcine collagen membrane for focal chondral defects in the knee by 1 of 2 senior sports medicine fellowship-trained surgeons at a single institution (A.H.G. and S.M.S.) between August 2017 and September 2020. Of the 82 patients who were identified, 50 met the inclusion criteria. Inclusion criteria were (1) treatment with MACI for trochlear and/or patellar grade 3 and 4 chondral defects and (2) minimum 2-year postoperative follow-up with PROMs.
The indication for surgical treatment with MACI included symptomatic full-thickness chondral defects (grades 3 and 4; >2 cm2) of the trochlea and/or patella. All patients failed a minimum of 3 months of nonoperative treatment. Concomitant pathologies were addressed as needed. Patellofemoral maltracking was addressed with tibial tubercle osteotomy (TTO), and patellar instability was treated with medial patellofemoral ligament (MPFL) reconstruction. Concomitant chondral or osteochondral defects of the femoral condyles were addressed with MACI or OCA transplantation. MACI was contraindicated in patients with deep subchondral bone defects and inflammatory or septic joint disease.
Our institution’s electronic medical record system was reviewed to determine patient information such as age, body mass index (BMI), sex, diagnosis, previous procedures on the index knee, follow-up time, concomitant procedures, defect size and location, reoperation for chondroplasty, and failure. First-stage arthroscopy for cartilage biopsy was not counted as a previous procedure. Failure was defined as a (1) poor clinical outcome with graft failure on follow-up magnetic resonance imaging (MRI) or second-look arthroscopy, (2) revision with MACI or other chondral procedure, or (3) conversion to unicompartmental knee arthroplasty or total knee arthroplasty.
Surgical Technique
First Stage
Standard diagnostic arthroscopy is performed, and cartilage lesions are debrided. An arthroscopic probe is used to measure the size of the defect, and a gouge is used to obtain a cartilage biopsy specimen from the periphery of the intercondylar notch at either the medial or lateral trochlea. Approximately 200 to 300 mg of healthy cartilage is harvested and placed in a culture medium, which is then sent to the laboratory for cell growth. The cells are cultured and then seeded onto the rough side of a porcine collagen 1/3 matrix at a density of at least 500,000 cells/cm2 (Vericel Corporation).
Second Stage
After at least 3 to 4 weeks, the patient returns for the second phase of the procedure. A medial or lateral parapatellar arthrotomy is made based on the location of the cartilage defect. A scalpel and a curette are used to debride the base of the chondral defect. A foil template is created, and the membrane is cut. A “cookie cutter” device is used to prepare an appropriately sized graft for the defect. A thin layer of fibrin glue is placed on the subchondral plate of the defect, and the MACI is placed onto the defect with the cell layer facing down. Fibrin glue is then placed at the periphery of the implied membrane. The knee is ranged several times, ensuring full range of motion and membrane stability. The arthrotomy is closed in a standard fashion, and the limb is placed in a hinged knee brace locked in full extension for weightbearing.
Patient-Reported Outcome Measures
PROMs were assessed preoperatively, at 1 year postoperatively, and at ≥2 years postoperatively to evaluate patient pain and function. The questionnaires were completed electronically at their respective time points and included the International Knee Documentation Committee (IKDC) score, 21 the Kujala score for patellofemoral disorders, 24 and the Veterans RAND 12-item Health Survey (VR-12) score. 34 The minimal clinically important difference (MCID) for each PROM was calculated utilizing a distribution-based method. A clinically significant improvement was defined as half of the standard deviation of the mean difference in raw scores between the preoperative and postoperative time points.8,22,25
Statistical Analysis
Descriptive statistics were used to determine patient and lesion characteristics, using means and standard deviations for continuous data and frequencies and percentages for categorical data. Normal distribution was assessed using Shapiro-Wilk tests. Change in PROMs from the preoperative period to the latest follow-up was analyzed using either a paired t test or Wilcoxon rank-sum statistical test. The correlation between reoperation for graft hypertrophy/flap and failure was assessed using the Fisher exact test. Kaplan-Meier survival analysis was performed with survival defined as the absence of failure, as previously described. A comparison of survival between unipolar and bipolar patellofemoral cartilage lesions was conducted using the log-rank test. SPSS Statistics Version 22 (IBM Corp) was used to perform statistical analyses. All statistical tests were 2-tailed. A P value <.05 was considered statistically significant.
Results
Clinical and Surgical Characteristics
In total, 82 patients who underwent cartilage treatment with MACI between August 2017 and September 2020 were identified, 50 (61%) of whom met inclusion criteria. Eight (9.8%) patients did not undergo MACI for the patellofemoral joint, while 24 (29.3%) patients did not reach the minimum follow-up of 2 years. Both groups were excluded from the study. The mean age at the time of surgery was 32.43 ± 7.33 years, the mean BMI was 25.60 ± 5.44 kg/m2, the mean clinical follow-up was 2.71 ± 0.79 years, and 34 (68.0%) patients were female. Preoperative diagnoses included degenerative/chondromalacia in 28 (56.0%) patients, patellofemoral instability in 18 (36.0%), and trauma in 4 (8.0%). Seventeen (34.0%) patients had previous surgeries, and 39 (78.0%) had concomitant procedures, such as TTO (30 patients), MPFL reconstruction (15 patients), condylar MACI (5 patients), OCA (4 patients), ACL reconstruction (2 patients), and meniscal allograft transplantation (1 patient). The mean lesion size was 3.67 ± 1.70 cm2, and the mean time from biopsy to surgery in our cohort was 19.21 ± 17.85 weeks. MACI location was the patella in 35 (70.0%) patients, trochlea in 6 (12.0%) patients, and both the patella and trochlea in 9 (18.0%) patients. Of the 50 patients included in the study, 12 (24.0%) underwent reoperation for hypertrophy/flap and 5 (10.0%) had MACI failure (Table 1). One patient underwent reoperation for hypertrophy/flap before failure. The presence of hypertrophy/flap was not a statistically significant predictor of failure (P > .999). According to the Kaplan-Meier survival analysis, survival probabilities at 1, 2, and 4 years were 98.0%, 96.0%, and 85.7%, respectively (Figure 1).
Patient Characteristics and Operative Conditions a

Data are presented as mean ± SD or n (%). ACL, anterior cruciate ligament; BMI, body mass index; MACI, matrix-induced autologous chondrocyte implantation; MAT, meniscal allograft transplantation; MPFL, medial patellofemoral ligament; OCA, osteochondral allograft; TTO, tibial tubercle osteotomy.

Kaplan-Meier survival analysis for patellofemoral chondral defects treated with matrix-induced autologous chondrocyte implantation. The survival probabilities at 1, 2, and 4 years were 98.0%, 96.0%, and 85.7%, respectively.
Patient-Reported Outcome Measures
Statistically significant improvements in mean IKDC, Kujala, VR-12 Mental, and VR-12 Physical scores were observed between the preoperative and minimum 2-year postoperative interval (Table 2). Not only did scores improve from preoperatively to a minimum of 2 years postoperatively, but when evaluated over time, mean IKDC and Kujala scores improved from preoperatively to 1 year postoperatively and continued to improve from 1 year postoperatively to the ≥2-year follow-up (Figure 2). The MCID thresholds at ≥2 years for the IKDC, Kujala, VR-12 Mental, and VR-12 Physical scores were determined to be 9.8, 8.68, 4.3, and 5.9, respectively (Table 3). Of the determined MCID values, 77.8%, 69.0%, 56.1%, and 63.4% of patients achieved each respective threshold (Table 3).
Preoperative and Postoperative Patient-Reported Outcome Measures a

IKDC, International Knee Documentation Committee; VR-12, Veterans RAND 12-item Health Survey. Bold P values indicate statistical significance (P < .05).

Mean patient-reported outcome measures assessed preoperatively, at 1 year, and at ≥2 years of follow-up. IKDC, International Knee Documentation Committee; VR-12, Veterans RAND 12-item Health Survey.
MCID Thresholds and Achievement Rates for IKDC, Kujala, VR-12 Mental, and VR-12 Physical a

IKDC, International Knee Documentation Committee; MCID, minimal clinically important difference; VR-12, Veterans RAND 12-item Health Survey.
Subgroup Analysis
Of the 50 patients, 30 had a concomitant TTO, whereas 20 patients did not. There were no differences in pre- or postoperative IKDC, Kujala, VR-12 Mental, or VR-12 Physical scores between patients with and without a concomitant TTO (P > .05 for all).
MACI for the treatment of patients with patellofemoral bipolar lesions (n = 11) showed significant improvement in IKDC and Kujala scores, with 2 (18.2%) failures (Table 4). This subgroup consisted of 9 patients treated with MACI to both the patella and trochlea and 2 patients treated with MACI to the patella and an OCA to the trochlea. The MCID achievement in this subgroup was 88.9% for the IKDC score and 66.7% for the Kujala score (Table 5). Kaplan-Meier survival analysis with the log-rank test demonstrated no statistically significant differences in survival distributions between patients with unipolar and bipolar patellofemoral lesions (P > .05) (Figure 3).
Preoperative and Postoperative Patient-Reported Outcome Measures of Patients Treated With MACI for Bipolar Patellofemoral Lesions a
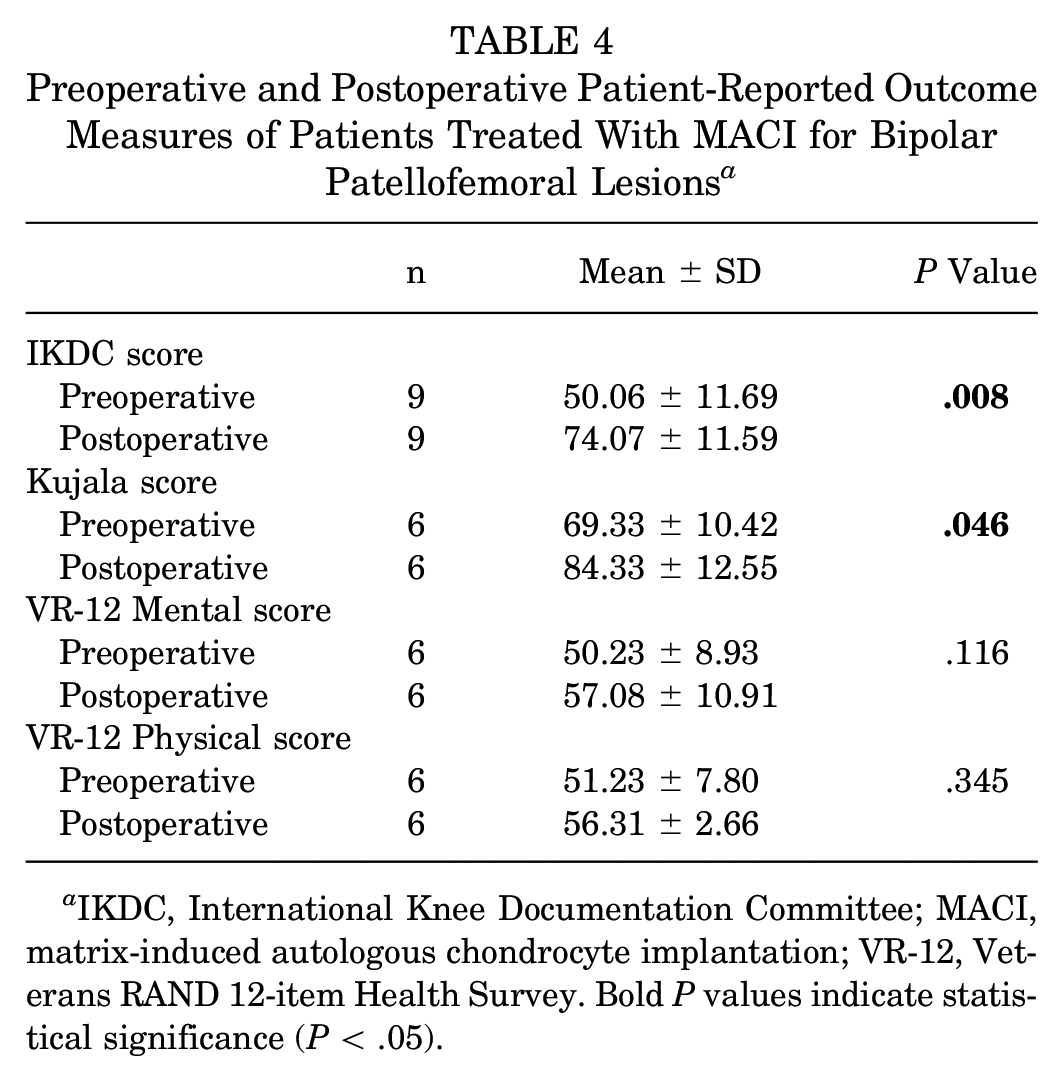
IKDC, International Knee Documentation Committee; MACI, matrix-induced autologous chondrocyte implantation; VR-12, Veterans RAND 12-item Health Survey. Bold P values indicate statistical significance (P < .05).
MCID Thresholds and Achievement Rates of IKDC and Kujala for a Subgroup of Patients Treated With MACI for Bipolar Patellofemoral Lesions a

IKDC, International Knee Documentation Committee; MACI, matrix-induced autologous chondrocyte implantation; MCID, minimal clinically important difference.

Kaplan-Meier survival analysis for unipolar versus bipolar patellofemoral lesions. The respective survival probabilities at 1, 2, and 4 years were 97.4%, 97.4%, and 88.2% for unipolar (blue line) lesions as compared with 90.9%, 90.9%, and 77.9% for bipolar (green line) lesions. The log-rank test showed no statistically significant differences in survival distributions between the groups (P = .387).
Discussion
This study demonstrated that MACI, when performed in conjunction with relevant concomitant procedures, is a successful and reliable treatment option for patellar and trochlear cartilage defects at midterm follow-up. Patients treated with MACI for patellar and/or trochlear cartilage defects had significant improvements in IKDC, Kujala, VR-12 Mental, and VR-12 Physical outcome scores and a 10.0% failure rate at a minimum 2-year follow-up. More than 60% of patients achieved the MCID in IKDC, Kujala, and VR-12 Physical scores. Survival probabilities in the Kaplan-Meier survival analysis at 1, 2, and 4 years were 98.0%, 96.0%, and 85.7%, respectively. Similarly, significant improvements were seen in IKDC and Kujala scores for bipolar patellofemoral lesions, with no statistically significant survival probability differences when compared with unipolar lesions, as assessed using Kaplan-Meier survival analyses. The MCID was achieved by more than two-thirds of bipolar patients in both IKDC (88.9%) and Kujala (66.7%). When evaluated over time, mean IKDC and Kujala scores continued to improve from 1 year to ≥2 years of follow-up, which may represent a longer tissue maturation time related to cell therapy. Overall, 24% of patients did require reoperation (chondroplasty) for graft hypertrophy or flap formation. However, no correlation was found between reoperation for chondroplasty and failure.
Patellofemoral cartilage defects are notoriously challenging to treat, given the high shear and compressive stresses experienced during knee range of motion. Although recent literature suggests that MACI is a promising treatment option for chondral lesions,1,11,23,33 original mid- and long-term outcomes of patellofemoral MACI are limited. According to Filardo et al, 11 matrix-assisted autologous chondrocyte transplantation (MACT) on a hyaluronic acid–based scaffold for patellofemoral lesions showed satisfactory clinical results in a 5-year follow-up, although not as good as those obtained in condyle lesions. When trochlear and patellar cases were compared in a subanalysis, the former showed significantly better results than the latter. The study results of Kon et al 23 followed the same path, demonstrating significant improvement in IKDC, EQ-VAS (EuroQol visual analog scale), and Tegner scores for patellofemoral lesions treated with MACT on a hyaluronic acid–based scaffold at 2-, 5-, and 10-year follow-ups. Patients with trochlear defects also achieved a higher mean IKDC score when compared with patients with patellar defects, albeit without reaching a statistically significant difference. In a study evaluating the treatment of patellofemoral lesions with MACI on a collagen 1/3 membrane, Meyerkort et al 26 demonstrated significant increases in all 5 Knee injury and Osteoarthritis Outcome Score subscales and the physical SF-36 at a 5-year follow-up. In addition, MRI analysis demonstrated good-to-excellent results for graft signal intensity, border integration, surface appearance, and structure. Patellofemoral chondral repair techniques are commonly associated with concomitant patellar realignment. The association of TTO with anteriorization of the patella is related to the reduction of patellofemoral stress forces, promoting a better environment for cartilage healing. 20 Moreover, patellar maltracking is a frequent cause of chondral lesions and should be addressed whenever diagnosed. 31
Most previous investigations of outcomes after ACI for patellar, trochlear, or bipolar patellofemoral chondral lesions have included first- or second-generation ACI.15,30,31 The first generation of ACI uses a periosteal flap to cover the chondrocyte implant. This technique has shown good functional results. However, a high rate of adverse events has been found, leading to the need for revision surgery in up to 44% of cases.18,27,28,31 The main adverse events found were delamination and dislodgment of the periosteal flap, in addition to its hypertrophy.18,20,27,28 Such events were mitigated with synthetic membranes in the second generation; however, the synthetic membrane was not seeded by chondrocytes before implantation in this case. The MACI procedure is a repair technique in which autologous chondrocytes are seeded onto a type 1/3 collagen porcine membrane before implantation into the defect. The membrane has a rough surface with a larger pore size, characterized by a looser collagen fiber arrangement that facilitates chondrocyte attachment.4,9,14,38 Previous studies have shown that chondrocytes adhere well to type 1/3 collagen membranes, forming a multilayer sheet.4,12 Furthermore, the cells maintain their chondrocytic phenotype.9,13,32 In addition to the histological advantages presented, MACI allows for a less invasive surgical procedure in areas of more difficult access because its fixation is sufficient with fibrin glue, unlike the first 2 generations that require sutures.4,28 MACI was approved by the Food and Drug Administration for clinical practice in December 2016. We present a challenging series of patients undergoing treatment for patellar, trochlear, or bipolar patellofemoral chondral lesions with third-generation ACI between August 2017 and September 2020. Third-generation ACI poses numerous advantages over first- or second-generation ACI, including a more even distribution of chondrocytes, smaller incisions required for surgery, and improved accessibility to areas that are difficult to treat. 16
There are several limitations to the present study. Given that this was a retrospective review, patients were not randomized to procedure type and selection bias may exist. In addition, the high rate of concomitant procedures in the study cohort (78%) may confound the results, as few patients had isolated patellar and/or trochlear MACI procedures. However, it is exceedingly rare to have an isolated patellar or trochlear full-thickness chondral lesion in the absence of other pathology; therefore, we believe that our study population is representative of this patient population and that our results are generalizable. In addition, the sizes of the chondral defects included in the study were variable. However, we acknowledge that it is not possible to control for the size of chondral lesions outside of the laboratory.
Conclusion
MACI, when performed alongside concomitant procedures to address other relevant pathologies, is a successful and effective treatment method for difficult-to-treat patellar, trochlear, and bipolar patellofemoral chondral defects.
Footnotes
Final revision submitted January 2, 2025; accepted February 17, 2025.
One or more of the authors has declared the following potential conflict of interest or source of funding: A.H.G. has received royalties from Moximed Inc and Organogenesis; consulting fees from Bioventus LLC, Flexion Therapeutics Inc, Joint Restoration Foundation Inc, Miach Orthopaedics Inc, Moximed Inc, Organogenesis Inc, Smith & Nephew Inc, and Vericel Corporation; hospitality payments from Aesculap Biologics LLC, Bioventus LLC, Cartiheal Inc, DePuy Synthes Sales Inc, Flexion Therapeutics Inc, Joint Restoration Foundation Inc, Linvatec Corporation, Miach Orthopaedics Inc, Organogenesis, Pacira Therapeutics, and Smith & Nephew Inc; honoraria from Fidia Pharma USA Inc, Joint Restoration Foundation Inc, and Vericel Corporation; acquisitions from Smith & Nephew Inc; research support from Aesculap Biologics LLC, Cartiheal Inc, Hyalex Orthopaedics Inc, Joint Restoration Foundation Inc, Miach Orthopaedics Inc, Moximed Inc, Organogenesis Inc, Smith & Nephew Inc, and Vericel Corporation; and is a paid presenter or speaker for Linvatec Corporation, Pacira Therapeutics, Smith & Nephew Inc, and Vericel Corporation; he holds stock or stock options in Engage Uni LLC and Smith & Nephew Inc. S.M.S. has received hospitality payments from Bioventus LLC, Cartiheal Inc, DePuy Synthes Sales Inc, Dynasplint Systems Inc, Flexion Therapeutics Inc, Joint Restoration Foundation Inc, Linvatec Corporation, Miach Orthopaedics Inc, Organogenesis Inc, Pacira Therapeutics, Smith & Nephew Inc, and Vericel Corporation; research support from Cartiheal Inc, Hyalex Orthopaedics Inc, Joint Restoration Foundation Inc, Miach Orthopaedics Inc, Moximed Inc, Organogenesis Inc, Smith & Nephew Inc, and Vericel Corporation; honoraria from Joint Restoration Foundation Inc and Vericel Corporation; consulting fees from Bioventus LLC, Flexion Therapeutics Inc, Miach Orthopaedics Inc, Moximed Inc, Smith & Nephew Inc, and Vericel Corporation; and is a paid presenter or speaker for Smith & Nephew Inc and Vericel; she holds stock or stock options in Engage Uni LLC, Moximed Inc, and Stryker. A.H.G. and S.M.S. are spouses. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was obtained from the Hospital for Special Surgery Institutional Review Board (2020-2123-AM35).
