Abstract
Background:
Bone-age determination remains a difficult process. An atlas for bone age has been created from knee-ossification patterns on magnetic resonance imaging (MRI), thereby avoiding the need for radiographs and associated costs, radiation exposure, and clinical inefficiency. Shorthand methods for bone age can be less time-consuming and require less extensive training as compared with conventional methods.
Purpose:
To create and validate a novel shorthand algorithm for bone age based on knee MRIs that could correlate with conventional hand bone age and demonstrate reliability across medical trainees.
Study Design:
Cohort study (diagnosis); Level of evidence, 2.
Methods:
Included in this study were adolescent patients who underwent both knee MRI and hand bone age radiographs within 90 days between 2009 and 2018. A stepwise algorithm for predicting bone age using knee MRI was developed separately for male and female patients, and 7 raters at varying levels of training used the algorithm to determine the bone age for each MRI. The shorthand algorithm was validated using Spearman rho (rS) to correlate each rater’s predicted MRI bone age with the recorded Greulich and Pyle (G&P) hand bone age. Interrater and intrarater reliability were also calculated using intraclass correlation coefficients (ICCs).
Results:
A total of 38 patients (44.7% female) underwent imaging at a mean age of 12.8 years (range, 9.3-15.7 years). Shorthand knee MRI bone age scores were strongly correlated with G&P hand bone age (rS = 0.83; P < .001). The shorthand algorithm was a valid predictor of G&P hand bone age regardless of level of training, as medical students (rS = 0.75), residents (rS = 0.81), and attending physicians (rS = 0.84) performed similarly. The interrater reliability of our shorthand algorithm was 0.81 (95% CI, 0.73-0.88), indicating good to excellent interobserver agreement. Respondents also demonstrated consistency, with 6 of 7 raters demonstrating excellent intrarater reliability (median ICC, 0.86 [range, 0.68-0.96]).
Conclusion:
This shorthand algorithm is a consistent, reliable, and valid way to determine skeletal maturity using knee MRI in patients aged 9 to 16 years and can be utilized across different levels of orthopaedic and radiographic expertise. This method is readily applicable in a clinical setting and may reduce the need for routine hand bone age radiographs.
Determining bone age, a metric used in a variety of medical fields, helps clinicians assess skeletal maturity and provides valuable information for proper surgical planning and management of patients. 25 Within pediatric orthopaedics, bone-age determination is particularly important for surgical planning, given the variation in patients’ skeletal maturity and remaining growth potential. This is especially critical when planning anterior cruciate ligament (ACL) reconstruction (ACLR) and limb-lengthening or guided-growth procedures that may involve crossing and potentially disturbing the physis.
Greulich and Pyle (G&P) published their first bone age atlas in 1950, with a revision published in 1959. 11 The G&P method for bone age determination is widely accepted as the gold standard for assessing skeletal maturity, preferred over other methods such as Tanner-Whitehouse because of its ease of use and short time requirement. 9,11 Although the G&P method is relatively convenient, it exposes patients to potentially unnecessary radiation and may increase clinical visits and costs. 22 Other authors have explored various methods to address some of these challenges, with varying success. Dual-energy x-ray absorptiometry has been proposed as an alternative to bone-age determination; however, it is associated with more radiation exposure than traditional radiographs. 12 Others have used ultrasonography to eliminate radiation altogether. 18,23
Despite these alternatives, ordering additional imaging for bone-age determination inevitably adds to clinical time as well as cost. Moreover, studies have shown that bone age varies depending on whether hand-wrist (G&P method) or knee radiographs are used, suggesting that bone age determinations of the knee might be better suited for addressing knee pathologies. 1,24 Pennock et al 22 created and validated a bone age atlas using knee magnetic resonance imaging (MRI) scans that provides the best bone age information yet for the treatment of knee pathologies in the skeletally immature. Their knee MRI bone age method obviates the added radiation and clinical time and potentially enfolds the cost of determining bone age into the required knee MRI, avoiding additional imaging studies.
Reducing the time cost of bone age interpretation has been the aim of multiple studies, including those creating faster digital methods, 5 those using automation, 10,29 and those using computer-assisted methods. 6,17,20 Using shorter and more reliable methods for bone age determination is not novel and was first proposed in 1989. 19 Most recently, Heyworth et al 13 created and validated a shorthand method of G&P to provide a faster bone age determination of this hand-wrist method. Shorthand bone age determination of knee MRI methods has not yet been reported. Given the multiple advantages of the knee MRI-based bone age determination created and validated by Pennock et al, 22 the present study sought to create and validate a novel shorthand version for determining bone age use knee MRI scans and demonstrate the reliability of this method across levels of training.
Methods
This retrospective descriptive study included adolescent patients who underwent a knee MRI and hand bone age radiograph at our tertiary care children’s hospital from 2009 to 2018. Imaging procedures were identified using the following Current Procedural Terminology codes: 77072 (bone age studies) and 73721 (MRI; any joint of lower extremity) or 73723 (diagnostic radiology procedure of the lower extremities). All patients were aged 9 to 16 years at the time of MRI and bone age examination, which were performed within a single 90-day period. Exclusion criteria included patients with nonknee (ie, hip or ankle) lower extremity MRI scans or endocrinopathies, including short stature, delayed or early puberty, and hypothyroidism, as well as more than 90 days between MRI and hand bone age studies. Patients younger than 9 and older than 16 years of age rarely underwent hand bone age examinations at a similar time to knee MRI because it was presumed that their physes were open and closed, respectively. Therefore, they were not eligible for inclusion in the study. For patients (n = 2) who underwent bilateral knee MRI scans on the same date, 1 side was randomly selected for inclusion in the study. This study was approved by our hospital’s institutional review board.
Chronological age, sex, and hand bone age, as calculated by our institution’s radiologists using the G&P method, were retrospectively collected from each patient’s electronic medical record. From the findings of Pennock et al, 22 a stepwise algorithm for predicted knee MRI bone age was developed separately for male (Figure 1) and female (Figure 2) patients, in collaboration with a board-certified and dual fellowship-trained pediatric musculoskeletal radiologist (J.C.N.).

Shorthand magnetic resonance imaging (MRI) knee bone age algorithm for male patients. Sagittal intermediate-weighted non–fat saturated images are provided. Arrows denote the region of interest within the MRI scan. **If a patient does not reach this stage, the bone age should be listed as 9 years to represent younger than 10.
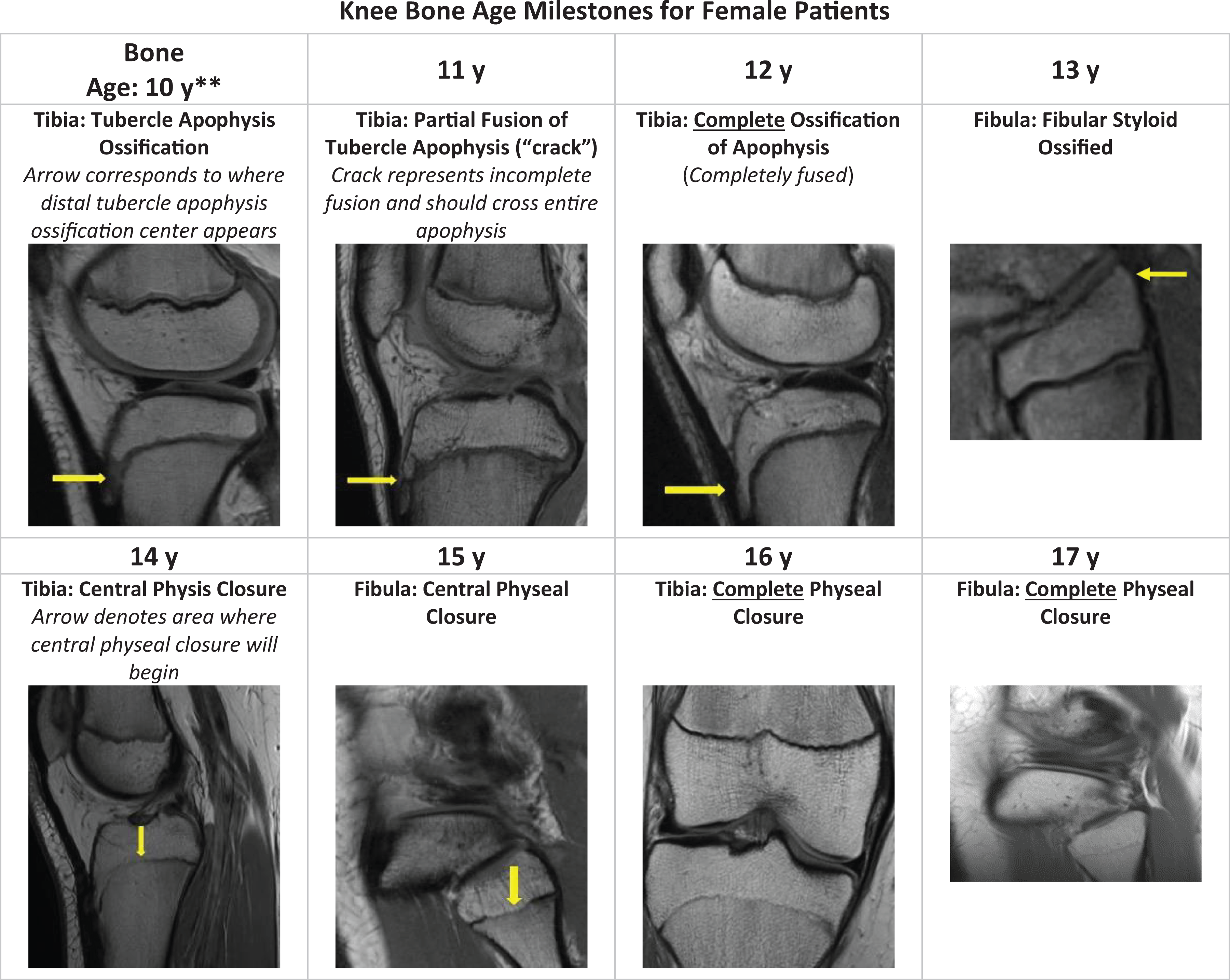
Shorthand magnetic resonance imaging (MRI) knee bone age algorithm for female patients. Sagittal and coronal intermediate-weighted non–fat saturated images are provided. Arrows denote the region of interest within the MRI scan. Note: The image shown for age 17 years is a representative image of what would be the next expected stage of physeal closure. No patients in the study achieved complete physeal closure of the fibula. **If a patient does not reach this stage, the bone age should be listed as 9 years to represent younger than 10.
Pennock et al 22 previously showed that a patient’s sex affects the timing of ossification and physeal closure. For example, in their study, complete ossification of the tibial epiphysis occurred at a median age of 14.6 years in males and 11.9 years in females. Therefore, we decided to create separate algorithms for each sex to control for pubescent variability in skeletal maturity. Our algorithms utilized a pattern of ossification and physeal patterns of the tibia, fibula, and patella on MRI, based on the median age of key radiographic milestones identified by Pennock et al, to guide raters in determining the bone age for each patient. Patients were imaged with either a 3.0-T or 1.5-T magnet. All patients were imaged prone with the knee placed in full or near-full extension, depending on the patient’s comfort preference. The clinical knee protocol consisted of multiplanar spin-echo pulse sequences that include non–fat suppressed sagittal and coronal intermediate-weighted pulse sequences. Deidentified full MRI scans were obtained and distributed to reviewers (2 medical students [B.C.M., S.M.L.], 2 orthopaedic surgery residents [C.J.D., B.M.S.], 1 pediatric musculoskeletal radiologist [J.C.N.], and 2 attending orthopaedic surgeons [J.L.C., T.J.G.]), who were given printed instructions to determine a patient’s knee bone age based on the patient’s sex and highest consecutive bone age milestone they had achieved. For example, if a patient met the bone age milestones for age 12 and 14 years, but not 13 years, they would be considered bone age 12 years. Raters were provided with each patient’s sex in order to choose the appropriate algorithm; however, they were blinded to chronological age.
Spearman rho (rS) was used to correlate the mean knee MRI bone age scores from all raters with G&P hand bone age and evaluate the accuracy of the average predicted knee MRI bone age for each level of training to G&P hand bone age. 22 These variables were treated as ordinal because of small range of ages included and the lack of fractional ages (ie, 13.5 years) that would be more suggestive with continuous variables. To validate the novel algorithm as a predictor of G&P hand bone age, Spearman rho was utilized to compare each rater’s predicted MRI bone age to the chart-recorded G&P hand bone age. Similar comparisons were made between chronological age and G&P hand bone age to demonstrate the superiority of the knee bone age algorithm in predicting hand bone age. The cohort was also subclassified and analyzed by sex to help compare the validity of each algorithm independently.
To demonstrate interrater reliability, the 2-way random, absolute agreement intraclass correlation coefficient (ICC) model was used. 16 A similar subanalysis, based on patient sex, was conducted to compare the reliability of the male and female algorithms. Additionally, MRI scans were redistributed to each rater at least 4 weeks after their initial readings to determine intrarater reliability of the algorithms. These predicted bone ages for each individual rater were compared across readings using a 2-way, mixed-effects, absolute agreement model of ICC. For all ICC comparisons, the proposed cutoffs by Cicchetti 7 were used: poor (0.00-0.39), fair (0.40-0.59), good (0.60-0.74), and excellent (0.75-1.00).
All statistical analyses were performed using SPSS Version 23 (IBM). Study data were collected and managed using Research Electronic Data Capture (REDCap) tools hosted at Children’s Hospital of Philadelphia. REDCap is a secure, web-based application designed to support data capture for research studies.
Results
In total, 38 patients (17 female; 44.7%) underwent knee MRI examinations at a mean chronological age of 12.8 ± 1.6 years (range, 9.3-15.7 years). Male patients were slightly older than female patients (13.3 ± 1.6 vs. 12.3 ± 1.5 years). The majority of patients were non-Hispanic (35; 92.1%) and Caucasian (20; 52.6%). The mean absolute time between hand bone age radiograph and knee MRI was 20.2 days (range, 0-88 days). Most frequently, knee MRI scans were obtained before hand bone age radiographs (27; 71.1%). Overall, knee MRI bone age scores were strongly correlated with G&P hand bone age (rS = 0.83; P < .001). The shorthand algorithm was also shown to be a consistent predictor of hand bone age across level of training, as medical students (rS = 0.75; P < .001), residents (rS = 0.81; P < .001), and attending physicians (rS = 0.84; P < .001) all demonstrated strong or very strong correlation. With respect to algorithm validity across raters, there was strong correlation between MRI knee bone age and G&P hand bone age for each individual rater (rS point estimate range, 0.71-0.82; P < .001) (Table 1). Our novel knee bone age MRI shorthand algorithm was shown to be superior to chronological age (rS = 0.67; P < .001) as a predictor of G&P hand bone age. When separating the MRI scans based on a patient’s sex, the male algorithm (rater rS range, 0.65-0.83) was found to be a better predictor of hand bone age than the female algorithm (rater rS range, 0.46-0.86).
Spearman Rho Between Rater Predicted Knee Bone Age Using MRI Algorithm and G&P Hand Bone Age, at Initial Rating and Minimum 4 Weeks Later a

aP < .001 for all measures. G&P, Greulich and Pyle; MRI, magnetic resonance imaging.
The mean interrater reliability for the application of our shorthand algorithm was 0.81 (95% CI, 0.73-0.88), indicating good to excellent interobserver agreement. This reliability was better for the male algorithm (ICC, 0.81; 95% CI, 0.69-0.90) than the female algorithm (ICC, 0.67; 95% CI, 0.50-0.84). Intrarater reliability was good to excellent for 6 out of 7 raters (Table 2), with a mean 2-way mixed-effects, absolute agreement ICC of 0.85. The final rater exhibited fair to good intrarater reliability.
Intrarater Reliability Between Individual Rater Scores Recorded a Minimum of 4 Weeks Apart a

a Two-way mixed-effects, absolute-agreement intraclass correlation coefficient (ICC).
Discussion
Bone age provides valuable information regarding a patient’s growth potential and must be considered whenever surgical approaches may cross or approach an open physis. Multiple methods have been proposed for determining bone age, each with its own reliability and associated ease of use. 1,4,26 In their meta-analysis of 3 different methods of bone age assessment, Serinelli et al 26 showed that despite its widespread use, G&P was the least accurate of the 3. Despite the G&P method still being widely accepted as the gold standard for bone age evaluation, MRI-based bone age determinations using the knee, 8,22 hand, wrist, 28 and clavicle 14 have been explored as alternatives.
Most recently, Pennock et al 22 created and validated a similar atlas using the ossification pattern of the knee on MRI for male and female patients aged 2 to 19 years. They also looked at 48 patients, aged 9 to 17 years, who had knee MRI and left-hand radiographs within 3 months of each other. They demonstrated both excellent interobserver reliability for the knee MRI atlas and correlation between bone age as determined by the G&P method and the knee MRI atlas. It is not clear if these comparisons were performed using a single author’s ratings or the mean from several raters.
The current study is the first to create and validate a shorthand version of a knee MRI atlas for determining bone age. The main drawback of Pennock et al’s 22 MRI knee atlas is that it fails to circumvent some of the biggest limitations of the G&P method, including the steep learning curve and time required to compare images with a lengthy atlas of standard images. Shorthand versions address these problems and have been employed by Heyworth et al, 13 who created a shorthand version of the G&P method using the pattern of physeal closure in the digits. Our study aimed to combine the central concepts of these studies and create a shorthand version of the MRI knee atlas based on the progression of ossification and physeal closure of the knee established in Pennock et al’s atlas.
Our algorithm demonstrated excellent reliability over time and between raters as well as superior predictability of G&P bone age compared with chronological age. Additionally, level of training did not significantly affect rater performance, suggesting that this method is easy to learn regardless of clinical expertise. Although some of the raters included in this study helped to create and were therefore more familiar with the algorithm, 4 raters (2 residents, 2 attending surgeons) did not have any knowledge of the algorithm before scoring the MRI scans, indicating that this shorthand method may be applied without significant training.
In addition to other benefits of shorthand methods, such as clinical efficiency and reducing the need for extensive training or use of the atlas itself, our novel shorthand MRI knee bone age algorithm may prevent the need for and enfold the costs ($123) 27 and radiation associated with hand bone age radiographs into the inevitable knee MRI these patients require. The idea of combining common methods of imaging to obtain information more efficiently, with less radiation exposure, is not entirely new. Some authors have proposed that patients with scoliosis should receive a single posteroanterior spine radiograph that includes their hands placed near the skull so that bone age and deformity can be quantified in a single image. 15
It is difficult to assess how rater performance changes over time. We found that the correlation between knee MRI bone age and G&P hand bone age improved slightly with subsequent rounds of ratings for some of the raters. Utilizing the algorithm regularly in a clinical setting may lead to a quicker and, perhaps, more drastic improvement in scoring, compared with the improvements observed in our study with at least 4 weeks between ratings of the cohort. Future studies should aim to evaluate the impact of consistent usage of the algorithms on performance and help quantify how many knee MRI bone age ratings are necessary for mastery.
In light of the novelty of MRI-based atlases for quantifying bone age, there are still many unknowns. First, it is not evident which physis of the knee is the most accurate predictor of bone age. Our algorithm focused on the patella, tibia, and fibula to limit the regions in which raters had to evaluate physeal closure and maximize efficiency of the shorthand method. The distal femoral physis closes across a similar age range to the fibula and should be evaluated for potentially superior accuracy in future studies. The G&P atlas was created decades ago and has been shown to have varying reliability across different ethnicities. 2,21 It is unknown how ethnicity or other demographic factors affect the knee MRI findings used to build our shorthand algorithms. Additionally, utilization of our proposed algorithms in clinical practice could be subject to other biases, such as provider knowledge of a patient’s chronological age. It has been shown that prior knowledge of chronologic age affects G&P determinations. 3 We blinded our raters to chronological age, and superior performance of the shorthand knee MRI bone age algorithm to age was exhibited. Therefore, in cases where the 2 ages are discordant, we believe that priority should be given to the predicted knee MRI bone age.
Further, this study has several important limitations to consider. First, the single-center, retrospective, descriptive nature of the study may preclude its widespread generalizability. Large-scale multicenter studies would help establish its validity across a variety of clinical environments and determine what demographic factors may impact its accuracy. Furthermore, subanalysis of each algorithm by sex demonstrated that the algorithm created for male patients was more reliable and consistent with G&P hand bone age. This could be explained, in part, by the smaller number of female patients included, as well as the limited number of female patients at the extremes of ages (ie, 9-10 years old, 15-16 years old) with hand bone age radiographs found within our picture archiving and communication system. It is likely that these patients were presumed to have a completely open or closed physis on the basis of chronological age alone and therefore did not require bone-age evaluation. Although it did not occur in this study, evaluating the ossification and physeal closure pattern on knee MRI could be limited by the specific knee pathology that is present, such as bone bruises associated with ACL tears. This is an inherent limitation of any shorthand method that relies on imaging of the injured area.
Finally, this study cannot fully quantify the clinical benefits associated with implementation of the knee bone age shorthand algorithms. It is expected that fewer hand radiographs would result in lower costs, less radiation exposure, and greater clinical efficiency; however, the exact amount of time and money saved has yet to be evaluated. Our goals include investigating these matters and determining how our algorithm can be used to effectively guide surgical planning. Specifically, potential future directions include studying the costs and time saved by the knee MRI shorthand method versus G&P hand bone age method and evaluating the effectiveness of knee MRI bone age calculations in determining appropriate ACL reconstruction techniques in adolescents.
Conclusion
This novel shorthand algorithm was shown to be a consistent, reliable, and valid way to determine skeletal maturity using the ossification pattern on knee MRI in patients aged 9 to 16 years across raters of different levels of orthopaedic and radiographic expertise. We believe that this method is readily applicable in a clinical setting and may reduce the need for routine hand bone age radiographs.
Footnotes
Final revision submitted January 7, 2021; accepted February 23, 2021.
One or more of the authors has declared the following potential conflict of interest or source of funding: T.J.G. has received research support from Vericel, Arthrex, and Allosource and education payments from Arthrex; and is a paid associate editor for The American Journal of Sports Medicine. J.L.C. has received research support from Allosource, Anika Therapeutics, Ossur, and Vericel; and consulting fees, honoraria payments, and nonconsulting fees from Vericel. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval for this study was waived by Children’s Hospital of Philadelphia Research Institute (No. 18-015693).
