Abstract
Background:
Malalignment of the lower extremity can lead to early functional impairment and degenerative changes. Distal femoral osteotomy (DFO) can be performed with arthroscopic surgery to correct lower extremity malalignment while addressing intra-articular abnormalities or to help patients with knee osteoarthritis (OA) changes due to alignment deformities.
Purpose:
To examine survivorship after DFO and identify the predictors for failure.
Study Design:
Case series; Level of evidence, 4.
Methods:
Data from the California Office of Statewide Health Planning and Development, a statewide discharge database, were utilized to identify patients between the ages of 18 and 40 years who underwent DFO from 2000 to 2014. Patients with a history of lower extremity trauma, infectious arthritis, rheumatological disease, skeletal dysplasia, congenital deformities, malignancy, or concurrent arthroplasty were excluded. Failure was defined as conversion to total or unicompartmental knee arthroplasty, and the identified cohort was stratified based on whether they went on to fail. Age, sex, race, diagnoses, concurrent procedures, and comorbidities were recorded for each admission. Statistically significant differences between patients who required arthroplasty and those who did not were identified using the Student t test for continuous variables and a chi-square test for categorical variables. Kaplan-Meier survivorship curves were constructed to estimate 5- and 10-year survival rates. A Cox proportional hazards model was used to analyze the risk for conversion to arthroplasty.
Results:
A total of 420 procedures were included for analysis. Overall, 53 knees were converted to arthroplasty. The mean follow-up time was 4.8 years (range, 0.0-14.7 years). The 5-year survivorship was 90.2% (range, 85.7%-93.4%), and the 10-year survivorship was 73.2% (range, 64.7%-79.9%). The mean time to failure was 5.9 years (range, 0.4-13.9 years). Survivorship significantly decreased with increasing age (P = .004). Hypertension and a primary diagnosis of osteoarthrosis were significant risk factors for conversion to arthroplasty (odds ratio [OR], 3.12 [95% CI, 1.38-7.03]; P = .006, and OR, 2.42 [95% CI, 1.02-5.77]; P = .045, respectively), along with a primary diagnosis of traumatic arthropathy (OR, 10.19 [95% CI, 1.71-60.65]; P = .01) and a comorbid diagnosis of asthma (OR, 2.88 [95% CI, 1.23-6.78]; P = .02). Patients with Medicaid were less likely (OR, 0.11 [95% CI, 0.01-0.88]; P = .04) to undergo arthroplasty compared with patients with private insurance, while patients with workers’ compensation were 3.1 times more likely (OR, 3.08 [95% CI, 1.21-7.82]; P = .02).
Conclusion:
Older age was an independent risk factor for conversion to arthroplasty after DFO in patients ≥18 years but ≤60 years. Hypertension, asthma, and a diagnosis of osteoarthrosis or traumatic arthropathy at the time of surgery were predictors associated with failure, reinforcing the need for careful patient selection. The high survivorship rate of DFO in this analysis supports this procedure as a reasonable alternative to arthroplasty in younger patients with valgus deformities about the knee and symptomatic unicompartmental OA.
Malalignment of the lower extremity can lead to pain, functional impairment, and early degenerative changes. Symptomatic genu varum is usually caused by excessive varus of the tibia and may be treated with high tibial osteotomy, while excessive valgus malalignment about the knee is often caused by excessive valgus of the femur and is less frequently encountered. Genu valgum, whether resulting from a hypoplastic lateral femoral condyle or acquired from previous trauma or previous intra-articular procedures, may lead to early osteoarthritis (OA) of the lateral compartment. 3
Although total knee arthroplasty or lateral unicompartmental knee arthroplasty may be employed to treat unicompartmental OA associated with a valgus deformity, their utility in younger patients with OA is limited by implant longevity and the eventual need for revision surgery. 14 Therefore, in young active patients with lateral compartment OA and valgus malalignment, correction is sometimes attempted with distal femoral osteotomy (DFO) to offload the diseased compartment. 3,17 Realignment procedures of the lower extremity, including DFO, are useful for the treatment of habitual patellar dislocations with good outcomes. 12 However, Eberbach et al, 7 in a study of 420 patients, found that valgus malalignment was more commonly caused by a tibial deformity compared with a femoral deformity. In addition, the authors suggested that varus osteotomy to address OA of the lateral compartment must be performed at the tibial site or as a double-level osteotomy procedure (femoral and tibial). Nevertheless, DFO has been shown to achieve satisfactory pain relief and functional improvement in patients with unicompartmental OA, with a complication rate comparable with that of high tibial osteotomy. 1 –3,5,8,10,11
Previous studies investigating DFO have been limited by small sample sizes, constraining the generalizability of the results, especially pertaining to survivorship estimates. In their retrospective review, Backstein et al 1 identified only 38 patients (40 knees), despite analyzing records from a 30-year time period. A systematic review 3 included 14 studies representing 236 patients (248 knees); however, 2 of the studies that provided the largest patient contributions originated from the same institution and therefore likely represented much of the same patient cohort. 1,14 Recently, Voleti et al 15 reported a 100% (13/13) return-to-sport rate in a group of athletic patients who underwent DFO. The mean age of that patient group was 24 years (range, 15-35 years), and the mean time to return to sport was 11 months (range, 9-13 months). 15 The survivorship rate after DFO in a recent systematic review ranged from 64% to 87% at 10 years, with similar rates between open and closed DFO. 9
The aim of this study was to conduct a population-based investigation to examine survivorship after DFO and to identify the predictors for failure, defined as conversion to arthroplasty. We hypothesized that DFO would display a reasonable level of survivorship in patients aged between 18 and 60 years.
Methods
Data from the California Office of Statewide Health Planning and Development (OSHPD), a mandatory statewide discharge database, were utilized for this study. This database contains information from all public and private inpatient hospitals, ambulatory surgery centers, and emergency departments in the state of California, as well as demographic data for each patient and up to 25 medical diagnoses and total hospital charges with each admission. Diagnosis and procedure codes are listed as International Classification of Diseases, Ninth Revision (ICD-9) and Current Procedural Terminology (CPT) billing codes. Patients are tagged with a unique record linkage number that remains consistent throughout all admissions within the state of California, allowing patients to be tracked longitudinally regardless of where they receive postsurgical follow-up or future medical care.
Patients who underwent DFO from 2000 to 2014 were collected by identifying all admissions containing ICD-9 procedure codes 77.25 (osteotomy, femur) and 77.35 (wedge osteotomy, femur) as well as CPT codes 27448 (osteotomy, femur, without fixation) and 27450 (osteotomy, femur, with fixation). Those with a history of lower extremity trauma, infectious arthritis, rheumatological disease, skeletal dysplasia, congenital deformities, malignancy, or concurrent arthroplasty were excluded. A full list of inclusion and exclusion codes is provided in the Appendix. Patients younger than 18 years and older than 60 years were also excluded (Table 1). Concurrent diagnosis and procedure codes were reviewed for all patients with 2 qualifying osteotomy procedures to determine whether the second DFO procedure should be categorized as a revision or contralateral procedure.
Inclusion and Exclusion Criteria a

a CPT, Current Procedural Terminology; DFO, distal femoral osteotomy; ICD-9, International Classification of Diseases, Ninth Revision; OSHPD, Office of Statewide Health Planning and Development.
From 2000 to 2014, there were 6911 procedures identified based on coding alone. However, only 420 procedures remained after exclusions (Figure 1). Over 6000 procedures were excluded by age alone. Of the procedures included for analysis, 17 patients underwent bilateral DFO.
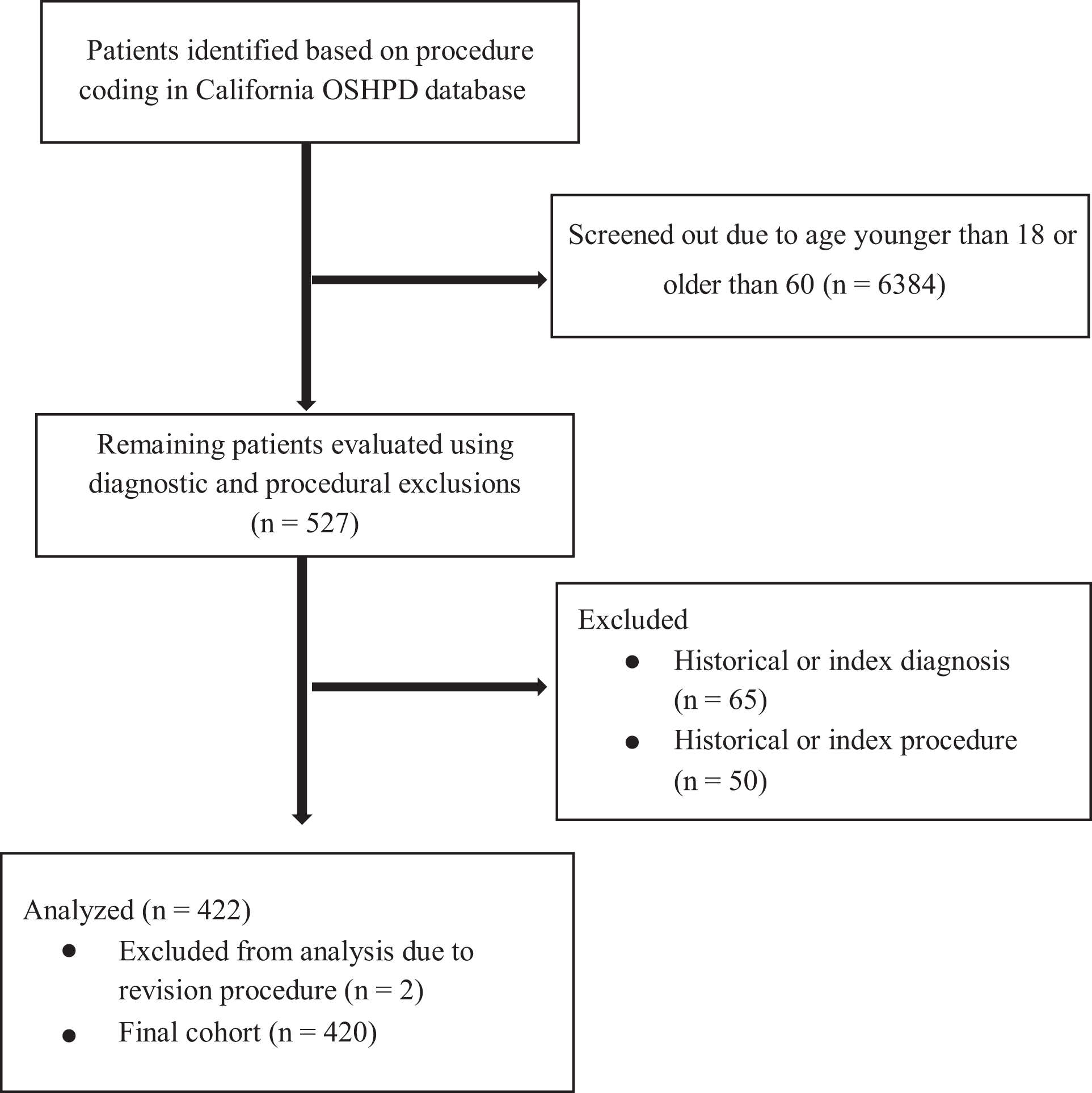
Patient identification and screening flow diagram. OSHPD, Office of Statewide Health Planning and Development.
Failure was defined as conversion to total or unicompartmental knee arthroplasty, and the identified cohort was stratified based on whether they went on to fail. Because of inherent limitations of the OSHPD database, we were unable to record the laterality of the procedure. Age, sex, race, diagnoses (OA, acquired genu valgum, other acquired deformity, derangement of internal knee structures, osteochondral defects, traumatic arthritis, and other arthropathy), concurrent procedures (arthroscopic surgery, osteochondral grafting, synovectomy, and meniscectomy), and comorbidities (asthma, chronic kidney disease, congestive heart failure, depression, diabetes mellitus, hypertension, obesity, and peripheral vascular disease) were recorded for each admission. Subsequent readmissions to an inpatient hospital, ambulatory surgery center, or emergency department in California after the index procedure were identified and sequenced using the record linkage number.
Statistically significant differences between patients who required arthroplasty and those who did not were identified using the Student t test for continuous variables and a chi-square test for categorical variables. Kaplan-Meier survivorship curves were constructed to estimate 5- and 10-year survival rates. If a patient underwent multiple revision procedures, only the time to the index arthroplasty procedure was included for analysis. Patients who underwent bilateral osteotomy were considered as 2 separate patients from the time of their contralateral DFO, to maintain the single failure per DFO procedure model. To compare survivorship for specified groups, a log-rank test of equality was employed. A Cox proportional hazards model was used to analyze the risk for conversion to arthroplasty. The results of this model were expressed as hazard ratios (HRs) with 95% CIs and P values. With simple Cox regression (unadjusted), we analyzed the following factors: age, sex, race, primary health insurance, diagnoses, comorbidities, and concurrent procedures. A multiple Cox regression model (adjusted) was constructed using all of these variables. Subsequent analysis using both simple and multiple Cox regression models was performed to evaluate the effect of age group (18-29, 30-39, 40-49, and 50-60 years) as well as the number of concurrent comorbidities. Statistical significance was set at P < .05. All statistical analyses were performed using Stata/IC 16.1 software (StataCorp).
Results
From 2000 to 2014, a total of 420 procedures remained after exclusions and were included for analysis. Overall, 53 knees were converted to arthroplasty. The mean follow-up time was 4.8 years (range, 0.0-14.7 years).
Patient Characteristics
Patients who underwent an arthroplasty procedure after their DFO tended to be older than patients who did not (mean age, 43.6 ± 8.9 vs 36.8 ± 11.1 years, respectively; P < .001). Patients who converted to arthroplasty also had a higher incidence of hypertension (32.1% vs 10.9%, respectively; P < .001) and a higher number of comorbidities (47.2% vs 27.5%, with at least 1 comorbidity, respectively; P = .021). These patients who converted to arthroplasty were also more likely to have a diagnosis of osteoarthrosis at the time of their initial DFO (81.1% vs 53.7%, respectively; P < .001). Patient characteristics are listed in detail in Tables 2 and 3.
Patient Demographics a

a Data are shown as n (%) unless otherwise indicated. Bolded P values indicate statistically significant differences between the arthroplasty and nonarthoplasty groups (P < .05).
Comorbidities, Diagnoses, and Concurrent Procedures a

a Data are shown as n (%). Bolded P values indicate statistically significant differences between the arthroplasty and nonarthoplasty groups (P < .05).
Risk of Conversion to Arthroplasty
Crude HR analysis demonstrated that patients were 3% more likely to undergo arthroplasty for each additional year of age (hazard ratio [HR], 1.03 [95% CI, 0.99-1.06]; P = .05). Patients indicated for DFO with a primary diagnosis of osteoarthrosis were 2.4 times more likely to convert to arthroplasty (OR, 2.40 [95% CI, 1.16-4.95]; P = .02). Hypertensive patients were 2.5 times as likely to require arthroplasty (OR, 2.51 [95% CI, 1.32-4.74]; P = .005).
When utilizing multiple Cox regression to calculate the adjusted risk of conversion to arthroplasty, hypertension and a primary diagnosis of osteoarthrosis remained significant risk factors (OR, 3.12 [95% CI, 1.38-7.03]; P = .006, and OR, 2.42 [95% CI, 1.02-5.77]; P = .045, respectively). Age was no longer a significant risk factor (OR, 1.01 [95% CI, 0.98-1.04]; P = .59). Furthermore, patients with Medicaid were less likely (OR, 0.11 [95% CI, 0.01-0.88]; P = .04) to undergo arthroplasty compared with patients with private insurance, while patients with workers’ compensation were 3.1 times more likely (OR, 3.08 [95% CI, 1.21-7.82]; P = .02). Additional significant risk factors for conversion to arthroplasty according to multiple Cox analysis were a primary diagnosis of traumatic arthropathy (OR, 10.19 [95% CI, 1.71-60.65]; P = .01) and a comorbid diagnosis of asthma (OR, 2.88 [95% CI, 1.23-6.78]; P = .02). A full list of HRs can be found in Table 4.
On subanalysis of age groups (18-29, 30-39, 40-49, and 50-60 years), there was a significantly increased risk in each group compared with the 18 to 29–year age group (Table 5). When analyzing for the risk of multiple comorbidities, multiple Cox regression demonstrated that patients with 3 comorbidities were 6.6 times as likely to convert to arthroplasty compared with those without comorbidities (OR, 6.62 [95% CI, 1.21-36.37]; P = .03) (Table 5).
Simple and Multiple Cox Regression a

a Bolded P values indicate statistical significance. HR, hazard ratio.
b Crude (unadjusted).
c Adjusted (all variables mentioned above entered into Cox analysis).
d Analyzed as separate independent variables given the possibility of concomitant presence in each patient.
e Chronic kidney disease and congestive heart failure omitted because of insufficient prevalence.
Subgroup Analysis Using Simple and Multiple Cox Regression a

a Bolded P values indicate statistical significance. HR, hazard ratio.
b Crude (unadjusted).
c Adjusted (computed using multiple Cox model presented in Table 4).
Survivorship
The 5-year survivorship was 90.2% (range, 85.7%-93.4%), and the 10-year survivorship rate was 73.2% (range, 64.7%-79.9%) (Figure 2). The mean time to failure (ie, conversion to arthroplasty) was 5.9 years (range, 0.4-13.9 years). Patients with a diagnosis of osteoarthrosis at the time of their index procedure had a 5-year survivorship of 88.49% (range, 73.82%-93.18%) compared with 93.50% (range, 85.65%-97.12%) for patients without and a 10-year survivorship of 67.32% (range, 56.52%-76.00%) compared with 86.37% (range, 73.82%-93.18%), respectively (P = .012) (Figure 3). Survivorship also significantly decreased with increasing age (P = .004) (Figure 4).

Kaplan-Meier survival estimate for survivorship to knee arthroplasty after distal femoral osteotomy.

Kaplan-Meier survival estimate for survivorship to knee arthroplasty after distal femoral osteotomy based on diagnosis of osteoarthritis.
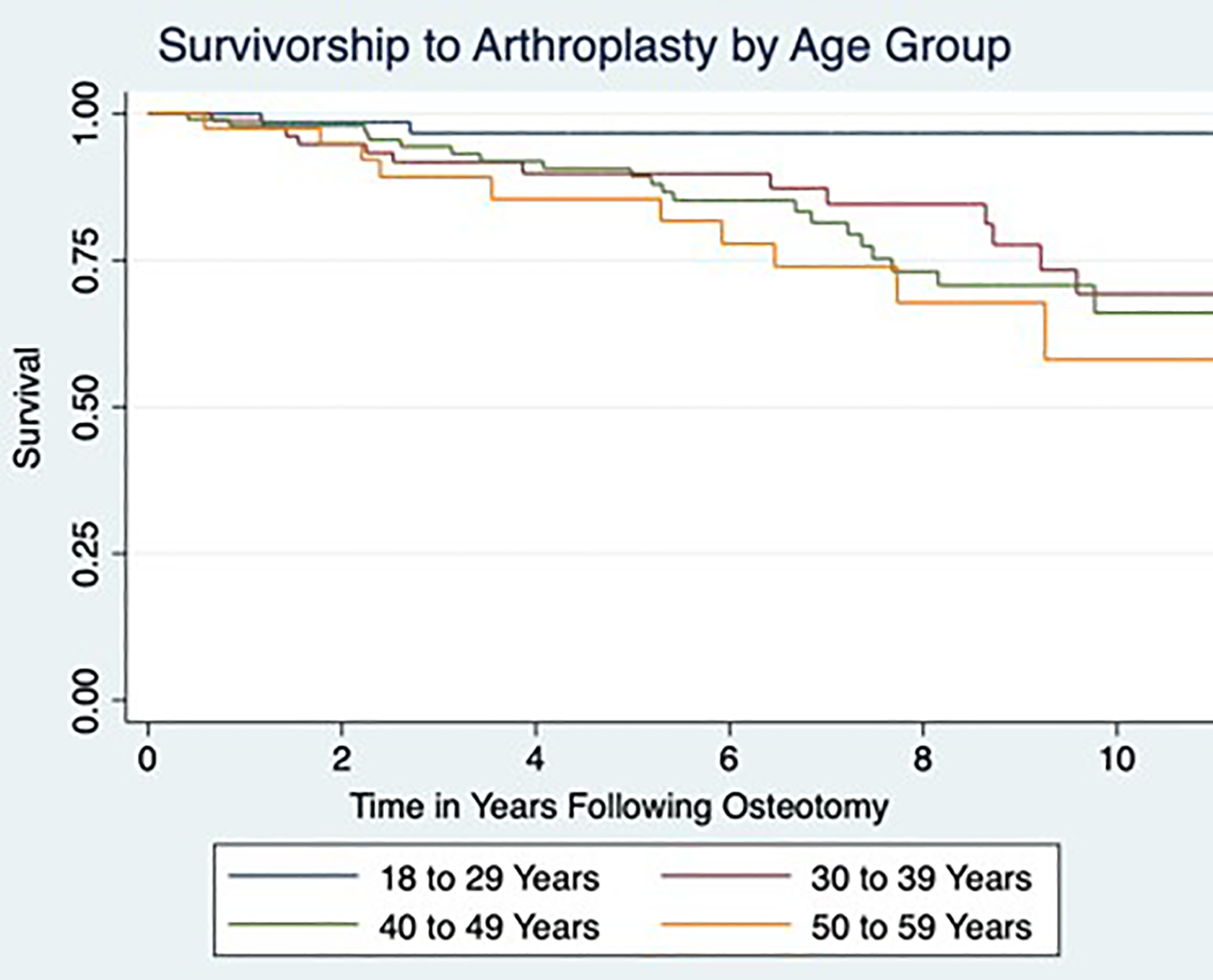
Kaplan-Meier survival estimate for survivorship to knee arthroplasty after distal femoral osteotomy by age group.
Discussion
According to this OSHPD analysis, the 5- and 10-year survivorship of DFO in patients between 18 and 60 years were 90.2% and 73.2%, respectively. Risk factors for conversion to arthroplasty after DFO were older age, hypertension, a primary diagnosis of osteoarthrosis or traumatic arthropathy, and a comorbid diagnosis of asthma. Patients with Medicaid were less likely to undergo arthroplasty compared with patients with private insurance, while patients with workers’ compensation were 3.1 times more likely.
The survivorship rates in the current study are consistent with reports in the existing literature. Ekeland et al, 8 in a study including 24 patients with a mean age of 48 years, reported the DFO survival rate as 88% at 5 years and 74% at 10 years. Similarly, in the study of Sternheim et al, 14 the survivorship of DFO at 10, 15, and 20 years was 90%, 79%, and 21.5%, respectively. In their systematic review, Chahla et al 3 included the results of 14 studies investigating DFO for the treatment of genu valgum with lateral OA. Overall, 5 of the studies used a lateral opening wedge technique, and 9 studies utilized a medial closing wedge technique, with a total cohort of 307 patients (323 knees). They reported a mean survival rate of 80% (range, 64%-90%) at 10 years. 3 We did not report survivorship at 20 years, which seems to be significantly lower than the survival rate at 5, 10, and 15 years in previous studies. 14,16 The last point should be taken into consideration during patient counseling regarding the longevity of DFO, especially beyond 15 years from the time it was performed.
As mentioned previously, DFO is not only indicated in patients with established OA in the lateral compartment, but it is also useful as an adjunct procedure for the correction of realignment in knee preservation cases of cartilage or meniscal transplantation. Drexler et al 6 reported a survivorship of 88.9% at 10 years, 71.4% at 15 years, and 23.8% at 20 years in a group of patients who underwent DFO combined with osteochondral allograft for failed lateral tibial plateau fractures. A significant drop in the survival rate at 20 years was observed, which corroborates the findings of the studies mentioned previously. Cameron et al 2 reported their outcomes of DFO by dividing the patient cohort into a joint preservation group (cartilage or meniscal defect with a valgus deformity) and an OA group (lateral compartment OA with a valgus deformity). The authors reported a 5-year survivorship of DFO of 74% in the OA group and 92% in the joint preservation group. To our knowledge, no other research group has reported comparative outcomes based on the preoperative diagnosis or procedures performed in patients who underwent DFO. Our results showed that a similar percentage of patients in the arthroplasty and nonarthroplasty groups had osteochondral allograft transplantation performed at the time of DFO. However, we calculated the survival rates in patients who underwent DFO concurrently with other procedures, and this constitutes a limitation in our analysis.
An additional 2 studies have investigated the outcomes of DFO in young and active patients by reporting the rate of return to physical activity postoperatively. In the study of de Carvalho et al, 4 there was a significant improvement in the Lysholm score (mean postoperative score was 77.1 compared with 53.1 preoperatively) in 26 patients who underwent DFO for symptomatic OA of the lateral compartment of the knee and who were physically active. In that group, the rate of return to physical activity was 57.7% at a mean follow-up time of 48 months. More recently, Voleti et al 15 reported a 100% rate of return to sport at a mean time of 11 months in 13 patients who underwent DFO for unloading valgus knee malalignment. Of those patients, 9 (69.2%) had concomitant chondral, meniscal, or ligamentous procedures performed on the ipsilateral knee joint. Our study did not evaluate the level of physical activity of the included patients or the rate of return to physical activity, and we were unable to compare our results. More research is necessary to determine whether DFO should be routinely performed in athletes with lower extremity malalignment who wish to return to physical activity, but these past 2 studies showed promising outcomes. 4,15
Our analysis revealed age as an independent predictor for failure. On subanalysis of age groups (18-29, 30-39, 40-49, and 50-60 years), there was a significantly increased risk in each group compared with the 18 to 29–year age group. Because of small cohort numbers, previous studies have been unable to draw associations between poor outcomes and age. 6 In the 14 studies they reviewed, Chahla et al 3 found no stratification of survival rates based on age. While decreased survivorship in the presence of OA is well documented in the high tibial osteotomy literature, such reports are fairly scarce with regard to DFO procedures. As mentioned above, Cameron et al 2 reported a 5-year survival rate of 74% in their OA group compared with 92% in the group without radiographic signs of OA. Similar to our findings, the mean age of the OA group was significantly higher than in the group without OA (41 vs 26 years, respectively).
Our results also showed that hypertension and a primary diagnosis of osteoarthrosis were significant risk factors for conversion to arthroplasty (OR, 3.12 [95% CI, 1.38-7.03]; P = .006, and OR, 2.42 [95% CI, 1.02-5.77]; P = .045, respectively). Additional significant risk factors for conversion to arthroplasty included a primary diagnosis of traumatic arthropathy (OR, 10.19 [95% CI, 1.71-60.65]; P = .01) and a comorbid diagnosis of asthma (OR, 2.88 [95% CI, 1.23-6.78]; P = .02). Unfortunately, we did not record body mass index, and therefore, we could not evaluate whether patients with metabolic syndrome are at a higher risk of DFO failure. 13 In addition, we are not aware of whether these patients were appropriately treated for hypertension. Based on the above and given that no previous studies have conducted a similar investigation, we were unable to make any conclusions regarding the impact of hypertension on the survival rate of DFO.
Our study has several strengths that are worth noting. The utilization of a statewide database allowed us to assess a larger cohort than previous studies. The larger numbers identified in the present study provided increased power to identify the risk factors for failure. Furthermore, patients identified in the current study came from several different hospitals and various practice settings throughout the state of California, making our findings more generalizable than previous single-center studies. Additionally, the unique record linkage numbers used in the OSHPD database allowed for long-term follow-up while limiting attritional loss of patient data. To our knowledge, this is the first epidemiological study investigating the survivorship of DFO using a population cohort.
There are several limitations to this study. Administrative databases such as the OSHPD do not allow for the assessment of outcome scores, severity of the deformity, grading of OA, surgical technique, postoperative protocols, or patient activity level, which limits the level of detail provided in our analysis. Despite this limitation, we were able to estimate procedure survivorship and demonstrate an age-dependent risk of failure. With any administrative data that rely on ICD-9 and CPT coding, there is a risk of coding errors. This risk is inherent with any study that relies on these types of databases, together with the possible loss to follow-up that might have resulted in overestimation of the survivorship rates. In contrast, the lack of laterality data in the database constitutes a major limitation of this study, which might have affected the accuracy of the reported survival rates. Patient body mass index was not recorded in our analysis. Because of this, not only were we unable to provide any information on whether obesity was a risk factor for DFO failure, but we also could not examine the impact of metabolic syndrome on DFO outcomes. We did not have any information on the treatment that the included patients received for hypertension, and although hypertension was found to increase the risk for knee replacement after DFO, we were unable to make any valid conclusion. In addition, we did not have any information on the degree of knee OA of the included patients, and we were unable to identify the primary indication for DFO in our study population. Regarding the endpoint used to define failure (knee arthroplasty), we could not identify whether this was unicompartmental knee arthroplasty or total joint replacement, which would be clinically useful.
Conclusion
Older age was an independent risk factor for conversion to arthroplasty after DFO in patients between 18 and 60 years. Hypertension, asthma, and a diagnosis of osteoarthrosis or traumatic arthropathy at the time of surgery were predictors associated with failure, reinforcing the need for careful patient selection. The high survivorship rate of DFO supports this procedure as a reasonable alternative to arthroplasty in younger patients with a valgus deformity about the knee and symptomatic unicompartmental OA.
Footnotes
Acknowledgment
The authors acknowledge the Cappo Family Research Fund.
Final revision submitted April 10, 2020; accepted April 14, 2020.
One or more of the authors has declared the following potential conflict of interest or source of funding: N.H. has received educational support from Smith & Nephew. W.C.P. has received hospitality payments from Zimmer Biomet. J.R.H. has received educational support from Elite Orthopaedics. C.T.V. has received consulting fees and honoraria from Osiris Therapeutics. G.F.H. has received educational support from Arthrex and Micromed, speaking fees from Arthrex, and honoraria from Fidia Pharma. A.E.W. has received educational support and speaking fees from Arthrex and hospitality payments from Stryker. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto.
Ethical approval was not sought for the present study.
