Abstract
Background
HIV index case testing is a successful early detection and prevention technique that includes providing HIV testing to the biological children and sexual partners of individuals living with HIV. There is inconsistent data about acceptance of HIV index testing in Ethiopia.
Methods
A systematic review and meta-analysis were conducted under the guidelines of PRISMA 2020. We searched Google Scholar, Hinari, Science Direct, PubMed, and the Cochrane Library. A total of 3952 participants from 11 cross-sectional studies were included. A random-effects model was used in STATA 17 for analysis. Subgroup, sensitivity, and meta-regression analyses were used to investigate heterogeneity.
Results
Pooled acceptance rate was 68.9% (95% CI: 57.6-80.2). Acceptance was higher among those who lived in an urban area, disclosed their HIV status, and had been on ART for more than a year.
Conclusion
Nearly 70% of ART patients in Ethiopia agree to undergo index testing
Introduction
HIV remains a serious worldwide health concern, particularly in sub-Saharan Africa, which has the greatest rate of new infections and fatalities. 1 In Ethiopia, the 2023 HIV Estimates and Projections report, there were 603,537 HIV-positive individuals (5.5% of whom were children) and 8257 new infections (18% of whom were children) and the prevalence of HIV in persons 15 years of age and older is about 0.87%, there are 610,000 HIV-positive individuals in the nation. Of these, approximately 510,000 people were receiving antiretroviral treatment (ART). 2
HIV index case testing, also known as partner notification or contact tracing, is an important method for detecting undiagnosed HIV infections. 3 Index testing enables early diagnosis and timely antiretroviral medication (ART) starting, improving individual health outcomes and reducing HIV transmission. 4 Since negative perceptions and judgments associated with being HIV infected continue to persist, stigma remains one of the biggest social challenges. 5 Although index testing and other HIV testing programs have been expanded in Ethiopia, HIV is still a major public health concern with regionally varied acceptance rates of HIV. 6
Index testing has been demonstrated to improve connection to care and increase case detection; nevertheless, its acceptance among ART patients in Ethiopia is uneven and occasionally below ideal. Numerous studies document significant geographical variations in acceptance rates, which can range from as low as 39% in some places to over 85% in others. 7 Social stigma, fear of disclosure, ignorance, and inequities in the healthcare system are frequently connected to this heterogeneity. 3
Efforts to optimize index testing in Ethiopia run the danger of being ineffective or misguided if these components and the overall acceptance rate are not thoroughly understood. Furthermore, policymakers’ capacity to create focused actions using nationally pooled data is constrained by the fragmented nature of the available research.
Therefore, this meta-analysis and systematic review aims to fill this gap by determining national pooled acceptance rate of HIV index testing among people living with HIV individuals and identify factors associated with it. In addition to identifying feasible objectives for intervention, such as improving counselling for patients new to ART, addressing urban-rural disparities, and strengthening disclosure support, this study will give a clear baseline for programs performance by synthesizing the available evidence. The results are meant to help Ethiopian HIV prevention and treatment programs be improved for more equal and successful results.
Methods
Protocol Registration
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 standards were followed in the conduct of this systematic review and meta-analysis. The protocol was developed beforehand and registered with the International Prospective Register of Systematic Reviews (PROSPERO) with a reference number CRD420251176875. The objective was to compile information on Ethiopian ART patients’ acceptance rates of index case HIV testing and related variables.
Study Design
Systematic review and meta-analysis
Study Setting
The included primary studies were conducted among HIV index patients in Ethiopia.
To ensure transparency and reproducibility, this assessment was carried out in compliance with the PRISMA 2020 guidelines (SF1). Using the CoCoPop (Condition, Context, Population) mnemonic to direct inclusion, a thorough search method was created: Condition – HIV index testing; Context
Two reviewers (ASA and TKD) independently performed systematic electronic searches across four main databases: PubMed, Cochrane Library, Hinari, and Science Direct. Both Medical Subject Headings (MeSH) and free-text phrases were used, together with Boolean operators (“AND,” “OR”) and truncation symbols when necessary, to optimize sensitivity. “Acceptance,” “uptake,” “HIV index testing,” “index testing,” “associated factors,” “predictors,” and “Ethiopia” were among the main search terms. Each database's indexing system was used to customize the search strings.
To find more relevant papers, the reference lists of every study that was included were manually checked in addition to database searches. To reduce publication bias, Google Scholar and manual searches of institutional repositories were used to investigate grey literature. Every study with human subjects that was released prior to the last search date was taken into account.
The Newcastle-Ottawa Scale (NOS) (SF1) for cross-sectional studies was used to objectively evaluate the methodological quality of each included study. Independent appraisals were completed by three reviewers (ASA, DMK, and JMK), and disagreements were settled by consensus or discussion with a fourth reviewer. Excluded studies were those with scores below the minimal quality level.
Study Selection
All relevant studies published up to September 09/2025, were included in this study. The selected and retrieved studies were managed using EndNote version 7 software. After the initial database searches, duplicates were removed by exporting all retrieved records into EndNote version 7. The titles and abstracts of the found articles were then compared to the predetermined eligibility criteria by two independent reviewers (TKD and ASA). Full-text articles were obtained for in-depth analysis if they seemed to fit the inclusion requirements or if the abstract did not provide enough details.
Every paper was evaluated for methodological quality, adequacy of result reporting, and relevance throughout the full-text review. Any disagreements among reviewers during the selection process were settled by discussion, and a third reviewer (JMK) was engaged to reach a resolution if consensus could not be reached. To ensure transparency, the justifications for the exclusion of studies such as an irrelevant research population, a qualitative design devoid of quantitative data, or a failure to submit results were methodically documented. The PRISMA diagram (Figure 1) shows the sequence of study identification, screening, eligibility, and inclusion. The selection process was conducted in accordance with the PRISMA 2020 principles.
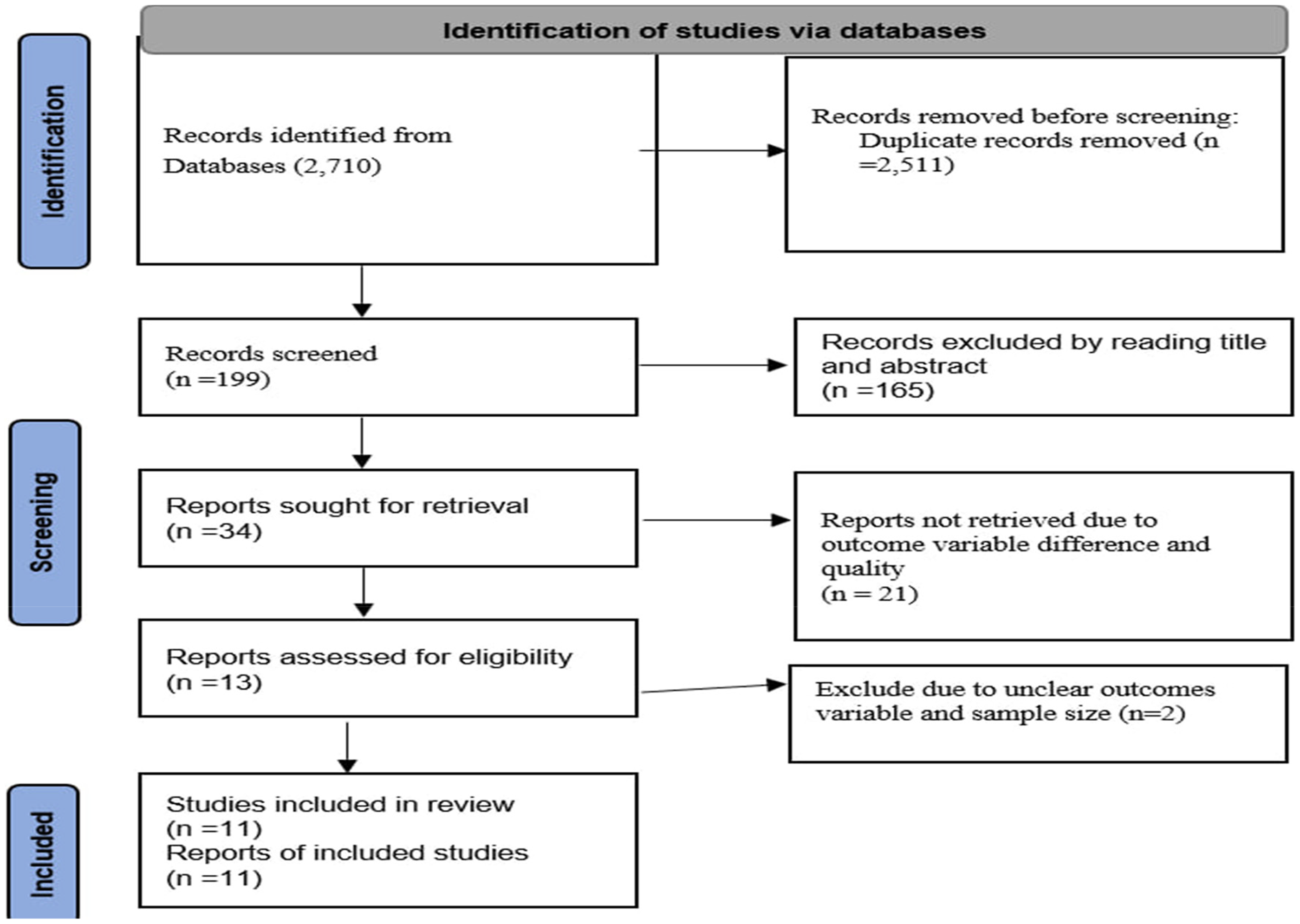
PRISMA flow diagram of study selection for HIV index testing and its associated factors in Ethiopia, 2025.
Condition
HIV index testing
Context
In Ethiopia
Population
HIV index individuals
Outcomes
The primary outcomes were the pooled acceptance of HIV index testing and associated factors among people living with HIV.
Data Extraction Process and Main Data Items
We used a pre-piloted, consistent data extraction format to obtain the necessary data from the included papers. Using the Newcastle Ottawa Scale (NOS) data extraction form (SF1), the three reviewers (ASA, DMK, and JMK) separately extracted the data. During the data extraction process, disagreements were settled by consensus and debate. The following study characteristics were extracted from the included studies: names of the first authors, region, study year, publication year, study design, study setting, participants, sample size, data collection method, response rate, and outcome measures (HIV index testing or not and its associated factor).
Inclusion and Exclusion Criteria
The studies included in this review were selected according to predefined criteria, focusing on the study population, exposure, and outcomes of interest. Studies were included if they were carried out in Ethiopia, reported on ART clients’ acceptance of index case HIV testing, reported on acceptance-related variables (eg, ART duration, disclosure status), included quantitative research, such as case-control, cohort, or cross-sectional studies, publication in English and supplied enough information to calculate odds ratios. Excluded studies were: those that were purely qualitative, did not report outcome of interest, were abstracts from conferences, case studies, editorials, or commentary that lacked complete data.
Three authors were rigorously evaluated for each study selected for inclusion in the systematic review and meta-analysis to assess its methodology and examine how effectively it addresses potential biases in design, implementation, and analysis. The methodological quality and risk of bias of each included article were assessed by three authors (ASA, DMK, and JMK) independently using the NOS critical appraisal checklist. 8
Statistical Analysis
An Excel spreadsheet was used to extract the data from each original study, which was then imported into STATA version 17.1 for in-depth analysis. To determine Ethiopia's pooled prevalence of acceptance of index HIV testing and related factors, meta-analyses were conducted. Heterogeneity across individual studies in terms of population characteristics, study designs, and measured outcomes was addressed by adhering to standard methodological principles. Each primary study was effectively described using a summary table.
Using a random-effects model, the pooled prevalence of acceptance of index HIV testing was determined. Results were displayed graphically in a forest plot with 95% confidence intervals (CIs), and the I2 statistic was used to measure study heterogeneity. To investigate possible sources of variation and take into consideration the observed heterogeneity, sensitivity and subgroup analyses were performed. A weighted Egger's regression test and funnel plot asymmetry were used to assess publication bias; a p-value of less than 0.05 was deemed suggestive of significant publication bias.
Result
Selection Result
Overall 2710 articles were retrieved from the online database. After being exported to EndNote 7, eleven studies included in this study.
Characteristics of the Included Studies
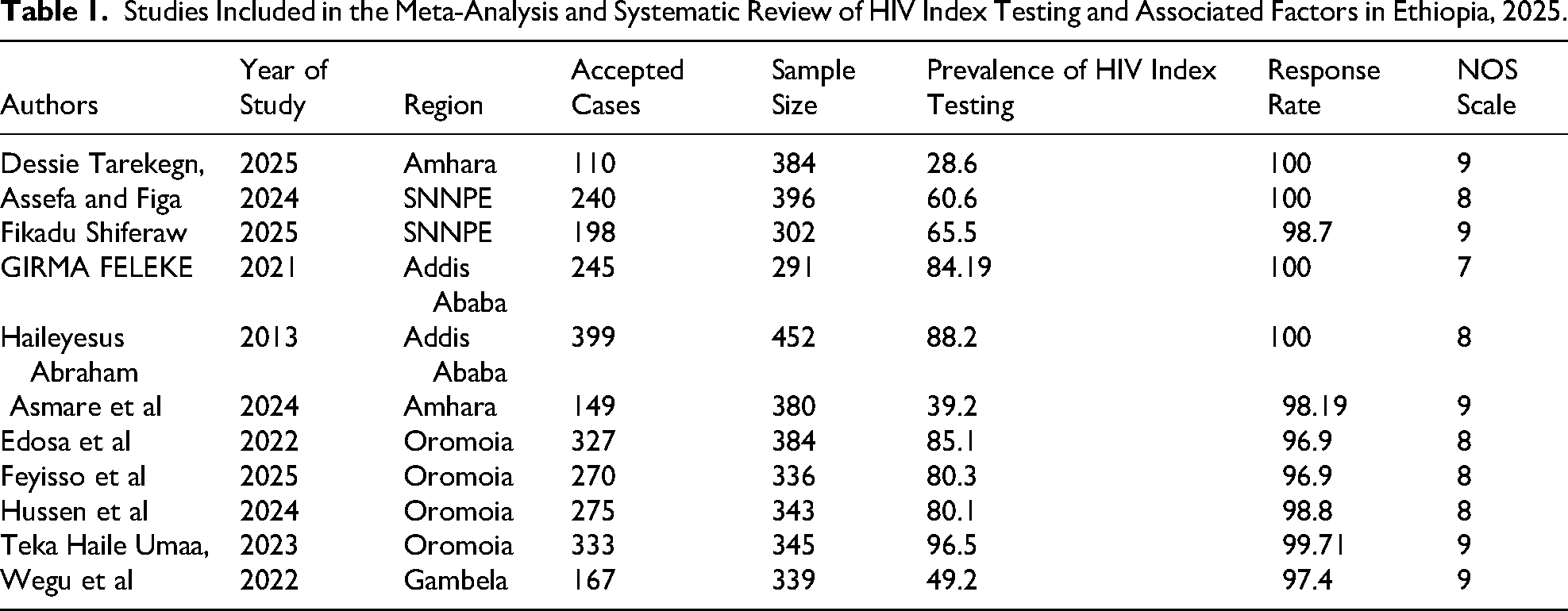
This analysis includes 11 cross-sectional studies with 3952 HIV index patients in total. Among those, 2 studies were from the Amhara region,9,10 4 studies were from Oromia,11–14 2 were from Addis Ababa,15,16 and 2 were from the Southern Nations, Nationalities, and Peoples’ Region of Ethiopia,7,17 and 1 study from Gambela 18 (Table 1).
Studies Included in the Meta-Analysis and Systematic Review of HIV Index Testing and Associated Factors in Ethiopia, 2025.
Pooled Acceptance of the Index Case HIV Testing
Among ART patients, the overall pooled acceptance rate of HIV index case testing is 68.9% (95% CI: 57.6% - 80.2%) (Figure 2).

Forest plot for pooled prevalence of HIV index testing among HIV infected individuals in Ethiopia, 2025.
Subgroup Analysis
Potential causes of the observed heterogeneity were investigated by subgroup analysis.
Research published after 2022 had a much higher acceptance rate (80.7%) than studies published prior to 2022 (59.1%), indicating a potential upward trend over time. By Region: There were significant regional differences in acceptance rates. In South Ethiopia (39.0%) and the Amhara & Gambela areas (39.0%), they were significantly lower. The Oromia region (85.6%) and the capital city of Addis Ababa (86.2%), on the other hand, had extremely high approval rates. Both the year of publication and the region were found to be statistically significant drivers of variability (Table 2).
Subgroup Analysis for HIV Index Testing and its Associate Factor Among People Live with HIV in Ethiopia, 2025.

Publication Bias and Heterogeneity Test
Egger's test confirmed possible small-study effects and was significant (p < .000). The significant test result, however, appears to be more likely due to the strong clinical heterogeneity among studies than to publication bias, as indicated by the visual symmetry of the funnel plot(Figure 3) and the trim-and-fill analysis, which imputed no missing studies.

Funnel plot for HIV index testing among people living with HIV in Ethiopia, 2025.
Meta Regression
The meta-regression indicates that while sample size does not significantly explain the variability, region and year of publication appear to be significant factors of variation in the acceptance of HIV index testing across studies. This suggests that the acceptance of HIV testing in Ethiopia is influenced by both region and year of publication (Table 3).
Meta Regression for HIV Index Among People Living with HIV in Ethiopia, 2025.

Sensitivity Analysis
The sensitivity analysis, which is shown in the accompanying figure, is a technique to evaluate the general robustness of the pooled result. One by one, the studies are eliminated, and the pooled estimate is recalculated. The research revealed that the total pooled acceptance percentage of 68.9% was not excessively impacted by any one study. This suggests that the meta-analysis's conclusion is trustworthy and consistent (Figure 4).

Sensitivity analysis for HIV index testing among people living with HIV in Ethiopia, 2025.
Factors Associated with HIV Index Testing
The study found a number of important variables that were strongly linked to a greater chance of accepting index case testing: Urban Residency: Patients in urban regions were 2.0 times more likely than those in rural areas to receive index testing (AOR: 2.0; 95% CI: 1.29, 2.71).
HIV Status Disclosure: Patients were 2.87 times more likely to agree to have their spouses or kids tested if they had told someone they were HIV positive (AOR: 2.87; 95% CI: 1.84, 3.91).
ART Duration >1 Year: Compared to patients with shorter treatment durations, those who had been on antiretroviral therapy for more than a year had a 2.06-fold higher likelihood (AOR: 2.06; 95% CI: 1.30, 2.82) of accepting index case testing (Table 4).
Factors Associated with HIV Index Testing Among People Living with HIV in Ethiopia, 2025.

Disclosure HIV Status Meta Regression
A univariable meta-regression was conducted to examine whether particular study-level characteristics could account for the remaining variation in the effect sizes, given the significant result of Egger's test for small-study effects (p = .020) and low statistical heterogeneity (I2 = 1.82%; Q-test p-value = .411). The study's location (p = .059), year of publication (p = .102), and sample size (p = .239) did not significantly alter the relationship between disclosure and acceptance (Table 5).
Possible Moderators of the Association Between Disclosure of HIV Status and Acceptance of Index Case Testing: A Meta-Regression Analysis.

Discussion
The purpose of this systematic review and meta-analysis was to ascertain the pooled acceptance of index case HIV testing and the factors related to it among Ethiopian ART patients. According to the study, which combined data from several studies, the pooled acceptance percentage of index case testing was 68.9% (95% CI: 57.6% - 80.2%). While a majority of ART patients are eager to participate in a crucial preventive strategy, this data shows that almost one-third are still apprehensive, indicating a considerable gap that needs focused intervention.
A significant degree of statistical heterogeneity (I2) was found in the meta-analysis's findings, suggesting that acceptance rates varied significantly among the included studies. However, the variation was not random; our meta-regression and subgroup analyses were able to identify particular study-level factors that contributed significantly to this difference. This takes the research beyond a single national statistic to a more detailed knowledge of regional and temporal dynamics and gives important context when analyzing the pooled acceptance rate.
Subgroup analysis revealed significant fresh insights into the causes of this heterogeneity. The acceptance rate of studies published after 2022 was significantly higher (80.7%) than that of studies published prior to 2022 (59.1%), suggesting a positive temporal trend. This progress could be attributed to the increased acceptance of HIV testing, the eradication of stigma through persistent public health campaigns, and the increasing assurance of medical professionals in prescribing index testing over time. The regional differences were even more pronounced. South Ethiopia and the Amhara & Gambela areas have much lower acceptance rates (both 39.0%),9–11 which contrast sharply with the unusually high rates in Oromia (85.6%)12–14 and Addis Ababa (86.2%).15,16
These disparities are probably caused by differences in healthcare facilities, access, and the rigors nature of health education efforts. In cities like Addis Ababa, there are more medical facilities, trained staff, and comprehensive HIV services available, which could lead to higher acceptance rates. 4 On the other hand, people may be discouraged from embracing index testing for their spouses and children in rural and isolated areas due to structural obstacles, increased stigma, and restricted access to private testing services. 19
The meta-regression quantitatively validated the patterns found in the subgroup analysis by statistically confirming that the year of publication and geographic region were significant predictors of the variation in acceptance rates (p-value < .05 for both). This offers strong evidence that the discrepancies are systematic and not the result of random variation. It firmly emphasizes the need to modify index testing approaches to particular geographic and temporal circumstances rather than relying on a national plan that works for everyone. These regional and temporal findings are further supported by the fact that sample size was not a significant source of discrepancy.
In Nigeria, the national HIV program was able to track down 97.7% of index cases’ contacts, ensure that 85.2% of those contacts were tested for HIV, and attain a 68.3% disclosure rate among index cases. 20 Compared to Ethiopia's national average, these figures show a significantly higher degree of contact tracing and index testing acceptance. This discrepancy would indicate that Nigeria have higher implementation fidelity and more reliable partner notification systems. Ethiopia has lower HIV index testing results than Kenya, where acceptance rate is 97%. 21 This discrepancy could be explained by differences in the study period, study area, and sociodemographic traits of the index HIV patients.
Ethiopia's findings are higher to those of research from eastern China. 22 Several factors, such as variations in the research duration, the architecture of the health system, and the methods employed for partner notification and index testing, could be responsible for this disparity. Furthermore, the chance that partners will present for testing and the desire of index cases to participate in partner notification may be influenced by cultural and societal norms regarding HIV disclosure, stigma, and healthcare-seeking behavior. While, it is consistent with other meta-analysis and systematic review conducted in China (68%). 23
Sample size, study period, and region were statistically confirmed to be important contributors to the observed heterogeneity by the meta-regression (p-value < .001 for all). This provides strong support for the subgroup findings and emphasizes the need to customize index testing methods for certain geographical circumstances instead of using a national strategy that works for everyone.
A clear road map for improving index testing acceptance is provided by the identification of related factors. The notion of spatial inequities is further evidenced by the substantial correlation (AOR: 2.0) between acceptance and urban residency. Higher levels of HIV information, more varied and easily accessible healthcare alternatives, and improved anonymity all help to lower perceived obstacles to testing, which is likely to benefit urban dwellers. 4
Disclosure of HIV status was the most powerful predictor (AOR: 2.87). A crucial phase in the cascade of care that lessens self-stigma, fosters a network of support, and instills a sense of duty to safeguard partners and children is disclosure. Index testing is legitimately and effectively hampered by the severe dread of social rejection, violence, and abandonment that patients who have not revealed their status confront. 6 Therefore, interventions that promote safe disclosure like community sensitization and couple's counseling are essential to increasing the acceptance of index testing and not merely supplementary.
Additionally, the likelihood of accepting index testing was more than doubled for patients who had been on ART for more than a year (AOR: 2.06). According to this, extended use of healthcare services promotes confidence in the medical system, enhances knowledge of the advantages of viral suppression and prevention, and permits more gradual, counselor-assisted decision-making about partner and kid testing. 20 It emphasizes the value of ongoing education and counselling not only at the beginning of ART but also throughout a patient's lifetime.
Conclusion
This meta-analysis concludes nearly 70% of Ethiopian ART patients consent to index case testing, which is a positive but below-optimal rate for efficient epidemic prevention. While the significant regional inequalities necessitate customized, decentralized public health measures rather than a standardized national approach, the significant impact of urban residency, HIV status disclosure, and treatment duration reveals actionable targets for clinical intervention. In the end, maximizing the promise of index testing as a pillar of HIV prevention in Ethiopia requires resolving differences in healthcare access and bolstering counseling support for disclosure.
Implications of the Research and Recommendations
At the individual and public health levels, the results of this meta-analysis have significant implications for enhancing HIV index case testing in Ethiopia. Enhancing ART patients’ access to counselling and support services should be the main goal of individual-level treatments. It is essential to promote long-term ART engagement, safe HIV status disclosure, and focused assistance for patients in low-acceptance or rural areas. Peer support groups, follow-up visits, and structured couple's counselling can all assist patients in overcoming stigma and fear, which increases the possibility that spouses and kids would agree to index testing.
The study emphasizes the necessity of region-specific approaches to address significant differences in acceptance rates, which vary from 39.0% to more than 86.0%, at the public health level. Reducing structural barriers requires investments in rural healthcare infrastructure, such as increasing access to qualified healthcare professionals and HIV testing services. Regional stigma-reduction initiatives can address social and cultural barriers, and training health professionals in partner notification protocols and index testing counselling can improve programme implementation. To guarantee fair access and successful results, public health policy should also encourage continuous monitoring and assessment of index testing initiatives.
Based on the finding meta-analysis, the following recommendation is proposed:
Implement region-specific actions to address the huge variations in acceptance, including concentrated spending in stigma reduction and access to testing in low-performing regions like south Ethiopia and Amhara and Gambela. To assist patients safely disclose their status, which is the best indicator of accepting index testing, incorporate structured disclosure support such as couple's counselling and support groups into standard HIV care. Consider follow-up visits as continuing chances for medical professionals to promote and assist index case testing in order to leverage on long-term ART participation. To create and assess culturally specific strategies that address regional barriers to testing adoption, conduct more implementation studies in low-acceptance areas.
Limitations
Despite being investigated by subgroup analysis and meta-regression, the significant heterogeneity observed in this study suggests underlying differences in research methodology or unmeasured contextual factors that were not fully taken into consideration. Due to the unique focus on Ethiopian studies and the possibility of missed research in regional languages because our search was restricted to English publications, the results might also be less applicable to other nations.
Supplemental Material
sj-docx-1-jia-10.1177_23259582251409296 - Supplemental material for Acceptance of HIV Index Case Testing and Associated Factors Among People Living with HIV and on Anti-Retroviral Therapy in Ethiopia: Meta-Analysis and Systematic Review
Supplemental material, sj-docx-1-jia-10.1177_23259582251409296 for Acceptance of HIV Index Case Testing and Associated Factors Among People Living with HIV and on Anti-Retroviral Therapy in Ethiopia: Meta-Analysis and Systematic Review by Addisu Simachew Asgai, Tsegaamlak Kumelachew Derse, Desalegn Mitiku Kidie, Tadios Lidetu, Demeke Shumu Negesse, Menberu Gete and Jenberu Mekurianew Kelkay in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-docx-2-jia-10.1177_23259582251409296 - Supplemental material for Acceptance of HIV Index Case Testing and Associated Factors Among People Living with HIV and on Anti-Retroviral Therapy in Ethiopia: Meta-Analysis and Systematic Review
Supplemental material, sj-docx-2-jia-10.1177_23259582251409296 for Acceptance of HIV Index Case Testing and Associated Factors Among People Living with HIV and on Anti-Retroviral Therapy in Ethiopia: Meta-Analysis and Systematic Review by Addisu Simachew Asgai, Tsegaamlak Kumelachew Derse, Desalegn Mitiku Kidie, Tadios Lidetu, Demeke Shumu Negesse, Menberu Gete and Jenberu Mekurianew Kelkay in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Abbreviations
Acknowledgements
We would like to thank all the authors of the studies included in this systematic review and meta-analysis.
ORCID iDs
Ethics Approval and Consent to Participate
This study is a meta-analysis and systematic review of previously published studies, therefore, ethical approval and consent to participant are not applicable.
Consent for Publication
Not applicable.
Authors’ Contributions
Conceptualization: ASA. Data curation: ASA, DMK. Formal analysis: ASA. Investigation: ASA, TKD. Methodology: ASA, TL. Project administration: TKD. Software: ASA. Supervision: MG. Validation: JMK and MG, Visualization: ASA, Writing original draft: ASA, DMK, TKD, Writing review & editing: ASA, DSN and JMK.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data is provided within the manuscript or supplementary information files.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
