Abstract

Keywords
Dear Editor,
During a prevalence survey of young people conducted between October 2022 and March 2023 in Zimbabwe, we collected dried blood spots (DBS) from youth aged 18-24 within 24 clusters in 3 provinces and tested them for HIV and viral load. 1 Participants who self-reported as HIV negative or not knowing their status, but whose HIV antibody test was positive and whose viral load was under 10,000 copies/ml, were further tested for the presence of antiretroviral drugs (ARVs) in the same sample to determine whether they were in fact receiving treatment. Out of 17,682 survey participants, 791 tested HIV-positive without self-reporting as HIV-positive, and 497/791 had a viral load <10,000 copies. Of these, 486 survey participants had ARV tests on their DBS sample. We tested for efavirenz, tenofovir, atazanavir, ritonavir, nevirapine, abacavir, lamivudine and dolutegravir. We used chi-square tests to investigate the association between efavirenz detection and exposure variables.
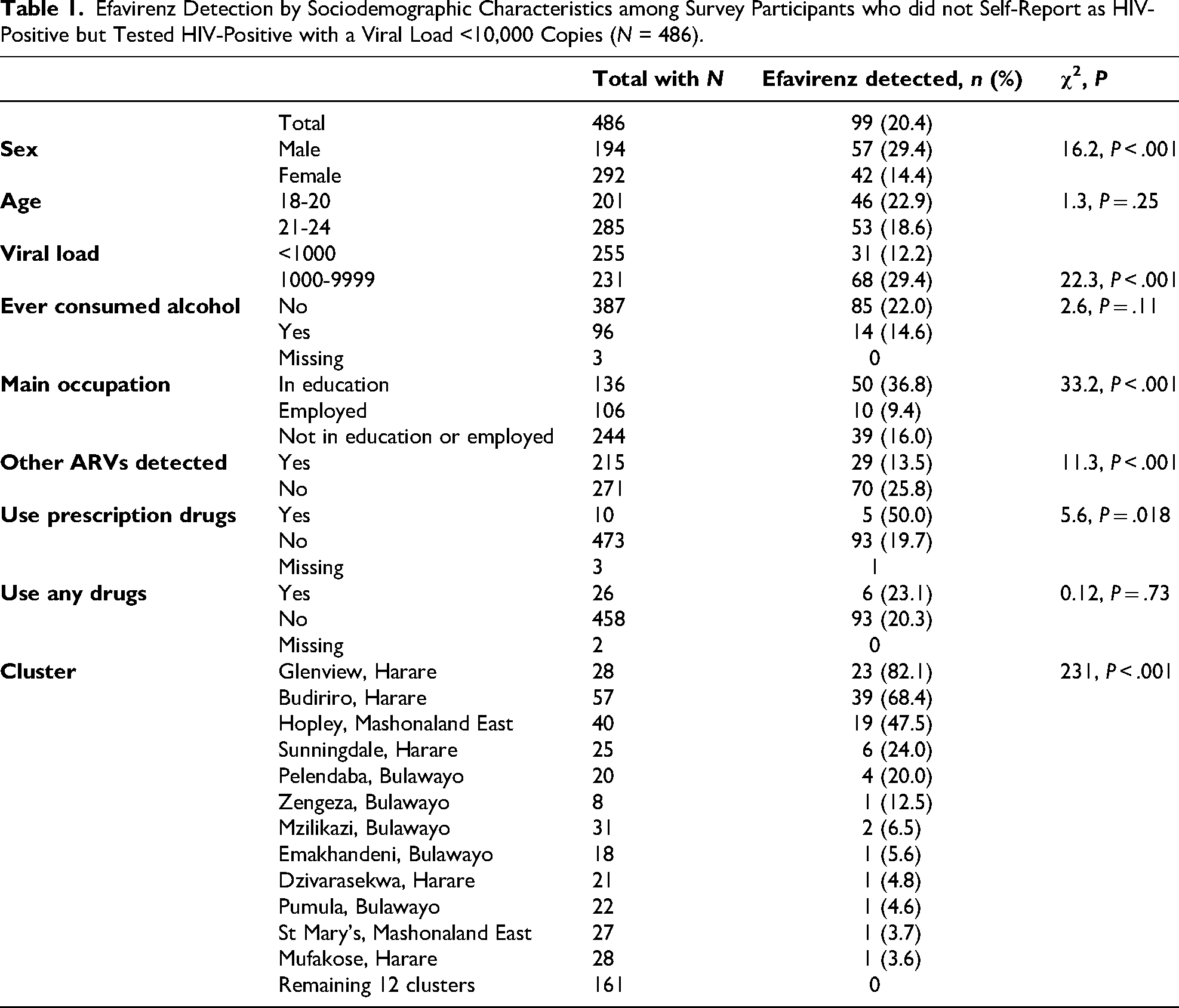
Efavirenz was detected in 99 (20.4%) of samples from youth who did not report being HIV-positive and had a viral load <10,000 copies (Table 1). In 70 (70.7%) of these, no other ARV was detected. Detection of efavirenz was particularly common in 3 of the 24 clusters surveyed (Glenview, Budiriro, Hopley), while 12 clusters had no cases at all. Efavirenz detection was more common in males (29.4%) than females (14.4%; χ2 = 16.2, P < .001), more common among those currently in education than not (χ2 = 33.2, P < .001), and more common among participants with an HIV viral load >1000 copies (defined as unsuppressed by WHO 2 ) than those virally suppressed (χ2 = 22.3, P < .001). By contrast, the other 7 ARVs were associated with female sex and viral suppression. The survey included questions about use of recreational drugs. Only 5.1% of the youth reported any substance use. Substance use was not associated with efavirenz detection (χ2 = 0.12, P = .73), but self-reported use of prescription drugs for non-medical reasons was associated with efavirenz detection (χ2 = 5.6, P = .018).
Efavirenz Detection by Sociodemographic Characteristics among Survey Participants who did not Self-Report as HIV-Positive but Tested HIV-Positive with a Viral Load <10,000 Copies (N = 486).
Efavirenz has psychoactive properties and is an ingredient in the drug cocktail known in South Africa as nyaope or whoonga. 3 Nyaope and the use of ARVs for recreational purposes in Zimbabwe have been reported in local mainstream media. A survey of 293 people who used drugs in 2022 found that 3/53 (5.7%) in Bulawayo and 1/61 (1.6%) in Harare reported using nyaope in the past year, while 20/53 (37.7%) in Bulawayo and 27/61 (44.3%) in Harare reported using cough syrup, and 4/53 (7.5%) in Bulawayo and 16/61 (26.2%) in Harare reported taking other pharmaceuticals for recreational purposes. 4 In focus group discussions, participants reported use of ARVs, haloperidol, diazepam and other pharmaceuticals for non-medical reasons. Additionally, focus group participants in Manicaland reported use of nyaope. 4 In South Africa, 8 out of 43 participants in a qualitative study in Soweto reported recreational use of ARVs broadly, with 1 participant specifically mentioning efavirenz. 5 In a qualitative study on potential recreational use of HIV medication in Durban, 3 participants reported efavirenz being an active ingredient in whoonga. 6
We posit that the distribution of efavirenz detection in this survey among those who did not report being HIV-positive is suggestive of recreational use by some participants rather than use of efavirenz as part of antiretroviral therapy. While the 29 participants in whom other ARVs were detected are likely to be diagnosed and on treatment, the evidence suggests that at least some of the 70 in whom only efavirenz was detected were taking it recreationally. These young people may not have known they were HIV-positive. This poses risks of developing resistance to efavirenz and also of interaction with other medications. The clusters where efavirenz detection was most prevalent, Glenview and Budiriro, which border each other, are 2 of the oldest and most high-density neighbourhoods in Harare. Hopley is a new and informal settlement on the outskirts of Harare with reports of high unemployment, low incomes and high levels of sex work and drug use. 7
The possible recreational use of efavirenz by young people in urban areas is of public health concern, particularly given recent reports of a substantial increase in substance use among youth in Zimbabwe. 8 Tenofovir/lamivudine/dolutegravir is recommended for first-line therapy in Zimbabwe, with the exception of women who might become pregnant or are in their first trimester, for whom efavirenz is the recommended alternative. 9 Thus, efavirenz continues to have an important role in HIV treatment in Zimbabwe, while its detection in young men is suggestive of use outside therapy. The use of efavirenz-based cocktails has been shown to have various biopsychosocial consequences and warrants further investigation. Further research is required to determine the epidemiology of recreational efavirenz use in Zimbabwe. This will enable policymakers to develop interventions for harm reduction.
Footnotes
Acknowledgement
We thank the CHIEDZA staff and survey participants.
Authors’ Contribution
R.A.F. was the principal investigator of the CHIEDZA trial. R.A.F. and V.S. conceived the study. K.G. analysed the samples. V.S. conducted data analysis. R.B.-J. and K.H. wrote the first draft. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval for the CHIEDZA study was granted by the Medical Research Council of Zimbabwe (reference number: MRCZ/A/2387), the Institutional Review Board of the Biomedical Research and Training Institute (ref: AP149/2018) and the London School of Hygiene & Tropical Medicine (LSHTM) Research Ethics Committee (ref: 12063).
Informed Consent
Written informed consent was obtained from survey participants. Eligible individuals were shown a video of the study procedures enacted and narrated by the study team on a tablet with narration in English, Shona or Ndebele. Consent was documented electronically through a signature or fingerprint recorded on the tablet. Participants retained a brief information sheet, link to the video, and signed paper copy of the consent form.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CHIEDZA was funded by a Wellcome Trust award to Rashida Ferrand (ref 206316/Z/17/Z).
