Abstract
Background
Real-world comparisons of initial antiretroviral third-drug classes are limited in middle-income settings. We evaluated whether the initial third-drug class predicts time to loss of virologic suppression in Colombia.
Methods
Historical cohort of 609 ART-naïve adults (2016-2020). Outcome: first HIV RNA ≥200 copies/mL. We compared NNRTI, PI, and INSTI regimens using adjusted and IPTW-weighted Cox models.
Results
Regimens were NNRTI (72%), PI (20%), and INSTI (8%); 19.9% lost suppression. Compared with NNRTI, PIs showed higher but nonsignificant hazards (aHR 1.50, 95% CI 0.98-2.27). For INSTIs, we did not detect significant differences (aHR 1.22, 95% CI 0.64-2.31). Diagnosis-to-ART delays and treatment interruptions were associated with loss of suppression.
Conclusions
Treatment continuity and timely initiation predicted sustained suppression better than drug class. Low INSTI usage limits statistical power to detect differences. Structural barriers to continuity may limit the real-world benefit of newer regimens.
Plain Language Summary Title
Impact of treatment continuity and drug choice on HIV viral suppression in a Colombian cohort (2016-2020)
Plain Language Summary
Why was the study done? When people start HIV treatment, most first regimens include 2 “backbone” medicines plus a third medicine from one of several common drug groups. Newer “integrase inhibitor” drugs are often recommended, but there is limited evidence from everyday clinical care in middle-income countries. What did the researchers do? We reviewed medical records from 609 adults who started their first HIV treatment between 2016 and 2020 in 9 HIV care centers in Colombia. We checked whether the type of third medicine in the first regimen was linked to losing viral suppression over the next 2 years. Viral suppression means the amount of HIV in the blood stays very low; losing suppression means the virus becomes detectable again. What did the researchers find? About 1 in 5 people lost viral suppression within 2 years. Overall, we did not detect statistically significant differences in loss of suppression between the main third-medicine groups. However, the number of patients taking newer drugs was small, which limits the ability to detect differences. In contrast, treatment interruption (stopping treatment for a time) and starting treatment later after diagnosis were strongly linked to losing suppression. What do the findings mean? These results suggest that, in routine Colombian care, protecting continuous access to HIV medicines, supporting people to stay on treatment, and reducing administrative barriers are critical to maximize the real-world effectiveness of any drug regimen chosen at the start.
Keywords
Introduction
Antiretroviral therapy (ART) has transformed human immunodeficiency virus (HIV) infection into a controllable chronic condition, substantially improving survival and quality of life among people living with HIV (PLHIV). 1 Despite these advances, achieving and maintaining virologic suppression in routine care remains challenging, as lifelong treatment, barriers to continuity of access, and suboptimal adherence can lead to viral rebound and resistance. 2
Over time, the development of new antiretroviral agents has enabled more potent, safer, and simpler regimens, with improved tolerability and dosing schedules that support long-term adherence. 3 In treatment-naïve individuals, initial ART regimens have traditionally consisted of 2 nucleoside reverse transcriptase inhibitors (NRTIs) plus a third agent, most commonly an integrase strand transfer inhibitor (INSTI), a nonnucleoside reverse transcriptase inhibitor (NNRTI), or a boosted protease inhibitor (PI).4–6
Randomized clinical trials have shown that INSTI-based regimens achieve high rates of virologic suppression and have favorable tolerability and drug-drug interaction profiles compared with many NNRTI- or PI-based regimens.7,8 Consequently, international guidelines—and subsequently the 2021 Colombian national guideline—recommend INSTI-based regimens, particularly dolutegravir (DTG), as preferred options for initiating ART.4,6 However, in many middle-income settings, INSTIs have coexisted with NNRTI- and PI-based regimens for prolonged periods, and comparative real-world evidence on regimen durability and maintenance of suppression remains limited.
In Colombia, during the period when this cohort was assembled (2016-2020), routine care was still shaped by earlier recommendations, with predominant use of NNRTI-based regimens (notably efavirenz [EFV]) and substantial heterogeneity in initial treatment choices. Even after the 2021 guideline update, continued EFV use and variability in practice persist in some care settings. 9 This context provides an opportunity to evaluate the comparative effectiveness of third-drug classes under real-world conditions during a transition toward INSTI-based regimens.
Therefore, we aimed to assess the association between the initial third-drug class (INSTI, NNRTI, or PI) and time to loss of virologic suppression in ART-naïve PLHIV receiving routine care in Colombia. By leveraging multicenter programmatic data, our findings seek to inform clinical decision-making and service delivery in Colombia and other middle-income settings where the adoption of INSTI-based regimens has been gradual and heterogeneous. 10
Methods
Study Design
We conducted an analytical observational study using a historical cohort design to evaluate the association between the class of the initial third antiretroviral (ARV) drug in the first ART regimen (NNRTI, PI, or INSTI) and time to loss of virologic suppression during follow-up.
Setting
Data were obtained from clinical records at 9 specialized HIV care centers in Colombia, providing care to people living with HIV (PLHIV) across 18 regions of the country. In Colombia, the General System of Social Security in Health (SGSSS) guarantees universal coverage for HIV diagnosis and treatment. Consequently, antiretroviral therapy is provided free of charge to patients, regardless of their socioeconomic status. Access to specific drug classes during the study period was determined by national clinical guidelines and institutional formularies, rather than by the patients’ ability to pay. We included individuals who initiated ART between January 2016 and December 2020. Clinical information was abstracted at 4 time points: at HIV diagnosis (before ART initiation) and at routine follow-up visits approximately 6, 12, and 24 months after ART initiation.
Participants
Eligibility Criteria
We included adolescents and adults aged ≥14 years with confirmed HIV infection who initiated ART at one of the participating centers during the study period.
Sampling Strategy
Sampling was performed using simple random sampling within each center, with sampling fractions proportional to the number of eligible patients per center.
Exclusion Criteria
Individuals were excluded if the clinical record contained an explicit statement indicating that their information should not be used for research purposes.
Variables
Exposure
Initial third-drug class: pharmacologic class of the third ARV in the initial regimen (NNRTI, INSTI, or boosted PI).
Outcomes
Loss of virologic suppression (event): HIV RNA ≥200 copies/mL at any follow-up measurement (approximately 6, 12, or 24 months).
Time to event: time in months from ART initiation to the first follow-up time point at which HIV RNA was ≥200 copies/mL (visit-based event time).
Censoring: participants were censored at the last available follow-up visit with HIV RNA <200 copies/mL (up to 24 months), or at the follow-up visit when they changed the third-drug class for reasons not related to virologic failure (see below). Administrative censoring was applied at 24 months.
Secondary outcomes (descriptive): evolution of CD4+ T-cell count and body weight over follow-up, stratified by initial third-drug class, restricting these analyses to individuals who did not change third-drug class during the 24-month follow-up.
Covariates (baseline unless otherwise specified)
The following variables were recorded from clinical charts:
(i) Sex: biological sex at birth (female/male); (ii) Age: age in completed years at ART initiation, treated as a continuous variable in models and categorized as <40 and ≥40 years for descriptive purposes, given its potential effect on ART adherence in previous Colombian studies and adherence reviews11,12; (iii) Race/ethnicity: categorized as mestizo or Afro-descendant according to the clinical record; (iv) Municipality of care versus residence: based on the municipality of residence and municipality of care, a dichotomous variable was created (same vs different) as an approximation to care outside the municipality of residence; (v) Clinical stage of HIV infection: clinical stage at diagnosis according to the revised CDC classification, 13 recorded as a categorical variable; (vi) CD4+ T-cell count (LTCD4+): value in cells/µL at diagnosis and at 6, 12, and 24 months after ART initiation. It was analyzed as a continuous variable and, for some descriptive and analytical purposes, categorized as <200 versus ≥200 cells/µL, based on the ART effectiveness literature14–19; (vii) AIDS-defining event at diagnosis: presence/absence of an AIDS-defining event at the time of HIV diagnosis, classified according to CDC criteria 13 ; (viii) HIV viral load (VL): HIV RNA in copies/mL at diagnosis and at 6, 12, and 24 months. It was analyzed as a continuous variable and categorized as <100,000 versus ≥100,000 copies/mL for descriptive and causal analyses, following prior studies that use this threshold to discriminate risk and response to ART14–19; (ix) Initial ART backbone: NRTI combination in the initial regimen (eg, TDF/XTC, AZT/3TC). For the primary analysis, this was recategorized as “TDF/XTC” versus “Non-TDF/XTC,” given its recommendation as the preferred backbone in clinical practice guidelines 4 ; (x) Time from diagnosis to ART initiation: interval in days between the date of HIV diagnosis and the date of ART initiation, calculated from date variables. It was analyzed as a continuous variable and categorized as <30 versus ≥30 days, following the Colombian guideline recommendation to start ART within the first 30 days 4 ; (xi) Weight, height, and body mass index (BMI): weight (kg) and height (m) were recorded at ART initiation and at 6, 12, and 24 months. BMI (kg/m2) was calculated at each time point. Baseline BMI was analyzed as a continuous variable and categorized according to the World Health Organization classification (underweight, normal weight, overweight, and obesity).
Treatment changes and treatment continuity (post-baseline; complementary)
xii) Third-drug class change (censoring variable): yes/no for at least 1 change in the third-drug class during follow-up (eg, INSTI→PI or NNRTI; PI→INSTI). Changes within the same third-drug class were not considered. This variable was used to define censoring in survival analyses when the switch occurred for reasons other than virologic failure; xiii) ART interruption: because adherence assessed with formal instruments was not systematically recorded in charts, documented ART interruptions were used as a proxy for treatment discontinuity. ART interruption was defined as the presence of at least 1 interruption episode recorded at any follow-up visit (6, 12, or 24 months). Reasons for interruption documented in the clinical chart were extracted. Because this is a post-baseline variable and may lie on the causal pathway, it was examined in complementary analyses rather than treated as a baseline confounder.
Data Sources and Measurement
Data were abstracted from clinical records by trained staff (physicians and nurses at participating centers) and entered into REDCap20,21 following a standardized instruction manual. The database was exported to .csv format for analysis. We verified eligibility criteria, randomly replaced ineligible records, and conducted a 10% audit comparing electronic data against source clinical records.
Bias
To reduce selection bias, we used random sampling within centers with sampling fractions proportional to the eligible population size. To reduce information bias, abstraction procedures were standardized and audited. Confounding was addressed through (i) conventional multivariable adjustment informed by a directed acyclic graph (DAG) and (ii) propensity-score weighting (IPTW) to estimate marginal effects. Treatment interruption and treatment switching were handled cautiously as post-baseline processes (potential intermediates) and were not included as baseline confounders for the primary exposure-outcome association.
Study Size
Based on reports from the 9 centers, approximately 5260 PLHIV initiated ART between 2016 and 2020. From this source population, we performed simple random sampling weighted by center. The target sample size was calculated using prior comparative studies of INSTI-based regimens versus PI/NNRTI-based regimens at 48 to 96 weeks, assuming a 15%-20% absolute difference in the proportion achieving virologic suppression and an approximate 1:9 ratio between INSTI and non-INSTI regimens. With 80% power and a 2-sided 5% significance level, the required sample size was estimated at 610 participants (approximately 56 in the INSTI group and 540 in the NNRTI/PI groups).
Quantitative Variables
Continuous variables (age, baseline VL, baseline CD4, time from diagnosis to ART initiation) were modeled as continuous terms in primary models. For descriptive and selected analytical purposes, clinically meaningful thresholds were used: age (<40/≥40), baseline VL (<100,000/≥100,000 copies/mL), baseline CD4 (<200/≥200 cells/µL), and diagnosis-to-ART time (<30/≥30 days). BMI was analyzed continuously and categorized using WHO cutoffs.
Data Management and Missing Data
We conducted exploratory data-quality checks, including review of variable types, detection of outliers, and assessment of missingness patterns. Missingness was low overall (<1%) and was <9% for variables used in the main models. We handled missing covariate data using multiple imputation by chained equations (MICE), generating 20 imputed datasets, and combined model estimates using Rubin's rules. As a sensitivity analysis, we repeated primary models using complete-case data and obtained similar results. Outcome measures were not imputed.
Statistical Methods
Descriptive Analyses
Baseline cohort characteristics were summarized using medians and interquartile ranges for continuous variables, and frequencies and proportions for categorical variables. Comparisons by outcome status (loss of suppression vs no loss) used χ2 or Fisher's exact tests for categorical variables and Student's t-test or Mann-Whitney U test for continuous variables, as appropriate. Standardized mean differences (SMD) were computed to quantify baseline differences, with values <0.1 interpreted as good balance.
Time-to-Event Analyses
The primary outcome (time to loss of virologic suppression) was analyzed using Kaplan-Meier methods and Cox proportional hazards models. Kaplan-Meier curves were stratified by initial third-drug class and compared using the log-rank test. Because viral load was assessed at routine follow-up visits (approximately 6, 12, and 24 months), event times reflect the first follow-up time point at which HIV RNA was ≥200 copies/mL.
Multivariable Cox Models
We fitted 3 Cox models:
Crude model: third-drug class as the only covariate Conventional adjusted model: adjusted for baseline covariates identified via a DAG as potential common causes of exposure and outcome: sex, age, baseline viral load, baseline CD4+ T-cell count, and time from diagnosis to ART initiation (Figure 1). IPTW model: to estimate the marginal (population-average) effect of third-drug class.22,23

DAG of the assumed causal structure between the third antiretroviral drug class, treatment interruption, and loss of virologic suppression.
The DAG was developed using clinical and epidemiological knowledge to identify a minimally sufficient adjustment set.24,25
IPTW (Propensity Score) Approach
We estimated a multinomial propensity score for receiving each third-drug class (NNRTI, PI, INSTI) using multinomial logistic regression with NNRTI as the reference category. Covariates included sex, age (continuous), baseline VL (continuous), baseline CD4 (continuous), and time from diagnosis to ART initiation (continuous). Stabilized weights were calculated as:
Complementary Analyses
We described reasons for ART interruption by third-drug class using frequency tables and χ2 tests. We also described associations between ART interruption and loss of virologic suppression as complementary analyses, acknowledging that interruption is a post-baseline process and may act as an intermediate factor rather than a baseline confounder.
Software and Reproducibility
All analyses were conducted in R (version 4.3.0) within RStudio. We used tidyverse for data management, mice for multiple imputation, survival and survminer for survival analyses, nnet for multinomial logistic regression, survey and tableone for weighting/balance assessment and descriptive tables, and gtsummary and table1 for reporting.27–54 The analysis workflow was implemented in R Markdown to support transparency and reproducibility. 55
Results
A total of 609 individuals who initiated ART between 2016 and 2020 were included. The median age at ART initiation was 31.2 years (IQR 24.8-40.0), with a predominance of men (81.0%) and mestizo individuals (95.4%). Most participants initiated treatment with an NNRTI-based regimen as the third drug (72.2%), followed by boosted PIs (20.2%) and INSTIs (7.6%). The main NNRTI used was efavirenz (EFV), and among PIs, atazanavir/ritonavir (ATV/r) was the most common. Among those who initiated an INSTI-based regimen (n = 46), 63% received dolutegravir (DTG), 33% raltegravir (RAL), and 4% elvitegravir/cobicistat (EVG/c). At baseline, the median CD4+ T-cell count was 275 cells/µL (IQR 123-439) and the median viral load was 50,400 copies/mL (IQR 11,985-174,221). Approximately two-thirds of participants initiated ART ≥30 days after HIV diagnosis (68.1%), and 20.0% had an AIDS-defining event at diagnosis. A complete univariable description of baseline cohort characteristics is provided in the Supplemental material (Table S2).
Over the 24-month follow-up, 121 patients (19.9%) experienced loss of virologic suppression. Those who developed loss of suppression were, on average, slightly younger and had a longer interval between HIV diagnosis and ART initiation, as well as a markedly higher frequency of ART interruption during follow-up. In contrast, the distributions of sex, race/ethnicity, presence of an AIDS-defining event at diagnosis, baseline viral load, and baseline CD4+ T-cell count were similar between those with and without the outcome (Table 1).
Baseline Characteristics of the Cohort According to Virologic Suppression Status During Follow-up.
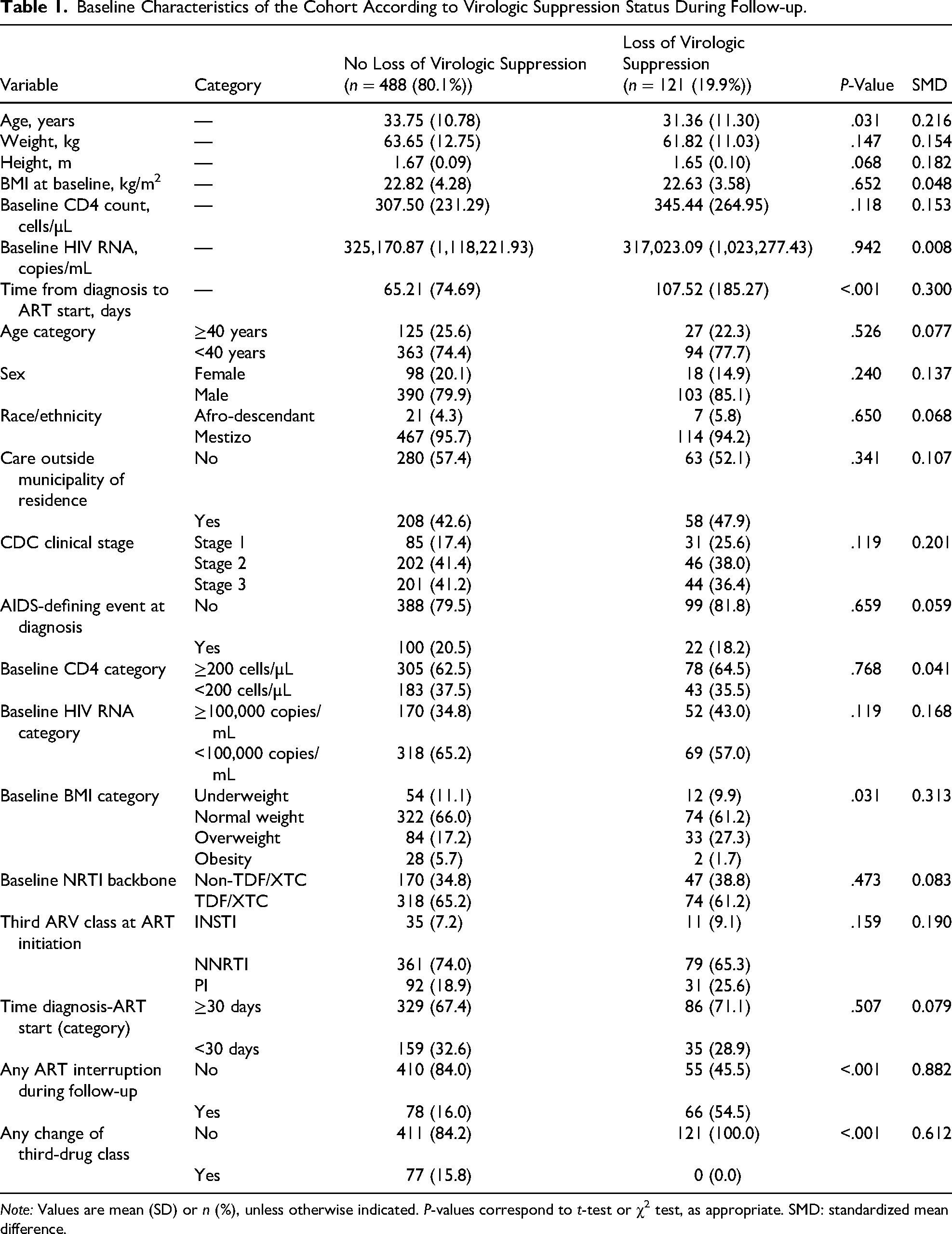
Note: Values are mean (SD) or n (%), unless otherwise indicated. P-values correspond to t-test or χ2 test, as appropriate. SMD: standardized mean difference.
Kaplan-Meier curves showed no statistically significant differences in virologic failure-free survival according to the class of the initial third ARV. Overall, the cumulative probability of maintaining virologic suppression through 24 months was similar across NNRTI-, PI-, and INSTI-based regimens, and the global log-rank test provided no evidence of differences between survival curves. Consistent with these findings, comparisons at fixed follow-up time points showed modest variation in the proportion of participants with virologic failure, with a statistically significant difference at 6 months (failure proportion: 4.3% in the INSTI group, 14.5% in the NNRTI group, and 19.5% in the PI group; P = .039), but no statistically significant differences at 12 months (P = .596) or 24 months (P = .078) (Figure 2). Detailed counts and proportions by time point are provided in the Supplemental material (Table S3).

Kaplan–Meier survival curves for time to loss of virologic suppression.
In the unadjusted Cox model, using NNRTI-based regimens as the reference, patients who initiated PIs had a hazard ratio (HR) of 1.47 (95% CI 0.97-2.23), and those who initiated INSTIs had an HR of 1.29 (95% CI 0.69-2.43), neither reaching statistical significance. After adjustment for the DAG-defined covariates (sex, age, baseline viral load, baseline CD4+ T-cell count, and time from diagnosis to ART initiation), the estimates remained of similar magnitude: HR 1.50 (95% CI 0.98-2.27) for PIs and HR 1.22 (95% CI 0.64-2.31) for INSTIs, compared with NNRTIs. In this model, time from HIV diagnosis to ART initiation was independently associated with failure (HR 1.002 per additional day, 95% CI 1.001-1.003). Detailed model outputs are provided in the Supplemental material (Table S5).
In the causal inference analysis, the multinomial propensity score model was used to estimate the probabilities of receiving each third-drug class as a function of baseline covariates. Before IPTW, standardized mean differences between treatment groups were low to moderate; after applying stabilized weights truncated at the first and 99th percentiles, all SMDs were <0.1, indicating adequate covariate balance across the 3 treatment groups (Table S4).
In the IPTW-weighted Cox model, results were consistent with those of the conventionally adjusted model: compared with NNRTI-based regimens, PI-based regimens showed an HR of 1.49 (95% CI 0.98-2.27), while INSTI-based regimens had an HR of 1.14 (95% CI 0.61-2.15), with no statistically significant differences (Table S5).
During follow-up, nearly one quarter of participants experienced at least 1 episode of ART interruption. This proportion differed by virologic outcome: among those who did not lose virologic suppression, 16.0% had any interruption, whereas among those with loss of suppression, 54.5% had at least 1 interruption over 24 months (Table 1). In bivariate analysis, ART interruption was strongly associated with loss of virologic suppression (P < .001), consistent with a potential mediating role rather than a baseline confounder.
Regarding the documented reasons for ART interruption, administrative causes were frequently recorded (21.6%-30.8% among those with interruption), although “other reasons” represented the largest category across groups (46.2%-66.7%). The overall distribution of recorded reasons differed across third-drug classes (P = .038) (Supplementary Table S6).
In the analysis of immunologic response among patients who did not change ART class during follow-up, CD4+ T-cell counts increased across all 3 treatment groups. Among those who initiated INSTIs, the median CD4+ count increased from 232 cells/µL at baseline to 472 cells/µL at 24 months; in NNRTI-based regimens, from 282 to 479 cells/µL; and in PI-based regimens, from 290 to 568 cells/µL over the same period (Figure 3).

CD4+ T-cell count trajectories over 24 months by initial third-drug class.
In parallel, body weight increased over follow-up in all 3 treatment groups. Median weight rose from approximately 61 to 62 kg at baseline to 66 to 69 kg at 24 months among participants receiving INSTIs, NNRTIs, or PIs. When stratified by baseline BMI category, the largest relative gains were observed among individuals who were underweight at baseline, whereas participants with overweight or obesity experienced smaller increases. Overall, this pattern was consistent with weight “normalization” over time rather than disproportionate weight gain attributable to any specific third-drug class (Figure 4).
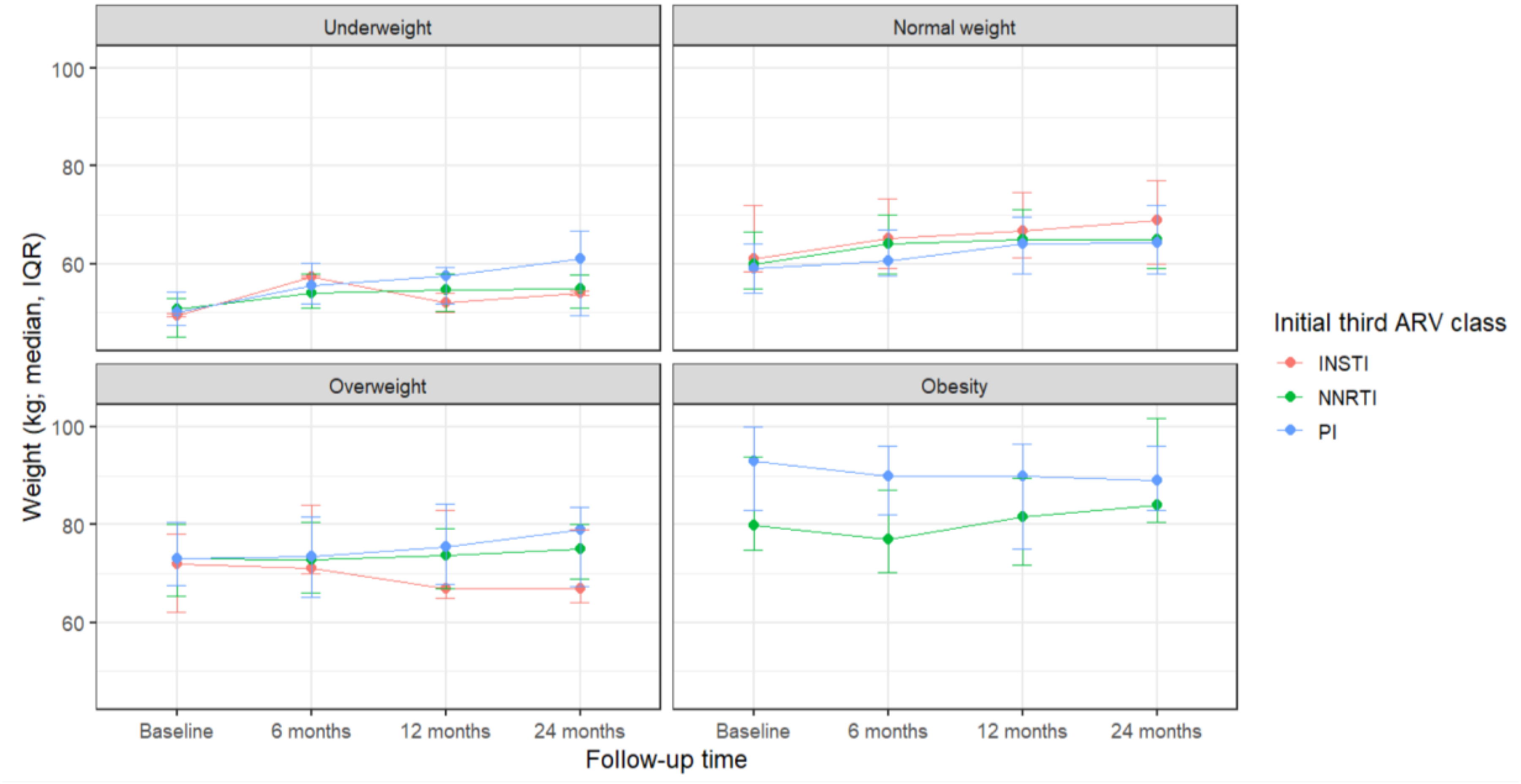
Weight trajectories by initial third ARV class, stratified by baseline BMI.
Discussion
This multicenter Colombian cohort study compared 3 initial third-drug classes (NNRTI, PI, and INSTI) with respect to time to loss of virologic suppression during 24 months of routine care. Across survival analyses—including Kaplan-Meier curves and Cox models (unadjusted, conventionally adjusted for a DAG-defined covariate set, and IPTW-weighted)—we did not observe statistically significant differences in the hazard of loss of virologic suppression between third-drug classes, using NNRTI-based regimens as the reference. Although failure proportions differed at 6 months (lower in the INSTI group), these differences were not clearly sustained at 12 or 24 months, and hazard ratio estimates remained imprecise with confidence intervals that included the null.
A consistent finding was the association between longer time from HIV diagnosis to ART initiation and higher risk of loss of virologic suppression. Even though the per-day effect size was small, the direction and statistical support are consistent with the rationale for rapid initiation strategies and with guideline recommendations to minimize delays between diagnosis and ART start. In practice, this result suggests that programmatic actions that shorten linkage-to-treatment intervals could contribute meaningfully to sustained virologic control at the population level.
While randomized clinical trials have consistently demonstrated the pharmacological superiority and higher genetic barrier of INSTI-based regimens compared with NNRTI- and PI-based protocols,56,57 our real-world findings did not replicate this clear advantage. This discrepancy suggests that in our setting, structural and administrative barriers—manifested as frequent treatment interruptions and administrative delays—may create a “ceiling effect” on therapeutic effectiveness. When continuity of care is disrupted by health system factors, the intrinsic pharmacologic advantages of a specific molecule may be negated by the lack of consistent drug delivery. This phenomenon is consistent with observational literature, where class-level differences often attenuate because treatment outcomes are strongly shaped by adherence, continuity of access, and health system constraints rather than efficacy alone.58–61 Furthermore, the absence of a statistically significant advantage for INSTI-based regimens in our cohort likely reflects the combined influence of this “ceiling effect,” the limited statistical power due to the small INSTI sample size, and the mixed composition of the INSTI group (predominantly dolutegravir but also raltegravir and EVG/c).
Treatment interruption emerged as a key correlate of virologic failure in this cohort. Participants with loss of virologic suppression had a markedly higher frequency of documented interruption compared with those who maintained suppression. Because interruption was assessed during follow-up and may lie on the causal pathway between regimen choice, access/adherence, and virologic outcomes, we did not treat it as a baseline confounder in the primary effect estimation; rather, we examined it descriptively and in complementary analyses. Notably, many documented interruptions were related to administrative and logistical barriers (eg, drug delivery delays and lapses in insurance coverage), underscoring the importance of health system factors and continuity of access. These findings emphasize that optimizing virologic outcomes requires not only selecting effective regimens but also ensuring uninterrupted treatment delivery and timely follow-up.
Regarding secondary outcomes, among participants who did not change third-drug class, CD4+ T-cell counts increased over 24 months across all groups, with clinically meaningful gains and no clear class-specific differences. Body weight also increased progressively in all 3 groups, with larger relative gains among those underweight at baseline and smaller gains among those with overweight or obesity, a pattern consistent with weight recovery after ART initiation rather than disproportionate weight gain attributable to a specific class. These findings should be interpreted cautiously, as the study was not designed as a dedicated safety analysis of metabolic or cardiometabolic outcomes.
Strengths
This study leveraged data from 9 specialized HIV care centers covering multiple regions of Colombia and applied prespecified causal inference strategies (DAG-informed adjustment and IPTW with weight truncation and balance assessment) to reduce confounding when estimating the association between third-drug class and virologic outcomes. Missing covariate data were addressed using multiple imputation, and sensitivity analyses yielded similar results.
Limitations
Several limitations should be considered. First and foremost, the study reflects prescribing patterns before the full implementation of the 2021 Colombian guideline, resulting in an extreme imbalance in group sizes with low INSTI use (8%). This limited the statistical power for the INSTI versus NNRTI comparison, yielding wide confidence intervals that suggest an “absence of evidence” of a difference rather than “evidence of absence.” Second, the definition of loss of virologic suppression was based on a single viral load measurement ≥ 200 copies/mL. We lacked the data to differentiate between persistent virologic failure and isolated viral “blips” (transient elevations), which may have led to an overestimation of the virologic failure rate. Third, adherence was not systematically measured with validated instruments; documented ART interruption likely captures only a subset of adherence-related problems and may underestimate intermittent nonadherence. Fourth, as a historical cohort using routine clinical records, the study is susceptible to information bias and incomplete measurement for clinical and social determinants. Fifth, despite DAG-guided adjustment and IPTW, residual confounding remains possible, particularly from unmeasured comorbidities, transmitted resistance (baseline genotyping is not routinely performed prior to ART initiation in Colombia), concomitant medications, psychosocial conditions, and structural determinants not captured in charts. Finally, INSTI estimates reflect an average across several INSTI agents used during 2016 to 2020, limiting direct extrapolation to settings where contemporary first-line therapy is predominantly DTG- or bictegravir-based.
Implications
Taken together, our results suggest that under routine clinical conditions in Colombia during 2016 to 2020, differences in loss of virologic suppression across third-drug classes were not large or consistently detectable, while programmatic factors—particularly timeliness of ART initiation and treatment continuity—were strongly related to virologic outcomes. These findings support the view that the benefits of contemporary regimens can be maximized when health systems reduce delays to ART initiation and prevent interruptions driven by administrative or logistical barriers. Future studies should incorporate standardized adherence measures, longer follow-up, and broader outcomes (including non-AIDS events), and should further evaluate the role of social and structural determinants in the comparative effectiveness of ART in middle-income settings.
To overcome these barriers, implementation strategies adapted to the local context are essential. These include the adoption of “Rapid Start” protocols to minimize the time between diagnosis and treatment, and differentiated service delivery models (eg, multi-month dispensing) to reduce the administrative burden on patients and prevent interruptions.
Conclusions
In this cohort of individuals aged ≥14 years who initiated first-line ART in routine care, we did not observe statistically significant differences in time to loss of virologic suppression between NNRTI-, PI-, and INSTI-based regimens in conventionally adjusted or IPTW-weighted Cox models. However, strict interpretation is limited by the low frequency of INSTI use (8%) in this transition period, which resulted in low statistical power and imprecise estimates that preclude ruling out clinically relevant differences. Conversely, a shorter interval between HIV diagnosis and ART initiation and uninterrupted treatment access emerged as the strongest predictors of sustained virologic suppression. These findings suggest that structural barriers to continuity may create a “ceiling effect” on the real-world effectiveness of newer regimens in this setting. Therefore, optimizing outcomes requires not only effective drugs but also programmatic strategies—such as Rapid Start protocols and differentiated service delivery—to ensure rapid initiation and prevent administrative interruptions.
Supplemental Material
sj-docx-1-jia-10.1177_23259582261436966 - Supplemental material for Initial Antiretroviral Third-Drug Class and Time to Loss of Virologic Suppression: A Real-World Colombian Cohort Study
Supplemental material, sj-docx-1-jia-10.1177_23259582261436966 for Initial Antiretroviral Third-Drug Class and Time to Loss of Virologic Suppression: A Real-World Colombian Cohort Study by Juan Carlos Alzate-Ángel, Otto Sussmann, Javier Andrade, Beatriz Hernández, Héctor Mueses-Marín, Ximena Galindo, Sandra Liliana Valderrama-Beltrán, William Lenis, Leonardo Montero, Mónica Mantilla, Leonardo Arévalo, Diana Alzamora, Eric Delgado, Martha García, Ernesto Martínez-Buitrago and in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Acknowledgments
We thank the participating VIHCOL sites and clinical teams for their contributions to data collection and patient care. We also acknowledge the people living with HIV whose clinical data made this study possible.
ORCID iDs
Ethical Considerations (Human Welfare Statement)
This study was conducted using de-identified clinical data, with no variables that could directly identify individual participants. The study protocol was reviewed and approved by the Research Ethics Committees of the Corporación para Investigaciones Biológicas (CIB) (Approval Letter dated October 6, 2020) and IPS Virrey Solís (Minutes No. 4, dated May 24, 2023). The study complied with national and international ethical standards for research involving human participants.
Consent to Participate
Informed consent was waived by the reviewing Ethics Committees because this study involved the retrospective analysis of de-identified secondary data collected for routine clinical care and presented no risk to participants. This waiver is in accordance with Colombian Law 1581 of 2012 (Data Protection Law), specifically Articles 6 and 10, which authorize the processing of sensitive data for scientific and statistical purposes without patient authorization when the identity of the data subjects is suppressed.
Consent for Publication
Not applicable.
Author Contributions
All authors contributed to data registration, manuscript drafting, and approval of the final version. J. C. Alzate-Ángel and E. Martínez-Buitrago additionally contributed to the study protocol design. J. C. Alzate-Ángel additionally performed the statistical analyses.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported with internal funds from the Valle del Cauca HIV/AIDS Network (Red de VIH/SIDA del Valle del Cauca, Reviva), which supports the VIHCOL research group. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
De-identified participant data are not publicly available due to legal considerations. Access may be granted upon reasonable request to the corresponding author and/or VIHCOL governance body, subject to ethics approval and data-sharing agreements.
Supplemental Material
Supplemental material for this article is available online.
