Abstract
Purpose of the research
To compare incident hypertension rates across antiretroviral therapy regimens among adults with HIV in routine clinical care in the US-based OPERA cohort.
Major findings
The hypertension incidence rate was 4.16 per 100 person-years (95% CI: 3.75, 4.61) among 3220 ART-naïve individuals, and 4.60 (95% CI: 3.99, 5.30) among 1442 ART-experienced individuals. There was no statistically significant difference in incident hypertension between regimens at the class-level (integrase inhibitor with/without tenofovir alafenamide (TAF), boosted protease inhibitor, and non-nucleoside reverse transcriptase inhibitor) nor the drug-level (dolutegravir three-drug regimen with/without TAF, dolutegravir/lamivudine, bictegravir/emtricitabine/TAF, and boosted darunavir with TAF three-drug regimen).
Conclusions
In this large cohort representing routine clinical care in the USA, classes of antiretrovirals and specific agent combinations with or without TAF were not statistically significantly associated with incident hypertension. Other factors are likely to be more important drivers of hypertension than antiretroviral therapy selection in people with HIV.
Plain Language Summary Title
Risk of hypertension with different HIV treatment regimens
Plain Language Summary
Background
There is no consensus on the role of different HIV treatment options in the development of high blood pressure or hypertension in people with HIV. Some studies suggest that people using medications in the integrase strand transfer inhibitors (INSTI) class, as well as those using tenofovir alafenamide (TAF), could have a higher risk of developing hypertension compared to people taking other types of HIV treatment. However, other studies found no relationship between specific HIV treatments and the risk of hypertension. This study compares the development of hypertension in people with HIV taking different types of HIV treatment.
Methods
Data from electronic health records from the OPERA cohort study were used. This study included adults with HIV starting a new HIV treatment between January 1, 2016 and December 31, 2022 if they had no evidence of hypertension at treatment start. We assessed and compared the rate at which individuals developed hypertension based on the type of treatment used. Analyses were conducted separately for people who started HIV treatment for the first time (ART-naïve), and those who switched from another HIV treatment (ART-experienced).
Results
Among 3220 ART-naïve individuals, the rate of hypertension was 4.16 cases per 100 person-years (95% confidence interval (CI): 3.75, 4.61). Among 1442 ART-experienced individuals, the rate of hypertension was 4.60 cases per 100 person-years (95% CI: 3.99, 5.30). There was no difference in the rate of hypertension between the different treatments investigated.
Conclusions
In this large cohort representing routine clinical care in the USA, there was no association between different HIV treatment options, including the INSTI class and TAF use, and development of hypertension. Other factors are likely to be more important drivers of hypertension in people with HIV.
Keywords
Introduction
Elevated blood pressure (BP) is associated with an increased risk of cardiovascular disease, chronic kidney disease, and mortality.1–3 The 2017 American College of Cardiology/American Heart Association (ACC/AHA) Task Force on Clinical Practice Guidelines 4 defines normal BP as a systolic BP (SBP) < 120 mm Hg and a diastolic BP (DBP) < 80 mm Hg, and stage 1–2 hypertension (HTN) as a SPB ≥ 130 mm Hg or a DBP ≥ 80 mm Hg. Hypertension is a common health concern in the USA, where the overall prevalence was estimated at 46% between 2011 and 2014. 4
While HTN is a common comorbidity in general, it may be an even greater concern for people with HIV, as people with HIV may have a higher likelihood of developing HTN compared to people without HIV. A meta-analysis of cross-sectional studies estimated that the risk of HTN was 12% higher among people with HIV than those without HIV in North America (risk ratio: 1.12; 95% CI: 1.02, 1.23). 5 Similarly, in the USA, people with HIV have a higher prevalence of HTN (66%) compared to those without HIV (54%). 6 HIV infection is believed to activate the renin-angiotensin-aldosterone system (RAAS), which may lead to HTN. 7 The prevalence of HTN may also be higher in individuals taking antiretroviral therapy (ART) than in ART-naïve individuals. 8 While the exact mechanism has not been identified, it has been proposed that HIV viral proteins and antiretroviral therapy (ART) may be interacting with the immune system, inducing kidney damage, vascular dysfunction and alterations in sympathetic nervous outflow, which may lead to HTN. 9
While older antiretroviral (ARV) agents such as nevirapine, indinavir, lopinavir/ritonavir, and stavudine have been associated with HTN, 10 evidence on the association between more modern ARVs and elevated BP or incident HTN is mixed. Notably, there have been some reports of an increased risk of incident HTN with integrase strand transfer inhibitors (INSTI) and with tenofovir alafenamide (TAF), alone or in combination with each other.11–15 However, other studies reported no association between INSTI use or TAF and incident HTN.16–19 While many studies have evaluated the role of ARV classes on incident HTN, others have compared specific ARV agents. Some found that dolutegravir (DTG) was associated with larger increases in BP or with a higher risk of HTN than efavirenz20–22 or atazanavir 20 although other studies showed no association compared to efavirenz,19,23 other ARV combinations (efavirenz or darunavir/ritonavir [DRV/r]),24,25 or protease inhibitors. 26
Given the lack of consensus regarding the impact of modern ART regimens on HTN in the literature, this study aimed to evaluate the association between common ARV classes or agents and the risk of developing incident hypertension in both ART-naïve and ART-experienced people with HIV across the USA in the Observational Pharmaco-Epidemiology Research & Analysis (OPERA) cohort.
Methods
Study Population
The study population was derived from the OPERA cohort, an electronic health records (EHR) database of prospectively collected health information from routine clinical care. At the time of the study, the OPERA cohort included data from 147,423 people with HIV from 103 clinics across 22 states and 1 US territory. All individuals meeting the eligibility criteria between 01JAN2016 and 31DEC2022 (inclusion period) were included, with follow-up through 31DEC2023.
From the OPERA cohort, all people with HIV aged 18 years or older were selected if they initiated ART or switched to a regimen of interest between 01JAN2016 and 31DEC2022. The regimens included consisted of (a) INSTI without TAF 3-drug regimen (3DR), (b) DTG/lamivudine (3TC) 2-drug regimen (2DR), (c) INSTI with TAF 3DR, (d) boosted protease inhibitor (bPI) without TAF 3DR, (e) bPI with TAF 3DR, (f) non-nucleoside reverse transcriptase inhibitor (NNRTI) without TAF 3DR, or (g) NNRTI with TAF 3DR. ART experienced individuals switching to a regimen of interest were included regardless of the composition of the regimen from which they switched. The availability of ≥2 BP measurement taken during follow-up was required for inclusion in the study. Finally, individuals with evidence of prevalent hypertension (ie, prescription for antihypertensives or HTN diagnosis) as well as those with elevated BP at baseline (ie, last SBP ≥ 120 to <140 mm Hg and last DBP ≥ 80 to <90 mm Hg 4 ) were excluded from the main analysis. Individuals with elevated baseline BP were, however, included in a sensitivity analysis.
All individuals included in the study population were followed from the start of the regimen of interest until the first of (a) starting or stopping a core agent and/or TAF, (b) >45 days without ART, (c) loss to follow-up, defined as 12 months without clinical contact, (d) death, or (e) study end (31DEC2023).
Measurements
Incident HTN was defined as the first occurrence of (a) blood pressure measurements consistent with stage 2 HTN, (b) a diagnosis of HTN, or (c) a prescription for an antihypertensive medication. In accordance with ACC/AHA guidelines, 4 two consecutive measurements of SBP ≥ 140 mm Hg or DBP ≥ 90 mm Hg, measured on separate days, were considered as consistent with stage 2 HTN. For HTN events identified by BP measurements, the date of incident HTN corresponded to the date of the second qualifying BP measurement. Diagnosis of hypertension (stage 1 or greater) were identified from ICD codes and diagnosis titles. Antihypertensive medications consisted of diuretics, beta-blockers, angiotensin-converting enzyme inhibitors, angiotensin II receptor blockers, calcium channel blockers, alpha blockers, central alpha-2 receptor agonists and other centrally acting drugs, combined alpha and beta-blockers, and vasodilators.
ART regimens of interest were categorized by anchor agent class and use of TAF for the class-level analyses. Specifically, regimens were classified as INSTI without TAF, INSTI with TAF, bPI with or without TAF, and NNRTI with or without TAF. In the main analysis, bPI- and NNRTI-based regimens were not stratified by TAF use due to the small number of individuals who did not use TAF in combination with a bPI or NNRTI, resulting in too few HTN events. Both bPI- and NNRTI-based regimens were however stratified by TAF used in the sensitivity analysis, given the larger sample size.
ART regimens of interest were also categorized by specific anchor agent and use of TAF for the drug-level analyses. Specifically, regimens were classified as DTG without TAF 3DR, DTG/3TC 2DR, DTG with TAF 3DR, BIC/emtricitabine (FTC)/TAF and boosted darunavir (bDRV) with TAF 3DR. Individuals on other ART combinations were also excluded as those specific combinations were not the focus for the analysis. Those on bDRV without TAF 3DR were excluded from the main analysis due to the small number of events, but were included in the sensitivity analysis.
Statistical Analysis
All analyses were stratified by ART experience at the time of start of the regimen of interest (ie, ART-naïve or ART-experienced) and repeated for the main analyses (individuals with normal blood pressure) and the sensitivity analyses (individuals with normal/elevated blood pressure). Univariate Poisson regression was used to obtain the incidence rate (IR) and 95% confidence interval (CI) of HTN, stratified by either class-level or drug-level ART regimen. Multivariate Poisson regression (complete case analysis) was used to obtain adjusted incidence rate ratios (IRR) for the association between class-level ART regimen (reference: INSTI without TAF) or drug-level ART regimen (reference: DTG without TAF 3DR) and incident HTN. Models were adjusted for age (quadratic), female sex, Black race, viral load (ART-naïve: quadratic; ART-experienced: ≥50 copies/mL), diabetes, estimated glomerular filtration rate (eGFR) calculated with the CKD-EPI 2009 equation (quadratic), SBP (quadratic), and time-updated body mass index (BMI; quadratic).
The reporting of this study conforms to the STROBE statement (Supplementary Table). 27
Results
Study Population
The application of inclusion criteria is detailed in the Supplementary Figure. Out of 10814 ART-naïve individuals included, 3242 (30%) had prevalent HTN. The remaining 7572 (70%) individuals had normal/elevated BP but no HTN and were included in the sensitivity analysis population. Of those, 3220 (43%) had normal blood pressure and were included in the main analysis population (Figure 1A).

Distribution of blood pressure in ART-naïve and ART-experienced people with HIV in OPERA.
Out of 6543 ART-experienced individuals included, 3115 (48%) had prevalent HTN. The remaining 1986 (52%) had normal/elevated blood pressure but no hypertension, and were included in the sensitivity analysis population. Of those, 1442 (42%) had normal blood pressure and were included in the main analysis population (Figure 1B).
Baseline characteristics of the ART-naïve and ART-experienced populations with normal BP (main analysis population) are presented in Table 1. Both groups included a majority of men (ART-naïve: 84%; ART-experienced: 78%), with an eGFR ≥ 90 mL/min/1.73 m2 (ART-naïve: 84%; ART-experienced: 63%), and a high prevalence of overweight or obesity (ART-naïve: 32%; ART-experienced: 45%). The ART-naïve group was younger, had a better kidney function, were less likely to be women, overweight or obese, and included more Black people than the ART-experienced group. Most ART-experienced individuals switched from a regimen that included only one core agent, most frequently from an NNRTI- or a PI-based regimen.
Baseline Characteristics of Individuals with Normal BP and No Prevalent HTN.
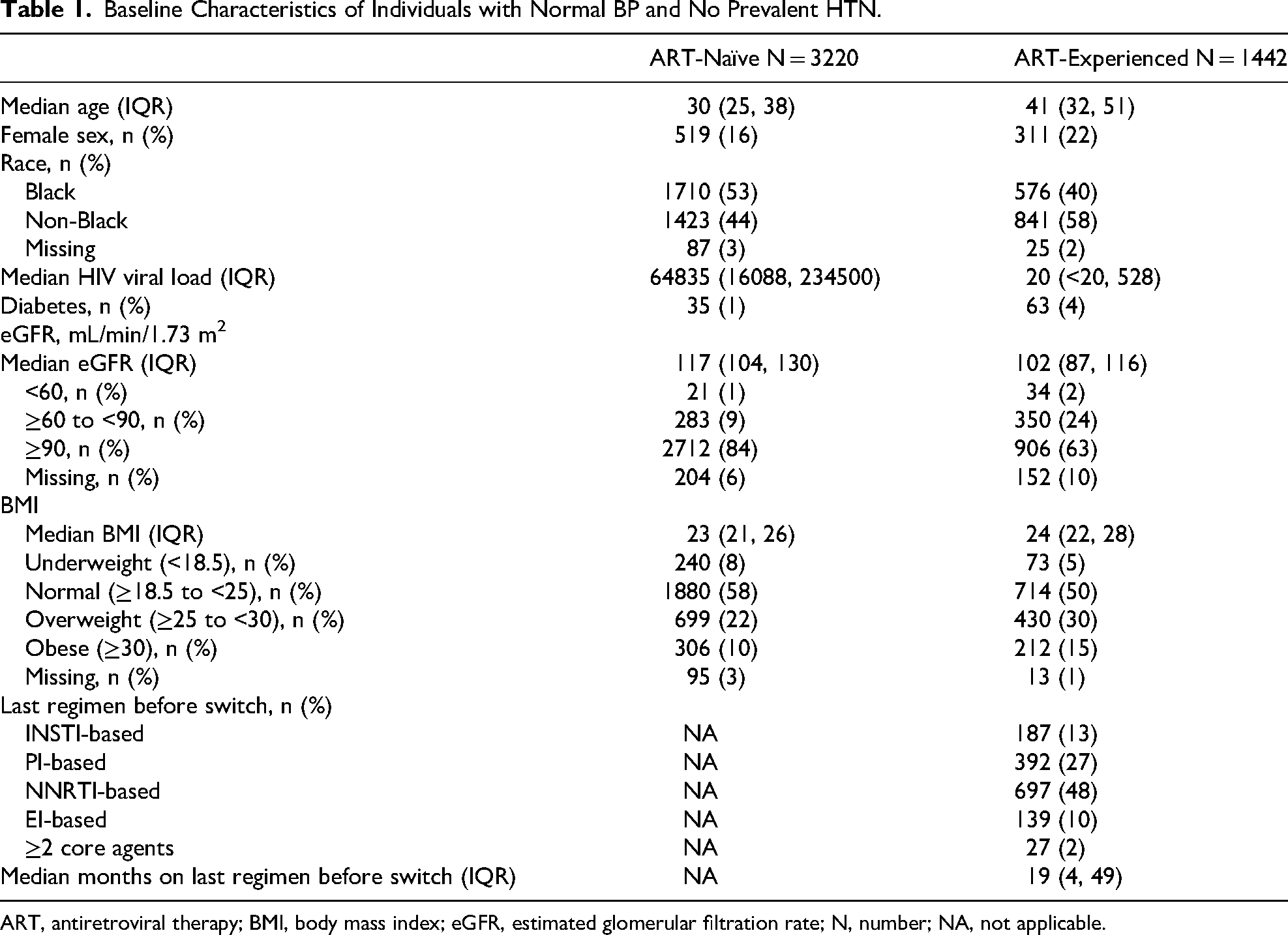
ART, antiretroviral therapy; BMI, body mass index; eGFR, estimated glomerular filtration rate; N, number; NA, not applicable.
Incident Hypertension Among Individuals With Normal BP and No Prevalent HTN
ART-naïve individuals with normal BP were followed for a median of 30 months and a total of 8538.5 person-years. Overall, the IR of HTN was of 4.16 per 100 person-years (95% CI: 3.75, 4.61). ART-experienced individuals with normal BP were followed for a median of 33 months and a total of 4155.1 person-years. The overall IR of HTN was of 4.60 per 100 person-years (95% CI: 3.99, 5.30). In both the ART-naïve and ART-experienced groups, over two thirds of incident HTN cases were identified through a prescription for an antihypertensive medication, and approximately half the cases had two consecutive BP measurements indicative of HTN (Table 2). Of note, incident HTN was identified solely based on an antihypertensive prescription (without a diagnosis or two consecutive qualifying BP) in 33% of ART-naïve and 41% of ART-experienced individuals with incident HTN.
Overall Incident Hypertension among Individuals with Normal BP and No Prevalent HTN.

ART, antiretroviral therapy; CI, confidence interval; DBP, diastolic blood pressure; IQR, interquartile range; IR, incidence rate; N, number; SBP, systolic blood pressure.
Criteria for incident hypertension are not mutually exclusive.
When comparing rates of HTN across regimens, there was no statistically significant difference between regimens at either the class-level or the drug-level among individuals with normal BP at regimen start (Figure 2). However, the rate of HTN was numerically higher with bPI regimens compared to INSTI without TAF for both the ART-naïve and ART-experienced groups. In addition, compared to DTG without TAF 3DR, the only regimen with consistently numerically higher HTN rates in the ART-naïve and ART-experienced groups was bDRV/TAF 3DR.

Association between ART regimen and incident HTN among individuals with normal BP and no prevalent HTN.
Incident Hypertension Among Individuals With Normal/Elevated BP and No Prevalent HTN
ART-naïve individuals with a normal/elevated BP were followed for a median of 30 months (IQR: 19, 50) and a total of 18962.4 person-years of follow-up. Overall, the incidence rate (IR) of HTN was 7.9 per 100 person-years (95% CI: 7.5, 8.3). ART-experienced individuals with a normal/elevated BP were followed for a median of 34 months (IQR: 19, 57) and a total of 9086.4 person-years of follow-up. Overall, the IR of HTN was 8.8 per 100 person-years (95% CI: 8.2, 9.4). No statistically significant difference was observed in the rate of HTN by core class or core agent/TAF combination in either the ART-naïve or the ART experienced groups (Figure 3).

Association between ART regimen and incident HTN among individuals with normal/elevated BP and no prevalent HTN.
Discussion
In this large, real-world cohort of people with HIV in the USA, the prevalence and IR of HTN was high, but there was no statistically significant association between ART regimens and incident HTN at the class- or drug-level. The high HTN prevalence of 30% in ART-naïve and 48% in ART-experienced individuals in OPERA was comparable to an estimated 28% HTN prevalence in a meta-analysis of studies including people with HIV in North America and in Western and Central Europe between 2007 and 2018, 8 and to a prevalence of 46% in ART-experienced individuals from a single clinic in Italy. 17 However, another US study using EHR data reported a much higher prevalence of HTN among people with HIV, estimated at 66% in 2022. 6 The higher prevalence in the latter may be due to the lower BP cut-off used (SBP ≥ 130 mm Hg, DBP ≥ 80 mm Hg) compared to this OPERA study (SBP ≥ 140 mm Hg, DBP ≥ 90 mm Hg).
Overall, incident HTN was observed in 11% of ART-naïve and 13% of ART-experienced individuals in OPERA, with IR per 100 person-years of 4.16 (95% CI: 3.75, 4.61) and 4.60 (3.99, 5.30), respectively. The proportion of people with HIV developing incident HTN in OPERA was comparable to the lower range from other studies (7% to 31%).11,13,18,19,24,26,28 Similarly, the IR of HTN among people with HIV was comparable to the lower range estimated in other studies (2.6 to 43.9 per 100 person-years).11,13,26,29 In the NA-ACCORD cohort, the IR was estimated at 2.6 cases per 100 person-years between 2000 and 2013 in North America. 29 In the RESPOND consortium of HIV cohorts from Europe and Australia, the IR ranged from 7.4 per 100 person-years (95% CI: 7.2, 7.7) 13 to 12.6 per 100 person-years (95% CI: 11.9, 13.4). 11 The IR for HTN was much higher (37.4 to 43.9 per 100 person-years) in the NEAT trial, which included virologically suppressed individuals aged ≥ 50 years or with a >10% 10-years Framingham CVD risk score from six European countries. 26 Differences in study populations, design, and outcome definitions likely explain this wide range of estimates. As expected, HTN incidence rates increased when individuals with elevated baseline BP were added to the OPERA study population for the sensitivity analysis.
In OPERA, no statistically significant association between regimen and incident HTN was observed, although there were some inconsistencies between ART-naïve and ART-experienced groups with regards to the direction of the point estimates. However, given that all confidence intervals crossed the null, these variations cannot be interpreted with certainty. A sensitivity analysis adjusting for baseline BMI instead of time-updated BMI yielded nearly identical results. Most clinical trials have reported no difference in the risk of HTN by regimen.18,19,24–26 As for observational studies, in a cohort of 1408 ART-experienced individuals in Italy, history of INSTI use (n = 634) was not associated with an increased risk of HTN compared to history of NNRTI or PI use (n = 774), with an adjusted odds ratio (OR) of 1.00 (95% CI: 0.98, 1.01). 17 It is thus possible that factors other than ARVs contribute to the risk of HTN in people with HIV. For example, lifestyle factors (eg, smoking, diet, exercise), family history, weight, comorbidities, or past use of certain older ARVs, as well as RAAS activation by the HIV infection itself may be more important drivers of HTN. However, two analyses of clinical trial data reported an increased risk of HTN in individuals randomized to DTG compared to EFV in regions where HTN was not routinely treated, 23 as well as greater increases in SBP and DBP in individuals randomized to DTG + DRV/r compared to DRV/r + 2 nucleoside reverse-transcriptase inhibitors. 28 Moreover, several cohort studies observed a higher likelihood of HTN with INSTIs and TAF than other regimens. In an analysis of 5162 individuals with overall low to moderate traditional atherosclerotic cardiovascular disease (ASCVD) risk in the REPRIEVE cohort, INSTI use was associated with statistically higher odds of HTN: a propensity-weighted hazard ratio of 1.38 (95% CI: 1.09, 1.70) was estimated for INSTI-base regimens (n = 2708) compared to non-INSTI-based regimens (n = 2454). 15 Of note, this was an update of an earlier analysis (N = 4500) in which no statistical difference was observed, although confidence intervals were close to reaching statistical significance. 16 The RESPOND consortium of HIV cohorts 13 reported that among 9704 people with HIV, the incidence of HTN was higher with regimens containing INSTI (5701 person-years; IRR: 1.41; 95% CI: 1.30, 1.53), TAF (2802 person-years; IRR: 1.22; 95% CI: 1.05, 1.42), or both (12515 person-years; IRR: 1.70; 95% CI: 1.54, 1.88) compared to regimens containing neither INSTI nor TAF (18976 person-years). Adjusting for BMI attenuated the association between INSTIs and HTN, although it remained statistically significant. 13 In a South African cohort study, 794 individuals switching from EFV to DTG had a 14% (95% CI: 11, 18) increased risk of incident hypertension compared to 794 propensity score-matched individuals remaining on EFV. 21 Of note, TAF has also been associated with an increased risk of HTN compared to tenofovir disoproxil fumarate (TDF) among people without HIV on pre-exposure prophylaxis (PrEP). 14 Differences in study designs, outcome definitions and study populations may have impacted the observed associations between ART exposure and incident HTN, resulting in conflicting conclusions.
Strengths of this study include the use of data derived from the real-world OPERA cohort, which contains all routine clinical data for the care received at OPERA-participating clinics. Statistical adjustment for potential confounders included time-updated BMI, which was shown to be an important factor to control for when estimating the association between ART and HTN. 13 This was a large study including a total of 4662 people with HIV without prevalent HTN and with normal BP. This allowed for stratification by prior ART experience, as well as the use of multiple categories of exposure, providing information on the risk of HTN for the most common types of regimen combinations currently in use in the USA. A sensitivity analysis was performed to assess the robustness of the results if individuals with elevated BP were included, thus increasing the sample size further.
However, despite the large study population, the study was limited by a small number of events in some regimens of interest. Therefore, some regimen categories were collapsed (NNRTI with/without TAF, bPI with/without TAF) or excluded from analysis (bDRV/no TAF). A complete case analysis was conducted, excluding 8–14% of the population with missing covariates. No information on lifestyle factors affecting HTN, such as diet, exercise, or family history, was available and therefore could not be controlled in the statistical analysis. While some smoking information was available in the EHR, it was often incomplete or missing, preventing its inclusion in the statistical models as up to a quarter of the population would have been excluded. Residual confounding may be present. Finally, misclassification of incident HTN cannot be excluded. Indeed, only BP consistent with stage 2 HTN (two consecutive measurements of SBP ≥ 140 mm Hg or DBP ≥ 90 mm Hg) was included in the outcome definition. However, the ACC/AHA guidelines consider that a SPB ≥ 130 mm Hg or a DBP ≥ 80 mm Hg is indicative of stage 1–2 HTN. 4 It is therefore possible that the incidence of HTN was underestimated due to the exclusion of stage 1 HTN. However, a large proportion of HTN cases were identified through an antihypertensive prescription only and did not have two consecutive BP indicative of HTN or a diagnosis of HTN at that time. Because some antihypertensive medications can be used to treat other conditions, this could have resulted in an overestimation of HTN incidence. Given that this study was derived from EHR data collected through routine clinical care, the study size was determined by the total number of individuals meeting the eligibility criteria.
Conclusion
In this large cohort representative of routine clinical care in the USA, prevalent HTN was common among both ART-naïve and ART-experienced people with HIV. However, classes of antiretrovirals and specific agent combinations with or without TAF did not appear to be statistically significantly associated with incident HTN. Other factors are likely to be more important drivers of HTN than antiretroviral therapy selection in people with HIV.
Supplemental Material
sj-jpg-1-jia-10.1177_23259582261445148 - Supplemental material for Incident Hypertension with Antiretroviral Therapy in the OPERA Cohort
Supplemental material, sj-jpg-1-jia-10.1177_23259582261445148 for Incident Hypertension with Antiretroviral Therapy in the OPERA Cohort by Gerald Pierone Jr, Laurence Brunet, Jennifer S Fusco, Philip C Lackey, Michael B Wohlfeiler, Douglas T Dieterich, Cassidy Henegar, Vani Vannappagari, Bryn Jones, Annemiek de Ruiter and Gregory P Fusco in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Supplemental Material
sj-pdf-2-jia-10.1177_23259582261445148 - Supplemental material for Incident Hypertension with Antiretroviral Therapy in the OPERA Cohort
Supplemental material, sj-pdf-2-jia-10.1177_23259582261445148 for Incident Hypertension with Antiretroviral Therapy in the OPERA Cohort by Gerald Pierone Jr, Laurence Brunet, Jennifer S Fusco, Philip C Lackey, Michael B Wohlfeiler, Douglas T Dieterich, Cassidy Henegar, Vani Vannappagari, Bryn Jones, Annemiek de Ruiter and Gregory P Fusco in Journal of the International Association of Providers of AIDS Care (JIAPAC)
Footnotes
Acknowledgments
This research would not be possible without the generosity of people living with HIV and their OPERA caregivers. Additionally, we are grateful for the following individuals: Michael Stagner (SAS programming), Kelly Oh (QA), Bernie Stooks (data architecture), Lisa Lutzi & Nicole Shaw (Data management/quality), and Judy Johnson & Quateka Cochran (Clinical data categorization).
Ethical Approval and Informed Consent
The OPERA® observational database complies with all HIPAA, HITECH, and HITRUST requirements and has received annual institutional review board (IRB) approval by Advarra IRB (Pro00023648), including a waiver of informed consent and authorization for use of protected health information. All data are anonymized to ensure confidentiality of all participants.
Author Contributions
LB, JSF, CH, and VV share the responsibility for the design of this study. GP, JSF, PCL, MBW, DTD, and GPF contributed the acquisition of data. LB is responsible for all the analyses. All authors have contributed to the interpretation of results, have critically reviewed and approved the manuscript, and have participated sufficiently in the work to take public responsibility for its content.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by ViiV Healthcare.
Declaration of Conflicting Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GP has received research funding paid to his institution from GSK/ViiV Healthcare, NIH/DAIDS, Gilead Sciences and Precision for Medicine, and is a member of the Epidemiology and Clinical Advisory Board for Epividian. LB, JSF, and GPF are employed by Epividian, which has had research funded by AIDS Healthcare Foundation, EMD Serono, Gilead Sciences, Janssen, Merck & Co., Theratechnologies, and ViiV Healthcare. PCL is a member of the Epidemiology and Clinical Advisory Board for Epividian. MBW has participated in post-conference advisory boards for the Conference on Retroviruses and Opportunistic Infections (CROI) and International AIDS Conference (IAC). MBW is also a member of the Epidemiology and Clinical Advisory Board for Epividian. DTD has received personal fees for participation in speaker's bureaus and advisory boards from Abbvie, Merck, and Gilead Sciences, consulting for VIR, and advisory board participation for Epividian. CH, VV, BJ, and AdR are employed by ViiV Healthcare and hold shares in GSK as part of their employment.
Data Availability
The datasets used in this study are not publicly available due to privacy concerns and the proprietary nature of the database but can be accessed upon reasonable request through the corresponding author to the OPERA® Epidemiological and Clinical Advisory Board. Access to codes may be granted upon request with parties agreeing to privacy restrictions and technological specifications and requirements.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
