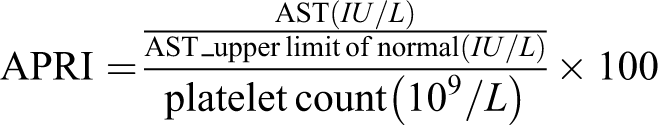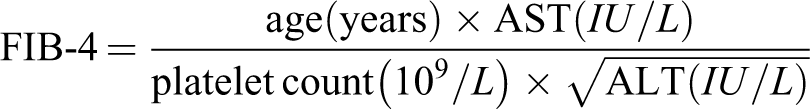Abstract
WHO recommends hepatitis C (HCV) screening for all people living with HIV (PLHIV). Yet, HCV coinfection was shown to be rare in some Sub-Saharan HIV cohorts, and targeted testing was suggested more efficient for such settings. We studied HCV prevalence among Ghanaian PLHIV, and assessed the external validity of a score to guide targeted testing. This score was initially derived from a Cambodian HIV cohort, and uses as predictors: age, household member/partner with liver disease, diabetes, generalized pruritus, AST, platelets, and AST-to-platelet ratio index. We enrolled 4,023 PLHIV, most from Greater Accra and Central regions, 28.4% were male, median age was 47 years, and high-risk behavior was reported to be rare. HCV seroprevalence was 0.57%, and HCV-RNA was detectable in 0.5%. Sequencing revealed genotype 1(b) and 2(q/r) infections. The discriminatory performance of the score was suboptimal in the Ghanaian setting. The area under the curve was 0.69 (95% CI 0.59-0.79). HCV coinfection prevalence was very low in this Ghanaian PLHIV cohort with reported low-risk of onward transmission. To avoid the cost of screening all PLHIV in similar cohorts in resource-constrained settings, further research to develop better tools/scores to guide targeted HCV testing is needed.
Keywords
Background
Worldwide, hepatitis C virus (HCV) infection is among the leading causes of chronic liver disease. An estimated 71 million people are chronically infected with hepatitis C. 1 This results currently in about 400,000 deaths each year, primarily due to liver cancer and cirrhosis caused by untreated infections. 2 Hepatitis C is found worldwide, but prevalence is highly unevenly distributed within and between different countries, depending on whether the epidemic is mainly driven by population subgroups with high-risk behavior, or by more widespread exposure (such as iatrogenic routes or poor infection control). 3 Low- and middle-income countries (LMIC) are estimated to account for about 75% of people living with HCV. 4
Until a few years ago, hepatitis C treatment was centered around the use of pegylated interferon and ribavirin, and was considered too complex and poorly successful for wide-scale use. However, with the advent of direct-acting antivirals, hepatitis C treatment has been revolutionized. Its high cure rates (>90%), near-excellent tolerability and ease of administration has generated interest in the global elimination of hepatitis C as a public health threat. 6 The new global HCV cascade targets established by the World Health Organization (WH0)—90% of infected diagnosed and 80% of diagnosed treated by 2030—reflect this paradigm shift. 7
What Do We Already Know about this Topic?
In a systematic review of 2016 the HCV/HIV coinfection prevalence in Ghana was estimated at 2.8% (95% CI 0.4-6%). 5 Resources were insufficient to offer HCV testing as part of the standard package of care.
How Does Your Research Contribute to the Field?
We found a very low HCV prevalence (0.5% viremic) in this large, primarily heterosexually infected HIV cohort. The low number of HCV/HIV coinfections did not allow to firmly conclude on the external validity of an earlier developed score for targeted screening.
What Are Your Research’s Implications toward Theory, Practice, or Policy?
Depending on local resources and epidemiology, offering targeted HCV screening to people living with HIV with low onward risk of HCV transmission might be a better (more efficient) choice. However, further research is needed to develop (and validate) better performing risk- and symptom-based scores for targeted HCV screening.
To allow timely scale up of treatment, efficient HCV testing strategies are crucial. By the end of 2017, only about 20% of those living with hepatitis C knew their status, with significantly lower proportions in LMIC. 8 WHO generally recommends to target HCV screening toward people with signs and symptoms of chronic liver disease, and toward population subgroups with known higher prevalence or risk profile, such as people who use/inject drugs (PWUD, PWID), men who have sex with men (MSM), and people living with HIV (PLHIV). However, if the prevalence in the general population exceeds 2 to 5%, universal screening, for the whole population, is recommended. 9
For many resource-constrained countries, it proves difficult to fully operationalize these HCV testing recommendations in the absence of specific donor funding. 10 To make best use of the available domestic resources for health, risk-based targeted HCV testing seems a more feasible option. However, countries often lack the national and sub-national epidemiological data to inform evidence-based decision making on who to prioritize for testing. 11
As for overall HCV epidemiology, prevalence of HCV coinfection among PLHIV varies widely and is highest in the subgroup of PWUD/PWID (seroprevalence of 82.4%), followed by MSM (6.4%), with much lower rates for those without specific risk behavior (2.4%). 12 For Sub-Saharan Africa, a recent study cumulating data from 5 countries reported even lower HCV coinfection prevalence (<1%), and suggested that in some contexts risk factor and symptom-based targeted HCV testing algorithms might be an appropriate choice for PLHIV. 13
We examined the prevalence of HCV coinfection among PLHIV in care in an HIV clinic in the Greater Accra region in Ghana. At the time of planning our study, a systematic review of HCV epidemiology in Ghana had estimated the seroprevalence of HCV/HIV coinfection at 2.8%, with a confidence interval ranging from 0.4 to 6%. 5 This estimate was based on small-sized studies, and on pre-selected subgroups of HIV cohorts. 14,15 More recent studies, among PLHIV from the Ashanti, Central, and Brong-Ahafo regions, reported seroprevalence estimates between 0.5 to 5.5%. 16 -18
In addition to contributing to a larger evidence-base on the prevalence of HCV antibody positivity and viraemia among Ghanaian PLHIV, we also aimed to externally evaluate the discriminatory performance of a clinical prediction score (CPS) for targeted HCV testing that we previously derived from a dataset of a Cambodian HIV cohort. 19 -21 This CPS aims to predict the risk of current hepatitis C infection, and to provide an easy-to-use tool to identify the sub-group of PLHIV who could be targeted for HCV diagnostic testing, if resources do not allow routine testing of all PLHIV.
Methods
Study Design and Setting
We conducted a single-center, cross-sectional study among adult PLHIV in care at the HIV clinic of the Fevers Unit of Korle Bu Teaching Hospital in Accra (Ghana). The hospital is the largest in Ghana with a bed capacity of 2,000 and a daily outpatient attendance of 1,500. The Fevers Unit houses the adult HIV clinic that serves as a national referral hub for PLHIV. The clinic does not target any specific subgroup of PLHIV. Key populations are expected to be a minority among those attending. Most did not yet know their HCV status at the time we started our study. Poor availability of hepatitis C treatment was the main reason for not offering HCV diagnostic testing as part of the routine care package. Availability of hepatitis C treatment is now slowly improving as direct-acting antivirals are becoming available, but still beyond reach for most patients.
Study Population
All adult PLHIV of the Fevers Unit cohort who reported for a routine follow-up consultation, and who were 1) on ART or yet to be initiated on ART, and 2) in regular HIV care follow-up, i.e. minimum of 2 HIV consultations in the last 6 months, and 3) never exposed to hepatitis C treatment, were offered participation in the study. They were enrolled in a consecutive manner, and without any other further selection until the sample size was reached. Enrolments took place between November 2018 and August 2019.
Background Information on the Clinical Prediction Score Derived from the Cambodian Dataset
Researchers from the Institute of Tropical Medicine Antwerp (also authors of this paper) and Sihanouk Hospital Center of Hope in Cambodia developed a CPS for targeted HCV screening based on a dataset of a large Cambodian HIV cohort. 20 They used the Spiegelhalter-Knill-Jones method to develop the predictive model. The score included older age (>50 years), having a household member/partner with liver disease, presence of diabetes, having generalized pruritus, high AST-to-platelet ratio index (APRI, ≥0.45), and low platelet count (<200 × 109 cells/L) as risk-increasing predictors; and low aspartate aminotransferase (AST, <30 IU/L) and low APRI (<0.45) as risk-lowering predictors. In the Cambodian derivation cohort, the score would have enabled identifying 85% of the coinfected while reducing the need for testing by 70%, and a score ≥0 fitted best as threshold-to-screen, with a negative predictive value of 99.2%.
Sample Size and Power
The primary objective of the study was to determine the HCV prevalence and to estimate the area under the ROC curve (AUROC) of the predictor score developed in Cambodia when it would be applied in the Ghanaian setting. The score had an AUROC of 0.84 in the Cambodian population. 20 A sample size of 4,000 participants was calculated initially to estimate the AUROC to within 7.5% (half-width of the 95% confidence interval), assuming a prevalence of current HCV coinfection (i.e. HCV RNA detectable) of 1.0% in Ghana. 15 The paper of King et al was the only published study to provide an estimate on current HCV infection in PLHIV in Ghana at the time of preparing the study protocol.
Subject-Related Procedures
After providing informed consent, enrolled PLHIV were—in addition to the usual procedures of an HIV follow-up consult—asked about 1) current socio-demographics (formal/informal profession, area of residence), 2) risk factors/behavior for HCV (injecting drug use, commercial sex work, sexual orientation, past blood transfusions, surgery, tattoos, partner/household member with liver disease), 3) clinical symptoms possibly related to liver disease (generalized pruritus, fatigue, myalgia/arthralgia, anorexia/weight loss), and 4) comorbid conditions (alcohol use, diabetes mellitus, tuberculosis, hepatitis B, hypertension). For alcohol use, we asked questions on the average number of days per week alcohol was consumed, and the type and number of drinks.
The data collection for the study was complemented by specific findings of the physical examination (clinical signs of hepatic decompensation) and retrieval of routinely collected data from the medical file such as date of enrolment in the HIV cohort, date of start on ART, baseline CD4, last HIV viral load, hepatitis B surface antigen (HBsAg) result, and current ART regimen.
This part of the study procedures covered also the systematic data collection on 4 of the predictors used in the Cambodian CPS (age, diabetes mellitus, partner/household member with liver diseases, generalized pruritus). All data were collected without prior knowledge on the HCV status of the patient.
At the end of the consultation, after hepatitis C pre-test counselling was done blood was drawn for HCV diagnostic testing (HCV antibody test, followed by HCV viral load testing if the HCV antibody test showed a positive or borderline result), liver function tests (ALT, AST) and full blood count. The above laboratory results were used to calculate the commonly used compound surrogates of the degree of liver fibrosis: AST-to-platelet ratio index (APRI) and Fibrosis-4 (FIB-4). Cut-offs of >1.5 for APRI, and >3.25 for FIB-4 are commonly used as being indicative of advanced fibrosis. 22 They are calculated as follows:
Laboratory Procedures
HCV antibody, biochemistry and full blood count analyses were done by a team of experienced laboratory technicians trained in Good Clinical Laboratory Practices (GCLP) at the Central Laboratory of Korle Bu Teaching Hospital. HCV viral load testing was performed at the University Hospital of Antwerp, Belgium. All analyses were performed according to the manufacturer’s instructions.
HCV antibody screening
The SD Bioline HCV test (Standard Diagnostics, Republic of Korea) was used to detect anti-HCV antibodies. It is an in vitro immunochromatographic rapid assay for the qualitative detection of antibodies specific to HCV. The test is listed on the WHO pre-qualification list for in-vitro diagnostics. 23 Serum samples were used in this study.
Biochemistry
Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels were determined using the VITROS ALT Slide method on the VITROS 5600 Integrated System (Ortho Clinical Diagnostics, Rochester, NY, USA).
Full blood count
Full Blood Count was performed on the SYSMEX Xi 2100 hematology automated analyzer (Sysmex Europe GmbH, Germany).
HCV viral load (quantitative PCR) and genotyping
The quantification of HCV RNA was determined using the COBAS TaqMan HCV Quantitative Test, v2.0 on the COBAS TaqMan 48 Analyzer from Roche, with lower limit of detection of 15 IU/mL. This method is approved by the Food and Drug Authority (USA). Genotyping was performed by NS5B sequencing at the Belgian National HCV Reference Centre (a consortium of the Institute of Public Health and Université Catholique de Louvain).
External quality control was performed on a subset of samples in the laboratories of the Institute of Tropical Medicine Antwerp and the University Hospital of Antwerp, Belgium. HCV antibody re-testing was done for 5% of the negative and 100% of the positive samples.
Data Collection
Routine patient files and laboratory result print-outs served as source data for the study. Standard templates were used for each of those. These source data and the study data, obtained during the consultation immediately following enrolment, were used to complete a study-specific paper case report form (CRF). On daily basis, data were transferred from the paper CRF into a password-protected Epidata database (v4.4.2.1).
Data Analysis
Statistical analyses were performed using Stata 15.1 (StataCorp LP, College Station, TX, USA). Descriptive statistics, using medians and interquartile ranges (IQRs) for continuous variables and percentages for categorical variables, were used to summarize the baseline characteristics of the study participants, by HCV coinfection status. The information on alcohol use was resumed for each participant in average grams of alcohol consumed per week. An average alcohol use of more than 140g/week was considered as moderate to heavy alcohol intake. 24
The HCV seroprevalence (i.e. proportion with an HCV-antibody positive result among all enrolled), and prevalence of current hepatitis C coinfection (i.e. proportion with a detectable HCV viral load among all enrolled) were determined and reported with Wilson 95% confidence intervals.
The Cambodian CPS, 20,21 containing clinical and laboratory factors with a specific weight (age over 50 years: +1, diabetes mellitus: +1, APRI <0.45: −1, APR ≥0.45: +1, AST <30 IU/L: −1, platelets <200 ×109 cells/L: +1, household/partner with liver disease: +1, generalized pruritus: +1), was applied in the Ghanaian cohort based on the data collected in the cross-sectional study. Summing the weighted predictor scores of the individual’s risk factors yielded the total predictor score for each study participant. Presence of current HCV infection (=HCV RNA detectable) was defined as the outcome.
The overall performance of the CPS was assessed by calculating the AUROC with 95% confidence intervals (CI).
Ethical Approval and Informed Consent
The study was approved in Belgium by the Institutional Review Board (IRB) of the Institute of Tropical Medicine of Antwerp (approval no. ITG 1216/18), the Ethical Committee of the Antwerp University Hospital (approval no. 18/20/249), and in Ghana by the IRB of Korle Bu Teaching Hospital (approval no. KBTH-IRB-00077/2018) and the IRB of Noguchi Memorial Institute for Medical Research (approval no. CPN 001/18-19). All participants provided written informed consent prior to enrolment in the study.
Results
Socio-Demographic and Clinical Characteristics of the Study Participants
Of the 4,023 participants enrolled, the majority resided in the Greater Accra region (82.3%, where the study was conducted and the national capital Accra is located), followed by the Central region (12.3%) and Eastern region (3.7%). Less than a third (28.4%) were male, and the median age of all enrolled was 47 years (IQR 41-55). Members of key population groups were few: only 6 participants identified as MSM; none reported past or current injecting drug use.
Most study participants (91.9%) were on first-line ART, the most used first-line regimens were tenofovir(TDF)/lamivudine(3TC)/efavirenz(EFV) (42.6%), zidovudine(AZT)/3TC/EFV (29.2%), AZT/3TC/nevirapine(NVP) (21.9%) and TDF/3TC/NVP (3.7%). As second-line ART regimens, TDF/3TC/lopinavir-ritonavir(LPVr) (52.6%), TDF/3TC/atazanavir-ritonavir(ATVr) (22.8%) were most frequently used. The median duration on ART was 7.4 years (IQR 3.7-10.8). Among those with available HIV viral load result less than 1 year before enrolment in the study (N = 2,235), 72.8% had an undetectable HIV viral load. Three-hundred nineteen participants (14.3%) had an HIV viral load above 1,000 copies/mL. Forty-two participants had an ongoing opportunistic infection at enrolment, mostly tuberculosis and/or oral/esophageal candidiasis. Of those with available HBsAg result as part of routine care, 13.6% (210/1548) had a reactive result. The median APRI of those with reactive HBsAg result was 0.36 (IQR 0.24-0.49). At enrolment, 6.8% of the study participants reported that they still consumed alcohol at least once per month, but only 5 participants consumed more than 140g/week (i.e. moderate to heavy alcohol use). Table 1 shows the details of the participant characteristics.
Characteristics of the Ghanaian HIV Cohort, by Coinfection Status.

Abbreviations: ART (antiretroviral treatment), VL (viral load), ALT (alanine aminotransferase), AST (aspartate aminotransferase), APRI (AST-to-platelet ratio index), FIB-4 (fibrosis-4), HBs Ag (hepatitis B surface antigen).
Missing values were less than 5% except for 1) HIV viral load results within 1 year of study enrolment, and 2) hepatitis B surface antigen.
† For HIV viral load, 1775 participants without HCV coinfection, and 11 with HCV coinfection have missing results.
‡For HBsAg, 2462 participants without HCV coinfection, and 14 with HCV coinfection have missing results.
HCV prevalence
Twenty-three of the 4,023 participants tested positive for anti-HCV antibodies yielding a seroprevalence of 0.57% (95% CI 0.38-0.86). Of these, 20 had a detectable HCV RNA viraemia, corresponding to a prevalence of current HCV infection of 0.5% (95% CI 0.32-0.77). Five out of the 20 HCV coinfected participants had an APRI >1.5 and/or FIB4 >3.25, and thus were likely suffering from advanced liver disease. The prevalence of current HCV coinfection was similar among men (0.7%) and women (0.4%) (p = 0.3). For 11 out of the 20 HCV coinfected participants genotyping results were obtained by NS5B sequencing, the genotype distribution was as follows: 1 (n = 4), 1b (n = 2), 2 q (n = 3) and 2r (n = 2).
Discriminatory Performance of the Cambodian Clinical Prediction Score in the Ghanaian HIV Cohort
The distribution of the individual predictors included in the Cambodian CPS is presented in Table 2.
Distribution of the Cambodian CPS Predictors in the Ghanaian Cohort.

Abbreviations: CPS (clinical prediction score), APRI (AST-to-platelet ratio index), AST (aspartate aminotransferase).
When applying the CPS, the total individual scores in the Ghanaian HIV cohort range from −2 to +4 (see Table 3). Only 36% of the PLHIV in the Ghanaian cohort had a total score ≥ 0 (i.e. at the cut-off that best fitted as threshold-to-screen in Cambodia), including 13 of the 20 HCV/HIV coinfected people. The AUROC of the CPS applied in the Ghanaian cohort was 0.69 (95% CI 0.59-0.79) (Figure 1) compared to 0.84 (95% CI 0.80-0.89) in the Cambodian derivation cohort.
Probability of HCV Coinfection by Clinical Prognostic Score.


ROC curve of the performance of the Cambodian derived prediction score in the Ghanaian validation cohort.
Discussion
In our study among PLHIV, predominantly from the Greater Accra and Central regions in Ghana, the HCV seroprevalence and prevalence of current HCV coinfection was 0.57% and 0.5% respectively. These estimates range the lower end of the confidence interval of the 2.8% (95% CI 0.4-6.0) pooled estimate for HCV/HIV coinfection prevalence obtained by a systematic review of Ghanaian data published up to end 2015. 5 When this systematic review was compiled, only 3 studies had reported data about PLHIV, of which one described coinfection data for less than 50 PLHIV as the study was not designed for that purpose. 25 The other 2 reported quite diverging results on larger sample sizes. King et al reported, similar to the estimate we found, a prevalence of current HCV coinfection of 1.0% (95% CI 0.0-1.9) among PLHIV from the Ashanti region. 15 In the other study, from Sagoe et al, the seroprevalence was higher (3.6%) and measured among ART-naïve PLHIV in 2007 at the Korle Bu Fevers Unit in Accra. 14 Possible explanations, for the difference in prevalence found in 2007 and our study (2018-2019) in the same HIV clinic, might be the different types/generations of antibody tests used (later generations being more specific), or excess mortality among HCV/HIV coinfected PLHIV, compared to PLHIV without HCV coinfection.
More recently published studies reported HCV coinfection data for PLHIV in care in the Central, Ashanti, and Brong-Ahafo regions, but none for the Greater Accra region. Our findings are similar to the seroprevalence estimate of 0.5% among PLHIV of any age from the Central region, 16 and the current HCV coinfection rate found by Geretti et al in the Ashanti region in a subgroup of HBsAg negative PLHIV. 26 The HCV/HIV coinfection seroprevalence estimates obtained in 2 other studies, 17,18 in the Brong-Ahafo and Ashanti region, are higher (4.1%-5.5%). In-country regional epidemiology differences, preselection of study participants, and use of differently performing diagnostic tests and algorithms may explain the broad range of results. Over-estimation of HCV prevalence due to high rates of false positive anti-HCV test results has been documented at several occasions. 27,28
It is important to bear in mind that our study participants were mostly “general population” and reporting a low-risk profile. Key populations, like PWID, MSM, and female sex workers are expected to have a higher HCV prevalence due to increased risk of exposure. 12,29 Underreporting of risk behavior might have been an issue in our study as restrictive policies and stigmatization might have influenced self-reporting. However, while according to a recent report (2019) levels of overall drug use are increasing in Ghana, levels of injecting drug use are considered to be still low in Ghana, 30 and the large majority of PLHIV in care in Ghana (about 125.000) are general population. 31 The Korle Bu HIV cohort profile is therefore not thought to be different, from the other large HIV cohorts in Ghana. 32
In sum, the main contribution of our study has been to provide more data on HCV/HIV coinfection prevalence in the Greater Accra and Central region for PLHIV without increased risk behavior. The large sample size without further selection of subgroups, the use of a WHO prequalified HCV rapid diagnostic test, 23 the availability of HCV RNA testing, and external quality control enabled to obtain robust and precise prevalence estimates.
Our findings suggest moreover also that there is no excess risk of HCV infection in PLHIV in this region compared to the HIV uninfected population. Seroprevalence among blood donors in and around Accra was reported to be 0.9%. 5,33 Hence, screening all PLHIV for HCV infection (as recommended by the WHO) 9 in the Ghanaian setting may not be cost-effective, and targeted screening of a subgroup of PLHIV with higher risk or suggestive symptoms could be considered as alternative strategy, in case resources are insufficient to offer testing for all PLHIV. However, such targeted screening strategy can only work if low-cost, easy-to-apply tools are available to guide identification of those at greater risk of HCV coinfection.
We intended to assess the discriminatory performance of a score for targeted HCV screening that was developed based on data from a Cambodian HIV cohort where overt high-risk groups for HCV, such as PWID or MSM, were also rare. 20 As the number of participants with current HCV infection was however very low (n = 20), we were not able to obtain a robust estimate of the score performance in this external cohort. The possibility for further exploration of other factors associated with HCV infection in the Ghanaian setting was limited due to this low number of participants with the outcome event, and the lack of complete data on other diseases that may frequently cause chronic liver disease, such as hepatitis B. The AUC of the Cambodian score applied in the Ghanaian cohort was 0.69, so potentially of some use, but difficult to interpret because of the wide confidence interval. Applying the score cut-off (≥ 0) as had been proposed for Cambodia, 36% would have needed screening and 13 out of 20 coinfections would have been found, if the score would have been applied once. Our study did not provide data to evaluate whether the missed cases would have been picked up timely if the score would be applied on yearly basis. In contrast to the findings in Cambodia, we did not see a birth-cohort (older age) effect in the Ghanaian cohort. In absence of a well-performing validated score, clinical alertness focused on signs/symptoms compatible with (chronic) liver disease (such as increased ALT or AST, increased APRI, clinical symptoms compatible with chronic liver disease) may be the best available guide for targeted screening in such settings.
Conclusion
Hepatitis C coinfection prevalence was very low among this cohort of Ghanaians from the Greater Accra and Central regions in whom HIV was largely heterosexually-acquired. The performance of a clinical prediction score for targeted HCV testing, originally derived from data of a Cambodian HIV cohort, was suboptimal in the Ghanaian setting. In order to identify PLHIV at risk, tools that are tailored to local context and disease epidemiology are needed for targeted HCV testing.
Footnotes
Authors’ Note
KT, ADW, JB and LL conceptualized the study. KT, ADW, LO, VC, SA, AA-N, JOC oversaw data collection. ADW analyzed the data, KT, ADW, LO, VC, SA, AA-N, JOC and LL drafted the manuscript. KT, ADW, LO, JB and LL provided critical input in the development and revision of the manuscript. All authors read and approved the final manuscript. The datasets used and analyzed in this study are available from the corresponding author upon request. Personal identifiers were excluded from the data set before analyses were performed. We therefore did not report any individual identifier. The study was approved by the Institutional Review Board of the Institute of Tropical Medicine in Antwerp, the Ethical Committee (EC) of the Antwerp University Hospital, the Institutional Review Board of Korle Bu Teaching Hospital, and the Noguchi Memorial Institute for Medical Research Institutional Review Board (NMIMR-IRB). Only patients who provided written informed consent for study participation were enrolled.
Acknowledgment
We would like to acknowledge the staff and patients of the Korle Bu Teaching Hospital, Fevers Unit for their support during the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been funded by the Flemish Government—Department of Economy, Science & Innovation through a research grant for the Institute of Tropical Medicine, Antwerp, Belgium. The views expressed in this article are those of the authors and not the funding agency. The funder of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report.