Abstract
Background:
Pegylated interferon (PEG-IFN)/ribavirin (RBV)-related cytopenias have been associated with improved virological outcomes among hepatitis C virus (HCV)-monoinfected patients. This analysis evaluated PEG-IFN/RBV-related cytopenias with virological responses among HIV/HCV-coinfected patients.
Methods:
Pooled data from PARADIGM and AIDS Pegasys Ribavirin International CO-infection Trial (APRICOT) trials of HIV/HCV-infected patients treated with PEG-IFN/RBV. Virologic response was categorized as HCV RNA detectable (end of treatment nonresponders [ET-NR]) or nondetectable (end of treatment responders [ETR]). Declines in hemoglobin (Hgb), platelets, neutrophils, lymphocytes, and weight between the groups were compared via analysis of covariance and Cochran-Mantel-Haenszel test.
Results:
A total of 474 patients, 291 ET-NR and 183 ETR (67 relapsers, 116 with sustained virologic response), 81% male, 52% Caucasian, 88% noncirrhotic, and 67% nondetectable HIV. The ETR experienced greater Hgb declines (≥3.0 g/dL) from baseline (73.8% versus 55.0%; P < .0001), neutrophils ≤1 and ≤ 0.5 × 109/L (66.1% versus 56.4%; P = .0334 and 42.6% versus 33.3%; P = .0312, respectively), and lymphocytes ≤1.5 and ≤0.5 × 109/L (99.5% versus 87.6%; P < .0001 and 24.6% versus 14.9%; P =.0079, respectively).
Conclusions:
The HIV/HCV-coinfected patients with ETR experienced greater declines in Hgb, neutrophils, and lymphocytes than the ET-NR patients.
Background
The treatment of patients monoinfected with hepatitis C virus (HCV) includes combinations containing pegylated interferon (PEG-IFN) plus ribavirin (RBV). 1 Although beneficial, these agents are associated with significant adverse drug events that can be limiting. 2 Of particular concern are high rates of cytopenias, such as anemia, thrombocytopenia, lymphopenia, and neutropenia. These effects may directly impact the overall health of patients, lead to premature dose reduction, and increase overall health care costs. These cytopenias, however, represent the host’s immunologic response to treatment and therefore could be a marker for the likelihood of successful anti-HCV therapy. A number of retrospective studies have evaluated the impact of cytopenias on virologic outcomes among HCV-monoinfected patients. 3 –5 Lindsay et al observed lower rates of thrombocytopenia, lymphopenia, and weight loss during treatment with PEG-IFN/RBV to be independently associated with achieving a null response. 3 Rodriguez-Torres and colleagues noted that greater declines in neutrophil and hemoglobin (Hgb) concentrations from baseline after 12 weeks of PEG-IFN/RBV treatment were predictive of an undetectable HCV RNA, 4 and Chung et al reported significantly greater declines in neutrophils and platelets among virologic responders compared with nonresponders. 5 In addition, a number of studies have observed higher rates of virologic response with increased rates of anemia 6,7 and RBV dosage and/or exposures. 8 –13 Taken together, these data suggest that such cytopenic events not only represent adverse effects of treatment but may also provide useful information in predicting the likelihood of a beneficial virologic outcome.
Coinfection with HIV and HCV has been reported to occur in 15% to 30% of the patients infected with HIV. 14,15 The use of PEG-IFN/RBV in coinfected patients, compared to monoinfected patients, has been associated with lower sustained virologic response rates (27%-40%) and higher incidence of adverse drug events (eg, neutropenia and anemia). 16 –18 In the AIDS Pegasys Ribavirin International CO-infection Trial (APRICOT), neutropenia and anemia resulting in dose reductions or discontinuations occurred in 28% and 17% of the treated patients, respectively. 18 Butt et al, in a retrospective analysis of a prospective National Institutes of Health study, found that 40% of HIV/HCV-coinfected patients developed anemia during the first 12 to 18 weeks of treatment 19 ; among those patients who achieved an early virologic response, 55% eventually developed anemia, and only 27% completed the study without any RBV dose modification. Finally, Talal et al reported anemia and neutropenia in 47% and 27% of the 92 HIV/HCV-coinfected patients, respectively. 20
Clinical data evaluating the association of IFN-related adverse events on clinical response in HIV/HCV-coinfected patients are limited. The safety and efficacy of PEG-IFN alpha-2a and RBV in IFN-naive HCV/HIV-coinfected patients have been evaluated in 2 large randomized clinical trials, APRICOT and PEGASYS (PEG-IFN alpha-2a [40 kDa]) Plus Copegus (Ribavirin) in patients with chronic hepatitis C (CHC) Genotype 1 and Human Immunodeficiency Virus-1 (HIV-1) Co-infection (PARADIGM). After adjusting for differing RBV doses, we pooled these 2 trials to evaluate the pharmacodynamic impact of cytopenias and weight loss as a predictor of virologic response at the end of treatment for genotype 1 HCV in HIV-coinfected patients treated with PEG-IFN/RBV.
Patients and Methods
Patients
Patients with HCV genotypes 1a or 1b coinfected with HIV, not receiving zidovudine (ZDV), and who received 48 weeks of combination therapy of PEG-IFN alpha-2a (Pegasys; Roche, South San Francisco, California; 180 µg/week) and RBV (Copegus; Roche; 800 mg or 1000-1200 mg/d) were pooled from 2 large clinical trials. The APRICOT and PARADIGM were randomized, multicenter phase 3 and 4 trials, respectively, in IFN-naive patients with CHC and stable HIV infection; both the studies used standard PEG-IFN alpha-2a dosing; however, APRICOT used RBV 800 mg/d, whereas PARADIGM dosed RBV at either 800 mg/d or 1000 to 1200 mg/d based on weight. In addition to RBV dosing, APRICOT differs from PARADIGM in study era (2003 versus 2009), liver biopsy requirement (no requirement versus required before first dose), and HCV RNA undetectable limits (<50 versus <20 IU/mL). Only patients who received at least 1 dose of study medication were pooled and included in the analysis.
Study Design
The objective of the study was to review and assess the association of cytopenias (anemia, neutropenia, lymphopenia, and thrombocytopenia) and weight loss with virologic response in HIV/HCV-coinfected patients not receiving ZDV who are of genotype 1 and treated with PEG-IFN alpha-2a plus RBV.
Sustained virologic response (SVR) is the most common method used to evaluate the effectiveness of HCV treatment. However, the pooled results from APRICOT and PARADIGM showed an overall SVR of 24%. As a result, to increase the sample size for analysis, virologic response categories at the end of treatment (completion of 48 weeks of PEG-IFN/RBV or discontinuation of therapy) were used and defined as either HCV RNA detectable (end of treatment nonresponders; ET-NR) or nondetectable (end of treatment responders; ETR). By definition, ETR patients, poststudy, included relapsers and those with an SVR. Demographic differences between virologic response groups were compared using a 2-tailed Fisher exact test. The maximum decrease (eg, the lowest postbaseline measurement during treatment subtracted from baseline measure) in weight, Hgb, neutrophil, lymphocyte, and platelet counts were calculated for each patient and analyzed for both virologic response groups. The mean maximum decreases in values from baseline among the ET-NR and ETR groups were compared via analysis of covariance with a type I error rate of P < .05 using the following covariates: age, sex, race, weight in kilograms, cumulative PEG-IFN alpha-2a dose (µg), cumulative RBV dose (mg), histological diagnosis, HCV RNA titer (log10 IU/mL), HIV RNA titer (log10 copies/mL), alanine aminotransferase (ALT) values (U/L), and corresponding baseline values.
The incidence of anemia, neutropenia, lymphopenia, and thrombocytopenia were also assessed using categorical parameters: (1) anemia, percentage of patients achieving Hgb measurements of ≤9.4 g/dL or a ≥3.0 g/dL decline from baseline, (2) neutropenia, percentage of patients with a neutrophil count of ≤1.5 × 109/L, ≤1.0 × 109/L, or ≤0.75 × 109/L, (3) lymphopenia, percentage of patients with lymphocyte counts of ≤1.5 × 109/L or ≤0.5 × 109/L, and (4) thrombocytopenia, percentage of patients with a platelet count of ≤9 × 109/L. Groups were compared via Cochran-Mantel-Haenszel test stratified by sex and cirrhosis status with a type I error rate of P < .05.
The time to occurrence of first cytopenia event of anemia, neutropenia, lymphopenia, or thrombocytopenia (as described by the categorical values previously) was evaluated; the survivorship and hazard of events between the HCV detectable and nondetectable groups were evaluated using the Kaplan-Meier method and Cox proportional hazard models (with 95% confidence intervals).
Results
Patient Demographics and Clinical Characteristics at Baseline
Table 1 presents the baseline demographics and clinical characteristics of the 474 patients infected with HCV genotype 1 and coinfected with HIV from the 2 randomized clinical trials of 48 weeks of treatment with PEG-IFN alpha-2a and RBV. Overall, the majority of patients were male (81%), Caucasian (52%), and without cirrhosis (88%). It should be noted that while roughly one-half had an AIDS diagnosis, the majority of patients had well-controlled HIV, with 67% having an HIV RNA <50 copies/mL and 94% having CD4 counts >200 cells/mm3 at baseline.
Patient Demographics and Clinical Characteristics at Baseline (n = 474).

Abbreviations: ALT, alanine aminotransferase; HAI, hepatic activity index; HCV, hepatitis C virus; SD, standard deviation.
a P = .0003.
b n = 181.
c n = 290.
d P = .0063.
e AIDS diagnosis only collected in PARADIGM trial (total n for Responders and Nonresponders was 132 and 219, respectively).
At the end of treatment, 291 patients (61%) had detectable HCV RNA, whereas 183 patients (39%) had nondetectable HCV RNA. Of the 183 ETRs, 67 were relapsers and 116 achieved a sustained virological response for an overall SVR of 24.4%. African Americans (P = .0003) and those with cirrhosis at baseline (P = .0063) were more likely to have detectable HCV RNA at the end of treatment (Table 1).
Change in Pharmacodynamic Parameters by Virologic Response Category
The mean ± standard deviation (SD) maximum decrease from baseline was 3.5 ± 1.6 g/dL for Hgb, 86.0 ± 49.6 × 109/L for platelets, 1.1 ± 0.6 × 109/L for lymphocytes, 2.1 ± 1.3 × 109/L for neutrophils, and 5.0 ± 4.0 kg for weight. Overall, 13% of the patients experienced Hgb values of ≤9.4 g/dL, whereas 62% had Hgb declines >3.0 g/dL from baseline. With regard to neutrophil counts, 87%, 60%, and 37% had neutrophil declines of <1.5, 1.0, and 0.75 × 109/L, respectively, while receiving PEG-IFN/RBV. Lymphocyte declines of <1.5 and 0.5 × 109/L occurred in 92% and 19% of the patients, respectively. Finally, 35% of the patients experienced platelet declines of <99 × 109/L during treatment.
The least-squares means on maximum decreases in hematologic parameters and weight by virologic response categories are shown in Figure 1. No significant differences were observed between groups; however, the change in platelets approached but did not reach statistical significance (mean maximum decrease: 91.6 versus 83.3 × 109/L for the ET-NR versus ETR groups, respectively; P = .0551). Figure 2 shows the least squares means on changes over time for cytopenia and weight.
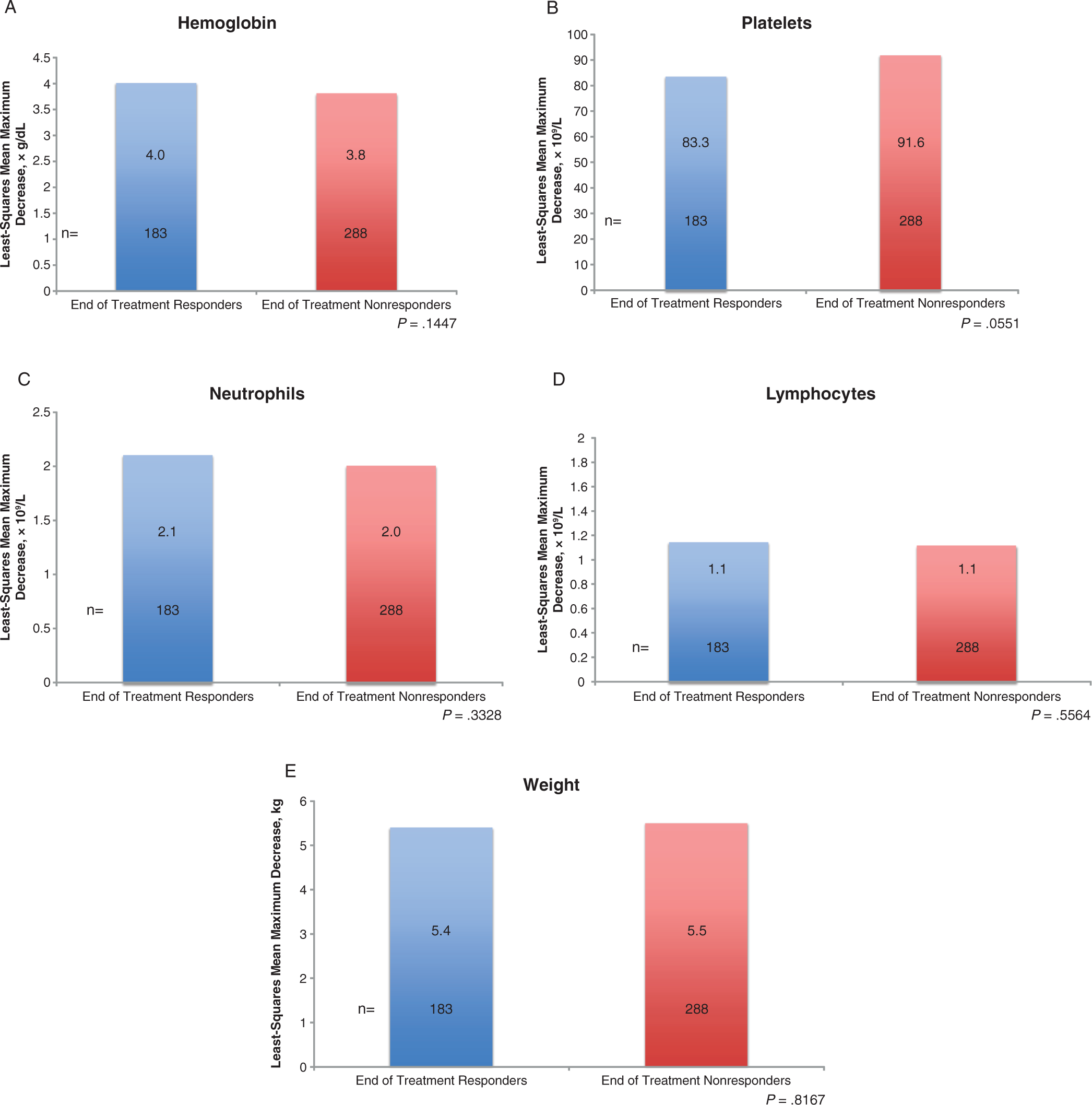
Least-squares mean on maximum decrease from baseline in pharmacodynamic parameters by virologic response categories. Groups were compared via analysis of covariance using a mixed effect model with a type I error rate at P < .05. Covariate parameters included age, sex, race, baseline weight in kilograms, cumulative PEG-IFN alpha-2a dose (µg), cumulative ribavirin dose (mg), histological diagnosis, HCV RNA titer (log10 IU/mL), HIV RNA titer (log10 copies/mL), alanine transaminase values (U/L), and corresponding baseline values. HCV indicates hepatitis C virus; PEG-IFN, pegylated interferon.

Least-squares mean on changes from baseline in pharmacodynamic parameters at each time point by virologic response categories. patients who experienced end of treatment nondetectable HCV RNA; solid lines are patients who had detectable HCV RNA at the end of treatment. Groups were compared via analysis of covariance using a mixed effect model with a type I error rate at P < .05. Covariate parameters included age, sex, race, baseline weight in kilograms, cumulative PEG-IFN alpha-2a dose (µg), cumulative ribavirin dose (mg), histological diagnosis, HCV RNA titer (log10 IU/mL), HIV RNA titer (log10 copies/mL), alanine transaminase values (U/L), and corresponding baseline values. *P < .05. BL indicates baseline; HCV, hepatitis C virus; PEG-IFN, pegylated interferon.
The categorical declines in hematologic parameters by virologic response category are shown in Figure 3. Significant differences between the ETR group compared with the ET-NR group were observed in Hgb declines ≥3.0 g/dL from baseline (73.8% versus 55.0%; P < .0001), neutrophils ≤1 × 109/L (66.1% versus 56.4%; P = .0334) and ≤0.75 × 109/L (42.6% versus 33.3%; P = .0312), and lymphocytes ≤1.5 × 109/L (99.5% versus 87.6%; P < .0001) and ≤0.5 × 109/L (24.6% versus 14.8%; P = .0079).

Categorical evaluation of changes from baseline in pharmacodynamic parameters by virologic response categories. Groups were compared via Cochran-Mantel-Haenszel test stratified by sex and cirrhosis status with a type I error rate at P < .05. Hgb indicates hemoglobin.
Table 2 lists the survivorship and hazard of events for the various cytopenias. Patients in the ETR group were 44% more likely to first experience a Hgb decline ≥3.0 g/dL from baseline (P = .0038) and 25% more likely to experience lymphocytes ≤1.5 × 109/L (P = .0346) than ET-NR patients. However, platelet declines to <99 × 109/L were more likely to first occur in ET-NR patients (P = .0330).
Time to First Event.a

Abbreviations: CI, confidence interval; HCV, hepatitis C virus; PEG-IFN, pegylated interferon.
a Covariate parameters included age, sex, race, baseline weight in kilograms, cumulative PEG-IFN alpha-2a dose (μg), cumulative ribavirin dose (mg), histological diagnosis, baseline HCV RNA titer (log10 IU/mL), baseline HIV RNA titer (log10 cps/mL), and baseline alanine transaminase values (U/L).
Discussion
The treatment of HCV in HIV-infected patients is often complicated by low sustained virologic response rates and increased incidence of adverse drug events. 16 –18 Data from the current analysis of 2 clinical trials of HIV/HCV-coinfected patients are consistent with these observations, in that 39% of patients treated with PEG-IFN/RBV for 48 weeks achieved nondetectable HCV RNA at the end of treatment, and 24% of patients achieved an SVR. Although not the focus of this analysis, it is also notable that 67 of the 183 ETR relapsed.
The primary focus of our analysis was on cytopenias and virological response. Within the ETR group, 60%, 35%, and 62% experienced neutropenia (neutrophils ≤1.0 × 109/L), thrombocytopenia (platelets ≤99 × 109/L), and decline in hemoglobin >3.0 g/dL from baseline, respectively. Many factors have been shown to be predictive of increased SVR rates, including low baseline HCV RNA measurements, Caucasian race, interleukin 28 genotype, non-HCV genotype 1 or 4, improved hepatic function prior to treatment initiation (eg, noncirrhosis, ALT ratio ≤3), high rates of adherence to therapy, and increased RBV exposures. 1,13,21 –24 In HCV-monoinfected patients, greater declines in platelets, white blood cells, and red blood cells during treatment with PEG-IFN/RBV have been associated with increased rates of virologic response. 3 –5 We set out to evaluate the impact of these parameters on HIV/HCV-coinfected patients by pooling data from 2 large randomized trials in genotype 1 patients who received 48 weeks of treatment with PEG-IFN/RBV.
The rate and magnitude of hemoglobin declines following initiation of treatment with PEG-IFN/RBV have been shown to be predictive of RBV dosage decreases and/or use of erythropoiesis-stimulating agents. 2,19,21,24,25 Although the overall maximum declines in Hgb values did not differ between virologic response groups, more ETR patients experienced a >3.0 g/dL decline in Hgb from baseline compared with ET-NR patients (Figure 3A). In addition, ETR patients were 44% more likely to experience this decline earlier than ET-NR patients (Table 2). Our findings are similar to those observed in HCV-monoinfected patients who were more likely to have an SVR when their Hgb values decreased by >3.0 g/dL. 6 Although we did not evaluate the time to first event on outcomes in the current study, early declines in Hgb values (eg, ≥1.5 g/dL at week 2) have been found to be predictive of both more significant Hgb declines during treatment and greater likelihood of achieving earlier virologic control (eg, 12 weeks). 26
A large percentage of patients experienced neutropenia or lymphopenia, with close to 90% and 96% having neutrophil or lymphocyte counts, respectively, of ≤1.5 × 109/L; however, no differences in the maximum decline from baseline were observed between treatment groups for either cytopenia. At the end of treatment, ETR patients had significantly higher proportions of lymphocyte counts (≤1.5 and ≤0.5 × 109/L) and neutrophil counts (≤1.0 and ≤0.75 × 109/L; Figure 3C and D). The impact on neutrophils is consistent with that observed in HCV-monoinfected patients 3 –5 ; however, these are the first data to evaluate the effect of lymphocyte changes on HCV virologic response to PEG-IFN/RBV in HCV-monoinfected or HIV/HCV-coinfected patients. To further evaluate these findings, we measured the mean changes over time in both the CD4 percentage and the CD4-CD8 ratio; however, no differences were observed between the groups (data not presented).
Platelet declines in HCV-monoinfected patients have been correlated with improved virologic responses. 3,5 In the current study, we did not find this association. In fact, the maximum platelet declines from baseline were greater in ET-NR patients; however, the difference between groups approached but did not reach statistical significance (Figure 1B; P = .0551). No differences were observed between groups with regard to the proportions of patients with platelets ≤99 × 109/L, but ET-NR patients were significantly more likely to experience this event earlier in the course of treatment with PEG-IFN/RBV (Table 2; P = .0330). The precise reason as to why our findings differ from those reported in HCV-monoinfected patients is unknown, but the difference may be reflective of the hepatic function of the patients in the 2 studies that were pooled for analysis. Overall, only 12% of the patients in this data set had cirrhosis at baseline (n = 58 of 474); as such, this small sample size could limit the ability to fully evaluate these findings. In addition, patients without cirrhosis at baseline were found to have a statistically significant lower hazard of time to first platelet count ≤99 × 109/L than patients with cirrhosis (P < .0001; data not shown). The impact of PEG-IFN/RBV on platelet counts in HIV/HCV-coinfected patients with more advanced liver disease has yet to be determined.
Previous studies in HCV-monoinfected patients who received treatment with PEG-IFN/RBV have shown weight loss to be correlated with improved virologic response. 3 We did not observe any difference in body weight changes between ETR and ET-NR groups in this cohort of HIV/HCV-coinfected patients. The precise reason for this observation and its potential impact on clinical decision making in HIV/HCV-coinfected patients has yet to be determined.
There are a number of limitations to the current study. First, 2 of the 3 treatment arms in the 2 pooled studies received RBV at a dose of 800 mg/d; although this dose is commonly used in this patient population, numerous studies have shown improved responses with increased RBV dose and/or plasma exposures. 8 –13 The studies that were pooled for this analysis did not record the RBV plasma levels, and, therefore, we were unable to evaluate their impact on treatment outcomes. Second, patient adherence to therapy is necessary for an SVR; studies of both HCV-monoinfected and HIV/HCV-coinfected patients have shown adherence rates of roughly 80% or higher to be associated with a beneficial virologic outcome. 13,22 We did not directly evaluate the effect of pill counts and adherence on outcomes; however, cumulative dosages of both RBV (average daily dose adjusted for weight) and PEG-IFN were built into the covariate models. Third, recent studies in both HCV-monoinfected and HIV/HCV-coinfected patients have shown genetic variants resulting in inosine triphosphatase deficiency (ITPA) being protective against RBV-induced hemolytic anemia. In HIV/HCV coinfection, reduced ITPA activity has been observed in 33% to 81% of the patients. 27,28 The 2 studies that were pooled for this analysis did not evaluate patients for ITPA deficiency, and, therefore, our analysis could not assess its impact on outcomes or adverse events. Finally, with the approval of the new HCV protease inhibitors telaprevir and boceprevir, the standard of care for genotype 1 HCV-monoinfected patients has shifted to triple therapy with these agents plus PEG-IFN/RBV. 29 Although treatment with triple therapy has significantly improved SVR rates in HCV-monoinfected patients, it has also increased the incidence of hematologic adverse events. For example, when compared with PEG-IFN/RBV in HCV-monoinfected patients, boceprevir results in anemia rates that are roughly doubled, 30,31 and telaprevir has been estimated to decrease Hgb values by an additional 0.5 to 1.0 g/dL by week 12. 32 In addition, the rates of grade 3 neutropenia were more common in both telaprevir- and boceprevir-treated patients in key registration trials (versus PEG-IFN/RBV; telaprevir: 37% versus 22%, boceprevir: ≈25% versus 14%). 30,33 How these increased rates of adverse drug events will impact the assessment of immunologic changes in cytopenias on treatment outcomes has yet to be determined in both HCV-monoinfected and HCV/HIV-coinfected patients. However, these study data expand the current understanding of the clinical impact of cytopenias in coinfected patients, while further studies are being conducted in the era of evolving HCV treatment paradigms.
In conclusion, among genotype 1 HCV-infected patients coinfected with HIV not receiving ZDV and being treated with PEG-IFN/RBV for 48 weeks, patients who had nondetectable HCV RNA at the end of treatment experienced greater declines in Hgb concentrations, neutrophil, and lymphocyte counts than patients who had detectable virus at the end of treatment. These are the first data suggesting a correlation of lymphocyte declines with HCV RNA outcomes in either HCV-monoinfected or HIV/HCV-coinfected patients.
Footnotes
Acknowledgments
The authors would like to acknowledge the third-party writing assistance for this manuscript furnished by Andrew D. Luber, PharmD, and Sue Currie, PhD, Health Interactions, which was provided by Genentech Inc and F. Hoffmann-La Roche Ltd.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Genentech Inc and F. Hoffmann-La Roche Ltd.
