Abstract
Several studies have demonstrated that HIV/hepatitis C virus (HCV)-coinfected patients experience more rapid fibrosis progression. In this study, to estimate the annual rate of direct liver fibrosis progression, we used analyses of paired biopsy samples from HIV/HCV-coinfected patients without prior treatment of hepatitis and assessed the possible association of fibrosis progression with certain clinical variables. We evaluated 30 HIV/HCV-coinfected patients, with no history of prior treatment of hepatitis C, who underwent paired liver biopsies. All patients were under antiretroviral therapy at first and second biopsies. The average annual progression rate was 0.13 fibrosis unit/year, with 36.7% of patients defined as progressors. Liver fibrosis progression was associated with alanine aminotransferase (ALT; P < .001) and aspartate aminotransferase (AST; P < .0340) levels over 3 times the upper limit of normal present at first biopsy. Elevated ALT and AST levels appear to be associated with more accelerated liver fibrosis progression among HIV/HCV-coinfected patients.
Keywords
Introduction
Currently, worldwide approximately 35 million individuals are infected with HIV worldwide. Of these, approximately 20% (around 7 million) are hepatitis C virus (HCV) carriers. 1,2 With the advent of antiretroviral therapy (ART) and increased life expectancy, chronic hepatitis C has also become a major cause of hospitalization and mortality among HIV-infected patients. 3 Several studies have demonstrated that HIV/HCV-coinfected patients experience more rapid fibrosis progression, particularly those most severely immunosuppressed. 4 –11 Other authors, when evaluating the same issue, did not observe more rapid fibrosis progression in this group of patients 12 –14 when compared to patients having HCV not infected with HIV. Several factors, such as infection duration, age, male sex, >50 g daily alcohol consumption, low CD4 count, and necroinflammatory activity, have been associated with more accelerated liver fibrosis progression. 5,7,10,15,16 Some metabolic conditions, such as obesity, steatosis, and insulin resistance, are also emerging as independent cofactors of fibrogenesis. 17
The annual rate of fibrosis progression can be directly calculated or estimated for each patient. This rate is calculated by the ratio between the difference in fibrosis stages between the 2 biopsies and the respective time interval. The estimated annual rate of progression is calculated by the ratio between the fibrosis stage at first biopsy and estimated duration of infection. 18 Most authors used the estimated rate in evaluating fibrosis progression in HIV/HCV-coinfected patients. 4,7,10,13,14
The present study aimed to evaluate direct liver fibrosis progression, using paired biopsy samples from HIV/HCV-coinfected patients who did not receive prior treatment of hepatitis C. We also intended to evaluate possible associations between certain clinical variables and the probability of the infection’s histological progression.
Patients and Methods
Study Population
The study population was derived from a cohort of HIV-infected patients followed at a specific ambulatory setting directed to HIV-infected patients and their sexual partners, in Saõ Paulo, Brazil. In this clinic, several tests are requested during the first consultation, including serology for hepatitis B and C viruses. Patients diagnosed with viral hepatitis are continuously monitored at the same clinic. An electronic and manual database, containing information of all patients, was the data source for this study.
Liver biopsies are generally used for staging liver disease, as they indicate the need for specific therapy. Clinical indication of biopsies followed recommendations by the Brazilian Ministry of Health, using specific protocol. 19
From June 2008 to February 2011, HIV/HCV-coinfected patients meeting the following criteria were evaluated: above 18 years, having undergone 2 paired liver biopsies with at least 12-month interval between them, and not receiving anti-HCV-specific treatment prior to biopsies. The following patients were excluded from the study: those with other causes of liver disease, including chronic carriers of hepatitis B virus, those with the presence of cirrhosis at first biopsy, those whose liver biopsies were not considered representative (less than 10 portal spaces in the histopathological assessment) by the on-site pathologist; those with any other cause of chronic liver disease, and those who received a liver transplant. The on-site ethics committee reviewed and approved the study.
Laboratory Tests for HIV and HCV Infection
All patients were diagnosed with HIV and HCV infection. For HIV diagnosis, we used an enzyme immunosorbent assay (EIA) and 2 commercial enzyme-linked immunosorbent assays (Organon Technika, Tournault, Belgium and Embrabio, São Paulo, Brazil) for confirmation. The specimens that tested positive by EIA and those with discordant or indeterminate results were confirmed by Western blot. To test hepatitis C, we used third-generation EIAs for HCV antibodies. To evaluate the presence of HCV RNA, we used nested polymerase chain reaction according to the method used by Garson and others. 20,21 Hepatitis C virus genotyping was performed using the INN0-LiPA-II HCV kit (Innogenetics, Ghent, Belgium) according to the manufacturer’s instructions.
Data Collection and Variables Evaluated
Medical records were retrospectively reviewed to obtain clinical and demographic characteristics, in addition to the laboratory data for the 6 months preceding liver biopsy. Demographic variables included age and sex. The variables associated with HIV and HCV were as follows: mode of HIV/HCV transmission; history of opportunistic diseases, according to the criteria adopted by the Centers for Disease Control and Prevention; and use of ART 6 months before each liver biopsy (regimens containing only nucleoside reverse transcriptase inhibitors or highly active ART—regimens containing protease inhibitors and/or nonnucleoside reverse transcriptase inhibitors).
Six months before each liver biopsy, the following laboratory data were evaluated: CD4counts, HIV viral load, alanine aminotransferase (ALT) and aspartate aminotransferase (AST), platelet count, γ-glutamyl transferase.
We also evaluated the nadir CD4 counts and HCV genotypes. The following comorbidities were evaluated: diabetes mellitus (fasting glucose above 125 mg/dL or above 200 mg/dL when sampled at random on more than 1 occasion or using oral hypoglycemic agents or insulin), dyslipidemia (serum triglyceride levels 3 150 mg/dL or low-density lipoprotein cholesterol 3 160 mg/dL on more than 1 occasion), and arterial hypertension (as described in medical records by the attending physician). In this study, the presence of metabolic disorder was defined as the presence of at least one of the following diagnosis: diabetes mellitus or dyslipidemia or arterial hypertension.
In this study, alcohol abuse was defined as more than 50 g alcohol consumption per day, within a period of 6 months prior to liver biopsy. The length of alcohol consumption is not included because it was a retrospective study and this information was not available.
Normal ALT and AST levels were defined as a value below 31 U/L and 41 U/L for women and men, respectively. Those values were determined according to the standard methods used at the Central Laboratory at Clinics Hospital of School of Medicine of University of São Paulo.
Liver Biopsies
Transcutaneous liver biopsies were guided by ultrasound performed only in those biopsy-indicated patients. Liver fragments were fixed, embedded in paraffin, and stained with hematoxylin–eosin, Masson trichrome, and reticulin (for collagen). All liver fragments had more than 10 portal spaces. Histological findings were classified according to the METAVIR classification (fibrosis score from F0 to F4 and inflammatory activity from A0 to A3). 18,22 Two experienced pathologists analyzed retrospectively the slides while remaining “blind” to each patient’s status.
Paired biopsies were analyzed in order to evaluate the rate of liver fibrosis progression. Histologic regression was defined as a decrease by at least 1 point in fibrosis score, progression as an increase by at least 1 point, and stability as unaltered fibrosis from the first to second biopsy according to the METAVIR classification. 5,9
The annual rate of fibrosis progression of the biopsy samples was calculated as follows: ratio between the differences in fibrosis stages (METAVIR fibrosis units [FUs]) from the 2 biopsies and the respective time interval (in months) multiplied by 12, expressed in FU/year. 18 We also assessed the degree of hepatic steatosis, which is classified as follows: grade 1 (5%-33% steatosis), grade 2 (33% to 66% steatosis), and grade 3 (≥ 66% steatosis). 23
Statistical Analysis
The descriptive analysis considered the frequency distribution (absolute and relative) of the study variables. Next, we calculated the prevalence of fibrosis progression with a 95% confidence interval. Considering the high prevalence of patients defined as progressors (36.7%), we estimated the prevalence ratio with 95% confidence interval for univariate analysis of the relationship between several variables and outcome occurrence of fibrosis progression.
After testing normality by Shapiro-Wilk test, we used Student t test to compare the means of quantitative variables for individuals with and without fibrosis progression and with or without moderate and severe necroinflammatory activities. For variables not normally distributed, we used nonparametric Mann-Whitney U test. Those variables with P values less than .20 were selected for multivariate analysis. The Cox regression model with robust variance was applied in this step. Variables with P < .05 for multivariate analysis remained in the final model. The tests were performed at 5% significance level.
Results
Characteristics of Patients
During the study period, we identified 106 patients who underwent 2 liver biopsies. Of these, 76 were excluded from the study for the following reasons: 1 (1.3%) was a chronic carrier of hepatitis B virus, 1 (1.3%) had less than 12-month interval between biopsies, 14 (18.4%) were cirrhotic at first biopsy, 23 (30.3%) were treated for hepatitis C before the first liver biopsy or between biopsies, and 37 (48.7%) had less than 10 portal spaces. Thus, 30 patients were included in this study (Table 1).
Demographic and Clinical Characteristics among 30 HIV/HCV-Coinfected Patients.a
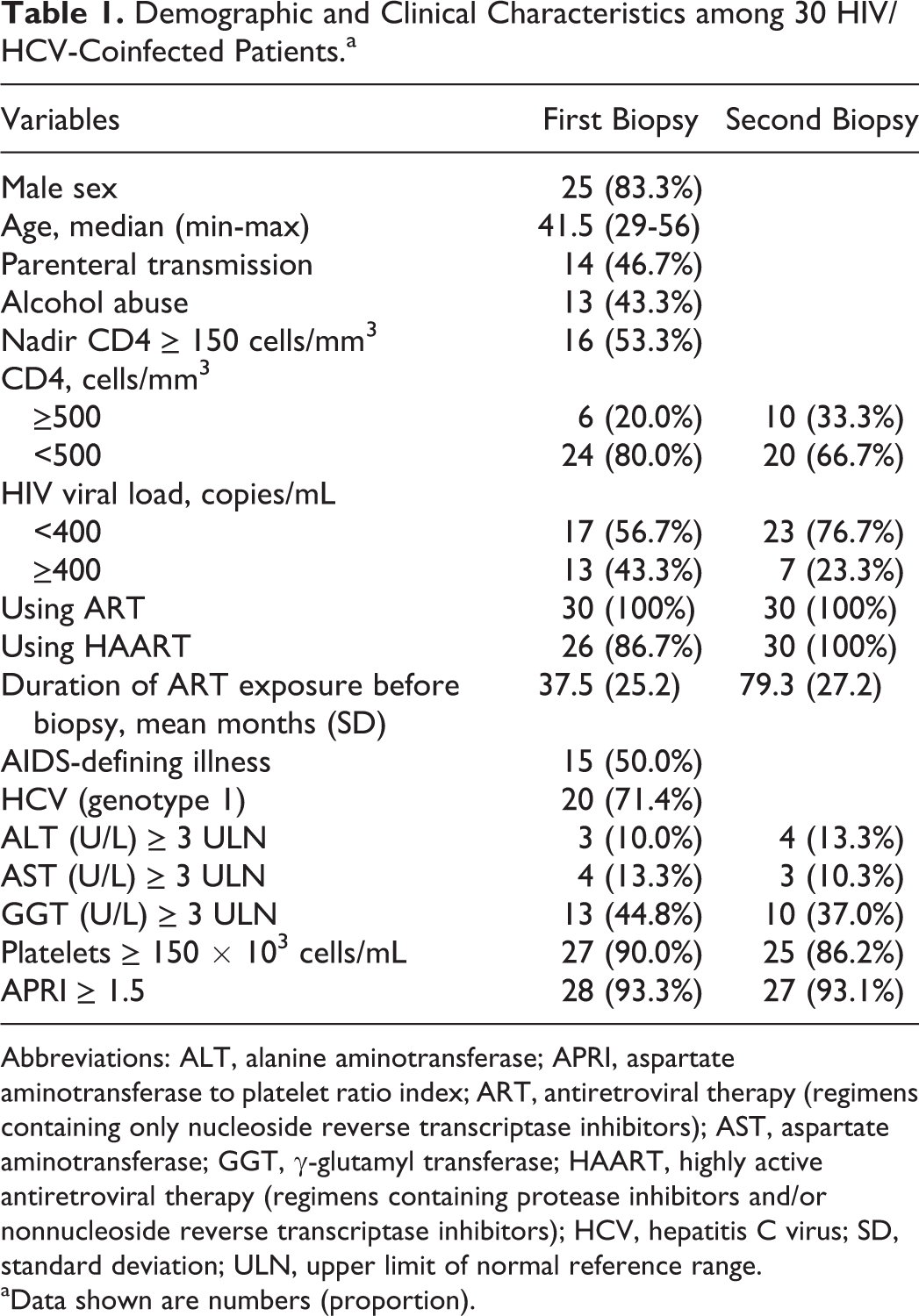
Abbreviations: ALT, alanine aminotransferase; APRI, aspartate aminotransferase to platelet ratio index; ART, antiretroviral therapy (regimens containing only nucleoside reverse transcriptase inhibitors); AST, aspartate aminotransferase; GGT, γ-glutamyl transferase; HAART, highly active antiretroviral therapy (regimens containing protease inhibitors and/or nonnucleoside reverse transcriptase inhibitors); HCV, hepatitis C virus; SD, standard deviation; ULN, upper limit of normal reference range.
aData shown are numbers (proportion).
Using the median values, we found that the CD4 count in peripheral blood was 331.5 cells/mm3 (132-1226 cells/mm3) at first biopsy and 450 cells/mm3 (13-1994 cells/mm3) at second biopsy for most patients. The median nadir CD4 count was 150 cells/mm3 (13-460 cells/mm3). We also observed that 43.3% of the patients had plasma HIV viral load higher than or equal to 400 copies/mL at first biopsy, and this value was reduced to 23.3% at second biopsy.
The following HCV genotype was identified in 28 patients: 20 with genotype 1 (71.4%), 1 with genotype 2 (3.6%), 6 with genotype 3 (21.4%), and 1 with genotype 4 (3.6%). All patients exhibited abnormal liver enzymes. This abnormality was below 3 times the upper limit of normal (ULN) for both enzymes, ALT (90%) and AST (86.7%), for most patients.
More than half of the patients (53.3%) exhibited metabolic disorders: 3 patients were hypertensive and dyslipidemic and 13 only dyslipidemic. No patients were diagnosed with diabetes mellitus.
The interval between first and second biopsies ranged from 13 to 128 months, with an average of 42 months, where half of the patients (median time) underwent 2 biopsies within a maximum interval of 35 months. Patients subjected to liver biopsy did not receive treatment of hepatitis C due to the following reasons: presence of clinical complications related to HIV or HCV infection (occurring after liver biopsy), contraindication for treating hepatitis C (occurring after liver biopsy), and patient’s desire to not get treated.
Liver steatosis was observed in most patients at both first (73.3%) and second (66.7%) biopsies. At first biopsy, 70% exhibited grade 1 steatosis and 3.3% exhibited grade 2. At second biopsy, 53.3% showed grade 1 steatosis, 3.3% grade 2, and 10.0% grade 3. None of the patients had steatohepatitis. We also found that 10.0% of the patients had METAVIR F3 and 30.0% had moderate or severe inflammatory activity (METAVIR A2 or A3) at first biopsy. Half of the patients had METAVIR A2 or A3, 4 (13.3%) had METAVIR F3, and 3 (10.0%) had METAVIR F4 at second biopsy.
Annual Rate of Fibrosis Progression
In 16 (53.3%) patients, the fibrosis score stabilized between the paired liver biopsies (average interval between biopsies of 50 months). However, 11 (36.7%) patients were defined as progressors (95% confidence interval [CI]: 18.4-55.0), with average interval between biopsies of 33 months. Of these patients, 1 (9%) had liver fibrosis stage F0, 4 (36%) F1, and 4 (36%) F2 (Table 2).
METAVIR Fibrosis Stage at First and Second Biopsies among 30 HIV/HCV-Coinfected Patientsa
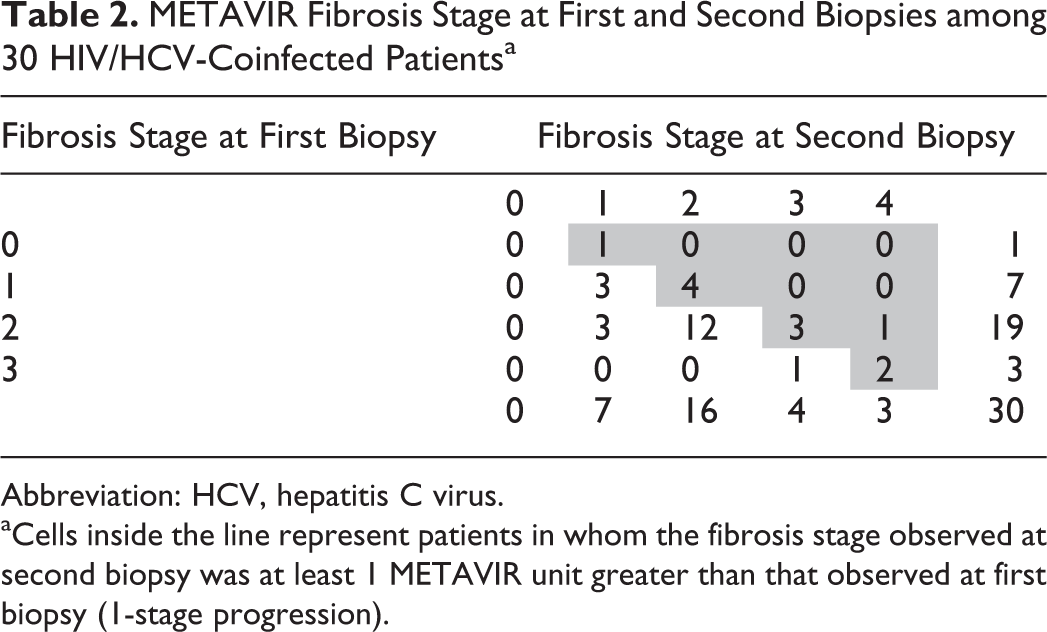
Abbreviation: HCV, hepatitis C virus.
aCells inside the line represent patients in whom the fibrosis stage observed at second biopsy was at least 1 METAVIR unit greater than that observed at first biopsy (1-stage progression).
Ten patients exhibited progression by 1 point (average interval of 31 months between biopsies), and 1 individual exhibited fibrosis by 2 points (average interval of 50 months between biopsies). There was histologic improvement in only 3 patients (10%), with fibrosis decreased by 1 point between the paired biopsies (average interval of 37 months between biopsies). Seven (23.3%) patients exhibited F3-F4 at second biopsy. Among the 30 patients studied, we found an average annual rate of fibrosis progression equal to 0.13 FU/year (95% CI: 0.02-0.24).
Factors Associated with Fibrosis Progression
We observed an association between elevated ALT higher than or equal to 3 times the ULN at first biopsy and hepatic fibrosis progression (P < .001). Similar results were found in assessing AST at first biopsy (P = .034).
There was no statistically significant difference in the prevalence of fibrosis progression according to the other variables analyzed in our study. Table 3 summarizes the results of our analysis.
Fibrosis Progression According to Hepatitis C and HIV Characteristics.a

Abbreviations: ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; GGT, γ-glutamyl transferase; HAART, highly active antiretroviral therapy (regimens containing protease inhibitors and/or nonnucleoside reverse transcriptase inhibitors); HCV, hepatitis C virus; PR, prevalence ratio; ULN, upper limit of normal reference range.
aData shown are numbers (proportion).
When measuring the central tendency of quantitative variables according to fibrosis progression, considering average interval between biopsies of 35 months, patients with fibrosis progression had shorter intervals between the biopsies compared to those without progression (P = .045). There were no factors independently associated with fibrosis progression at first biopsy by multivariate analysis.
Discussion
According to the data from our study, approximately one-third of the patients evaluated (n = 11, 36.7%) exhibited hepatic fibrosis progression over an average interval of 33 months. In most patients (n = 16, 53.3%), fibrosis stabilized, while histological improvement occurred in 3 (10%) patients. Our data are similar to those previously reported by other authors 5,6,8,9,11,24,25 who reported fibrosis progression in 20.4% to 50% of the HIV/HCV-coinfected patients evaluated, with varied time interval between biopsies, from 35 to 93 months.
The annual rate of fibrosis progression in our study was 0.13 FU/year. This rate does not differ much from what has been observed in studies on fibrosis progression in HCV mono-infected patients. According to these studies, this rate has varied from 0.086 to 0.13 FU/year. 4,12 –14,26
We observed that 4 (13%) patients with baseline fibrosis stage F2 progressed to advanced liver fibrosis. This finding is somewhat less significant than that found by Sanmartín who described a progression of 30% to advanced liver fibrosis in HIV/HCV-coinfected patients with baseline fibrosis stage F0-F2. 25 It is noteworthy that all patients in our study were undergoing ART at first biopsy, whereas HIV viral load was undetectable in 56.7% of patients.
The fibrosis stabilization observed in our patients may reflect the action of ART in HIV suppression and consequently antifibrogenesis. A study on paired biopsies found that fibrosis progression was independently related to suppressed HIV RNA in patients without anti-HCV treatment. 9 Recent studies 27,28 have postulated that in HIV/HCV coinfection, Kupffer cells, like other macrophages, can be infected with HIV and shift to a Th2 cytokine response, which affects the hepatic stellate cells, the major mediators of collagen deposition and fibrogenesis in the liver. Modulation of the antigen-presenting function of the Kupffer cells by HIV in HCV coinfection may also play a role in the progression of liver damage. In addition, stellate cells may also be targeted by HIV, where HIV infection, or gp120 exposure, leads to induce collagen synthesis. 29,30
Different studies have found an association between low CD4counts and advanced liver disease. However, the mechanisms underlying this association remain unclear. It is known that CD4 cells may modulate natural killer (NK) cell activity. 31 It has been postulated that CD4 cells are able to stimulate antifibrotic NK cell activity. Therefore, HIV-induced loss of CD4 cells together with an impaired activity of CD4 cells could contribute to accelerate progression of liver fibrosis as observed among HIV/HCV-coinfected patients.
However, we were not able to correlate CD4 counts (before first liver biopsy) with fibrosis progression. As mentioned earlier, it is possible that the clinical stability associated with HIV infection, observed in the majority of our patients, may have contributed to the fact that we were not able to associate CD4 counts and liver disease progression.
According to our data, elevated ALT (P < .001) and AST levels (P < .0340), higher than 3 times the ULN, were associated with liver fibrosis progression. Other authors have also made this observation. 5,6,10,14,24,32,33 Elevated levels of these enzymes may reflect hepatic inflammatory activity and higher activation of fibrogenesis. 34
To limit the staging error in our study, the slides were read “blindly” by 2 experienced pathologists, including only tissues with more than 10 portal spaces. This inclusion criterion increased the power of our findings and allowed for a more homogeneous sample. Furthermore, we classify progression as a change by 1 METAVIR stage. There were not interpathologist differences, which could have influenced the results. We observed only 3 patients with 1 lower stage, suggesting that sampling error was not an important factor.
Our study had some limitations. The first limitation is the fact that the number of patients included was reduced, due to applying the inclusion and exclusion criteria. Second, our cohort was mostly composed of patients with stable HIV disease, which may have prevented us from observing the disease’s natural progression that would occur if no antiretroviral (ARV) drugs were used. The use of ART may have influenced and decreased hepatic necroinflammatory process and consequently hepatic fibrosis in this group of patients. 35
However, while most studies characterize fibrosis progression indirectly via single liver biopsy, we analyzed paired biopsy samples from HIV/HCV-coinfected patients who did not receive prior treatment of hepatitis. Thus, we were able to increase the potential for forecasting progression by incorporating the elapsed time interval between first and second biopsies.
In summary, approximately one-third of the patients exhibited fibrosis progression with average duration of 3 years. In our study, we found a relationship between elevated ALT and AST levels with fibrosis progression, suggesting that AST and ALT serum levels play an important role in forecasting liver disease. Fibrosis progression should be monitored using shorter intervals than in HCV mono-infected patients, even as early as onset of ARV treatment. Elevated ALT/AST levels over 3 times the ULN may predict the risk for liver fibrosis progression in hepatitis C virus/HIV coinfected patients, particularly when biopsy or elastography is not readily available. Therefore, this technique serves as a very useful clinical marker.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
