Abstract
Objective
The objective of this study was to assess liver functions in tuberculosis patients after 6 months of treatment and determine the difference in the level of liver enzymes based on medication adherence and patient characteristics.
Methods
We used a prospective cohort design. The inclusion criteria were adult tuberculosis patients treated with the first line of antituberculosis drugs. The patients’ adherence to medication was measured using the Medication Adherence Rate Scale questionnaire. We also measured the total bilirubin, aspartate aminotransferase, and alanine aminotransferase levels over 2, 4, and 6 months of treatment.
Results
There were 233 tuberculosis patients enrolled in this study, with 58.91% male patients and the mean age of 40.31 (SD: 18.00) years. Overall, 86.3% of the patients had good medication adherence. The patients reported increased levels of aspartate aminotransferase (40%) and alanine aminotransferase (64%). There were no significant differences in total bilirubin between the adherence groups. Tuberculosis patients with poor adherence showed significantly increased levels of alanine aminotransferase and aspartate aminotransferase levels (p < 0.05). Female patients with poor adherence had high levels of alanine aminotransferase and aspartate aminotransferase.
Conclusion
The monitoring of alanine aminotransferase and aspartate aminotransferase in tuberculosis patients should be implemented during the 2nd, 4th, and 6th months of treatment, especially in female patients.
Keywords
Introduction
In 2020, Indonesia showed the second highest level of tuberculosis (TB) burden worldwide. TB has a significant impact on the medical, social, cultural, and economic growth in Indonesia. 1 The incidence of TB in Indonesia has increased from 339 in 2021 to 385 in 2022. 2 Approximately 845,000 people in Indonesia fell ill due to TB in 2020; 24,000 people fell ill due to drug-resistant TB in 2018; and 98,300 died of TB in 2018. 1
Poor adherence is one of the complex problems related to TB in Indonesia. A previous study stated that lack of information about the expected adverse effects of medication may lead to poor medication adherence in TB patients. 3 In China, more than 50% of TB patients were categorized as having good adherence. A study stated that some sociodemographic characteristics, treatment factors, knowledge about TB, mental health, and behavioral characteristics may influence TB patients’ medication adherence. 4 Our previous study in Indonesia showed that patients with good knowledge about TB experienced a less severe impact of hepatotoxicity and had good medication adherence. 5 In Ethiopia, the incidence of hepatotoxicity reached 7.9%, and factors such as comorbidity and older age had significant association with drug-induced hepatotoxicity. 6
Monitoring liver enzyme abnormalities is critically important in TB treatment due to the potential risk of drug-induced liver injury (DILI). Early detection of elevated liver enzymes allows timely interventions, such as dose adjustment or substitution with less hepatotoxic drugs, thereby preventing the progression of liver injury. Studies have emphasized that regular liver function monitoring is crucial in preventing severe outcomes of DILI, especially because symptoms of liver injury can be subtle and progression remains undetected in some patients. 7 Patients with elevated liver enzymes are at increased risk of developing serious hepatotoxicity, which can necessitate the discontinuation or alteration of the TB treatment regimen. This may complicate treatment and lead to the development of drug resistance if not managed effectively. Liver enzyme abnormalities may lead to drug side effects, such as nausea, fatigue, and jaundice, which can compromise the patient’s medication adherence. 8
During the coronavirus 2019 (COVID-19) pandemic, some innovations were launched to increase medication adherence in TB patients. A previous study showed that video directly observed therapy (DOT) was more effective in verifying the ingestion of TB medications. 9 Previous systematic reviews have shown that early recognition and appropriate management of adverse drug reactions (ADRs) during TB treatment may prevent the discontinuation of TB treatment. The ADRs experienced by TB patients included gastrointestinal disturbances, serious hepatotoxicity, ototoxicity, nephrotoxicity, and cutaneous ADRs. 10
The objective of this study was to assess liver functions in TB patients after 6 months of antituberculosis treatment and determine the difference in the level of liver enzymes based on medication adherence and patient characteristics.
Methods
This prospective cohort study enrolled adult TB patients treated with the first line of antituberculosis drugs (fixed dose combination) in 35 primary health care centers including hospitals in the area of Yogyakarta and Central Java from February 2021 to February 2022. We selected the patients purposively. The inclusion criteria were adult TB patients receiving antituberculosis treatments for 6 months. Pregnant women and patients with comorbidities related to liver function were excluded. We measured patients’ medication adherence using the Medication Adherence Rating Scale (MARS). The MARS questionnaire has five questions related to TB medication use. It uses a Likert scale with five choices: poor adherence is defined by a score of 1, good adherence is defined by a score of 5, and the gradual options are defined by scores from 2 to 4. 5 The patients were categorized as having good or poor adherence using the total score of MARS questionnaire. The good adherence group had a total score of 25, and the poor adherence group had a total score <25. We followed the patients during the 6 months of treatment.
We collected laboratory results, such as hemoglobin (Hb), total bilirubin, aspartate aminotransferase (AST), and alanine aminotransferase (ALT) levels, at the 2nd, 4th, and 6th months of treatment. We did not define DILI, but we only defined the elevation of bilirubin, AST, and ALT when they were over their upper limits. We considered the elevation in liver enzymes because we believe that these findings can be used to prevent DILI. We did not report the baseline liver enzyme levels because we had excluded patients with diagnosis related to abnormal liver functions. This study was approved by the Ethical Committee of Ahmad Dahlan University, Yogyakarta city, Indonesia (number: 012002010, date: 21 January 2021), located in Jl Prof Dr Soepomo Jaturan, Yogyakarta. We obtained written informed consent from the study patients. We have de-identified all patient details. We analyzed the data descriptively to describe the average Hb, bilirubin, AST, and ALT levels as well as TB patients with increased levels of bilirubin, AST, and ALT. Then, we analyzed the association between patients’ medication adherence and Hb, bilirubin, AST, and ALT levels. We used the Student’s t-test to assess differences in the liver enzymes levels between the good and poor adherence groups at 2, 4, and 6 months of antituberculosis treatment, revealing no significant difference. We found no missing data in this study. The methods in this study were conducted in accordance with the Helsinki Declaration of 1975, as revised in 2013. The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 11
Results
There were 233 TB patients enrolled in this study, with 58.91% male patients and the mean age of 40.31 (SD: 18.00) years. Overall, 86.26% of them had good medication adherence. Most of the patients had the highest education level of elementary to senior high school (86.26%). Most of the patients had no comorbidity (76.82%). The patients’ sociodemographic characteristics are presented in Table 1.
Patient characteristics (n = 233).

BMI: body mass index; SD: standard deviation.
Table 2 presents the association between laboratory data and medication adherence in TB patients at 2, 4, and 6 months. After 6 months of treatment, a significant association was observed between patients’ ALT and AST levels and their medication adherence (p < 0.05 and p < 0.02, respectively). Figure 1 shows the elevation in the patients’ Hb, bilirubin, ALT, and AST levels after 6 months of treatment. The level of bilirubin was above the normal range from 2nd to 6th months of treatment.
Association of Hb, bilirubin, AST, and ALT levels with medication adherence in TB patients.
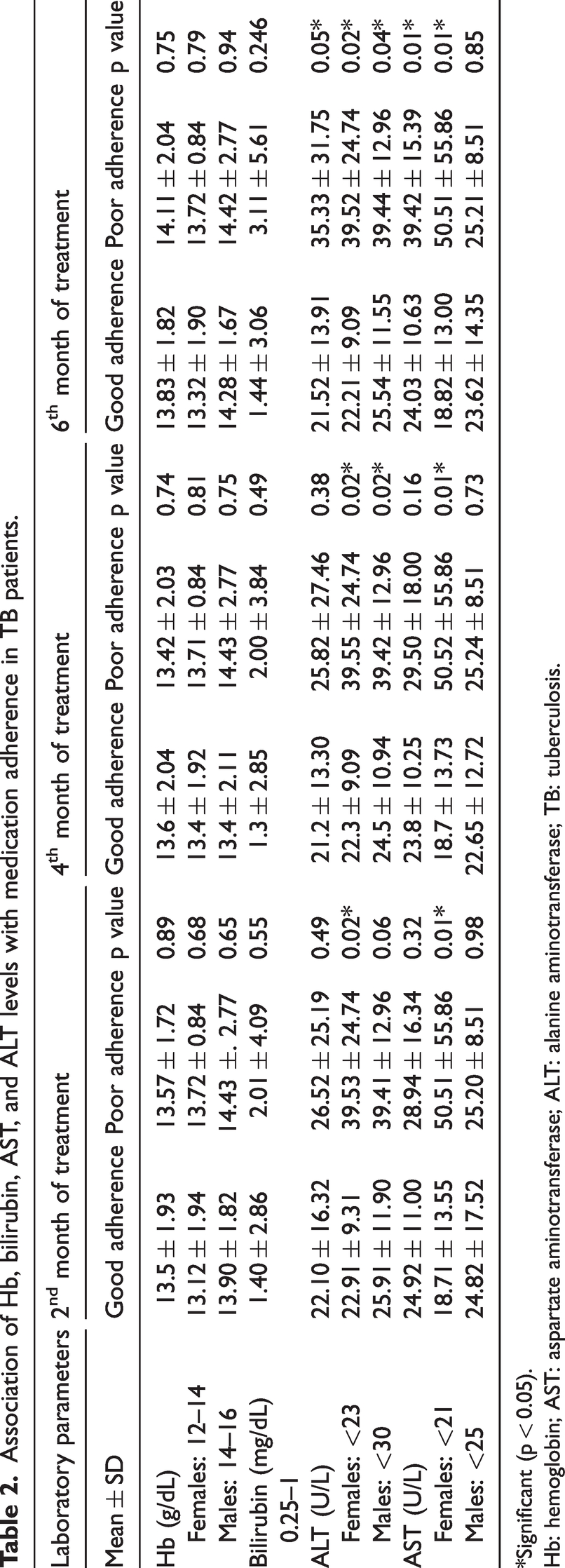
*Significant (p < 0.05).
Hb: hemoglobin; AST: aspartate aminotransferase; ALT: alanine aminotransferase; TB: tuberculosis.
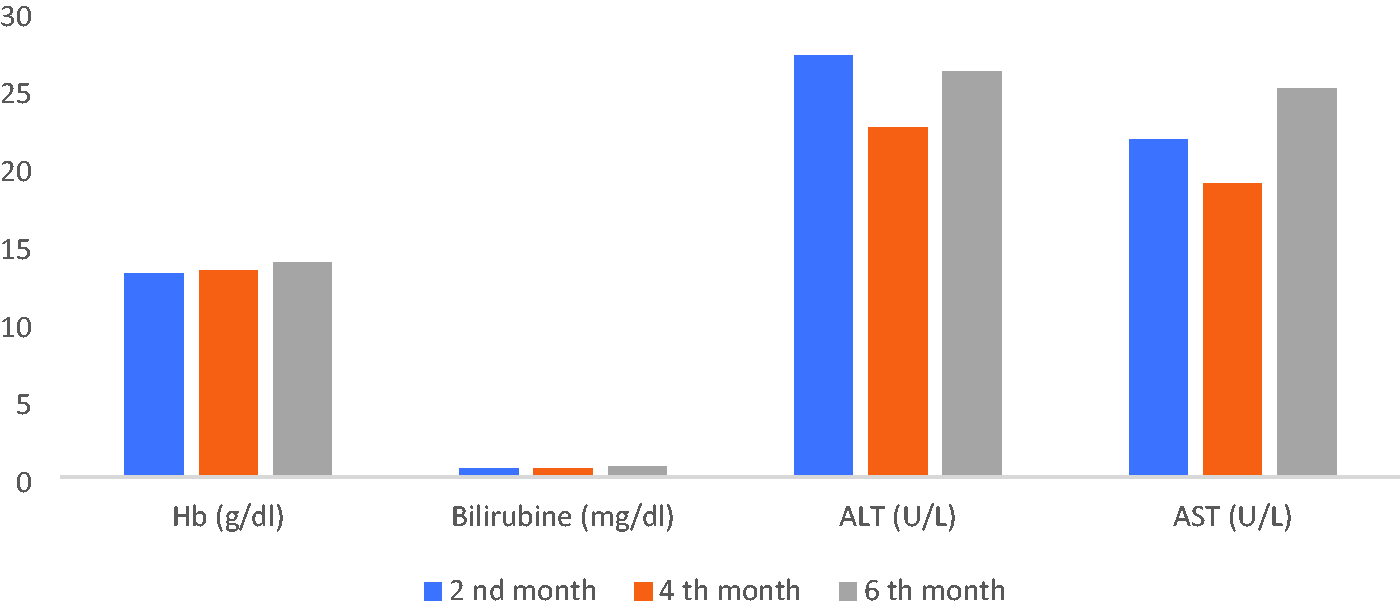
Levels of hemoglobin, bilirubin, ALT, and AST in tuberculosis patients at 2, 4, and 6 months of treatment. ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Female TB patients in the poor adherence group exhibited elevated AST and ALT levels. The differences in ALT and AST levels between the good and poor adherence groups were significant at 2, 4, and 6 months of treatment, with the AST level of females in the poor adherence group reaching two times that of the upper limit of the normal range. However, the ALT level of females in the poor adherence group reached 1.5 times that of the upper limit of the normal range. Significant differences were observed in the ALT levels of male patients at 4 and 6 months of treatment.
Figure 2 presents the proportion of TB patients who showed elevated levels of bilirubin, ALT, and AST during the treatment. The levels of bilirubin, ALT, and AST increased again at the 6th month of treatment.

Proportion of TB patients showing elevated ALT and AST levels at 2, 4, and 6 months of tuberculosis treatment. TB: tuberculosis; ALT: alanine aminotransferase; AST: aspartate aminotransferase.
Discussion
This study found that 64% and 40% of TB patients exhibited elevated ALT and AST levels, respectively. Although the levels of ALT and AST decreased in the 2nd month of treatment, they increased again in the 6th month of treatment. Female sex was found to be a predictor of medication adherence. Female patients with high levels of ALT and AST had poor adherence, owing to treatment discontinuation. Most of the TB patients in Indonesia are male; among the total TB patients, 6 million are adult males and 3.4 million are adult females. 12 Our study showed that the level of liver enzymes was significantly different between female TB patients with good and poor adherence. Many previous studies have shown that female TB patients are at increased risk of developing hepatotoxicity during antituberculosis treatment.13–15 Women and men metabolize drugs differently, partly due to hormonal variations and genetic factors. Women are often considered “slow acetylators” for drugs such as isoniazid (INH), which increases the concentration of hepatotoxic metabolites that can damage the liver. Women generally have higher levels of autoimmune responses than men. This can make them more susceptible to autoimmune-like reactions triggered by certain drugs, including antituberculosis medications, which may contribute to hepatotoxicity. In addition, women have a higher body fat percentage, which may alter drug distribution and elimination. Fat-soluble drugs or metabolites may be retained for a longer time in body tissues, leading to a higher cumulative dose exposure to the liver and thus an increased risk of hepatotoxicity. 16
The ALT and AST levels were elevated after 2 months of treatment, which decreased after 4 months and increased again after 6 months of treatment. A previous study stated that elevation of liver enzymes is observed in the early treatment of TB. 17 Moreover, these levels further increase during antituberculosis treatment. During the abovementioned study, there were no dose adjustment procedures. This is because during usual care, no dose adjustments are performed for the elevation of liver enzymes. The liver enzymes were closely monitored. 17 The physician monitored the liver function of patients who exhibited elevated liver enzymes.
INH is the most common drug related to DILI. The risk of DILI increases owing to the combined use of INH and pyrazinamide. INH is metabolized into hepatotoxicity intermediate metabolites. The direct hydrolysis of INH also results in potent hepatotoxicity.5,10 The pharmacogenomic role of INH in DILI has shown that some genes play a significant role in liver dysfunction. 18 Serum INH levels of >7 µg/mL posed a higher risk of DILI in patients in Indonesia. TB patients with slow acetylator also had a higher risk of DILI than those with rapid acetylator. 18 NAT-2 and HLA showed a significant association with DILI in Indonesian patients. 19 This association between DILI and gene polymorphism has been observed in other countries in addition to Indonesia.20–22 Rifampicin and pyrazinamide may also elevate liver enzymes. The underlying mechanism remains unclear; however, a study mentioned that superoxide generation can induce the hepatotoxic effects of antitubeculosis treatment. 23
Our study findings are supported by those of other previous studies, which revealed that age, sex, and other factors are common predictors of medication adherence in TB. 24 Contradictory results can be seen in the study by Chung et al. (2025), which mentioned that age, sex, and other sociodemographic characteristics did not influence medication adherence during the 9 months of treatment. Patients who experienced hepatotoxicity decided to discontinue the treatment. 25
Other factors related to adherence were patient-centered factors, perceptions, beliefs, lifestyle, treatment and side effect information, patient satisfaction, and comorbidities. 26 Forgetfulness and inadequate knowledge about TB were two major patient-centered factors that could influence TB patients’ medication adherence. DOT and health promotion are patient-centered solutions to resolve this issue.26,27 Removal of stigma and discrimination is important to support TB patients from the perspective of social aspects. 28 As Indonesia is famous as a country with thousands of islands, transportation and distance from public health centers remains a problem in Indonesia. Thus, DOT is mandatory for TB patients in Indonesia and may overcome the geographic problem in Indonesia. The government must think about the facilities and transportation that are affordable for TB patients. 29
Most TB patients are associated with poverty and have low education levels. 6 This study did not find any significant difference between the highest education level and medication adherence. However, the researcher found that most TB patients in Indonesia resided in houses with poor sanitation and less sunlight. Furthermore, these individuals graduated from elementary, junior high school, or senior high school. The Indonesian government has already shared many education leaflets on various topics, including TB. The Indonesian Ministry of Health has published reports on the role of pharmacists in the TB elimination program. Of these, important roles include providing motivation for medication adherence to TB patients; facilitating detection of TB; monitoring patients’ adherence; and preventing TB resistance, TB relapse, and death due to TB. 30 Pharmacists play a crucial role in improving medication adherence among TB patients. Until now, pharmacists in primary health care centers only had the responsibility of monitoring the drug management cycle, including oral antituberculosis treatment. The interaction between pharmacists and TB patients in primary health care centers is very limited. In the future, pharmacists must take responsibility for health promotion, information dissemination, and education on medication effectiveness and/or ADR monitoring. This activity will help improve medication adherence among TB patients and minimize the spread of TB by lowering the rates of TB treatment discontinuation. This responsibility must be taken up by pharmacists in collaboration with other health professionals.31,32
The limitations of this study are related to the factors that could be predictors of medication adherence in TB patients. We did not collect data regarding lifestyle, nutrition, and perceived end-beliefs, which could be the predictors of medication adherence, because of the COVID-19 pandemic. All TB patients in Indonesia follow the same pattern of TB care because of the existence of a national standard operating procedure, ensuring that the interaction between patients and healthcare providers would not influence the analysis. These data are part of the precision medicine model, which should be integrated with genomic, medication adherence, and other psychosocial aspects. We suggest that future studies must include predictors such as economic constraints, poor quality of healthcare providers, poor quality of healthcare services, patient satisfaction, being busy with work, level of health information/education, pill burden, stigma, and discrimination. The interpretation of the present study results is limited to the location where the study was performed—Yogyakarta and Central Java.
Conclusion
The 6-month antituberculosis treatment led to elevated ALT and AST levels. These levels were significantly different between the good and poor adherence groups at 2, 4, and 6 months of TB treatment in female TB patients. The levels of ALT and AST were significantly higher in females with poor medication adherence. Female sex was found to be a predictor of medication adherence in TB patients, considering the elevation in the levels of liver enzymes.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251348228 - Supplemental material for Liver functions and medication adherence in tuberculosis patients after 6 months of antituberculosis treatment in Indonesia
Supplemental material, sj-pdf-1-imr-10.1177_03000605251348228 for Liver functions and medication adherence in tuberculosis patients after 6 months of antituberculosis treatment in Indonesia by Dyah Aryani Perwitasari, Triantoro Safaria, Didik Setiawan, Haafizah Dania, Salma Auliya Fatimah and Imaniar Noor Faridah in Journal of International Medical Research
Footnotes
Acknowledgements
The authors would like to thank the Heads of the Hospitals, Lung Hospitals and Primary Health Care Centers, who provided permission to conduct this study and obtain the study results.
Author contributions
DAP designed the study, conceptualized the theories, and drafted the manuscript.
TS defined the patient, managed the funding, and drafted the manuscript.
DS collected data, analyzed the data, and drafted the manuscript.
HD designed the study, managed the data and revised the manuscript.
SAF collected data, analyzed the data, and revised the manuscript.
INF arranged for study permissions and ethical clearance, collected data, and drafted the manuscript.
Data availability statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors have no conflict of interest.
Funding
Grant research from the Ministry of Education, Cultural, Research and Technology, Number 075/E5/PG.02.00.PL/2023 date 12 April 2023; Number: 0254.8/LL5-INT/AL.04/2023 date 17 April 2023; and Number 004/WCR/LPPM UAD/IV/2023 date 24 April 2023.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
