Abstract
Background:
The importance of early initiation of antiretroviral therapy (ART) for tuberculosis (TB) and HIV-coinfected patients is controversial. We conducted a systematic review and meta-analysis to assess the effect of early initiation of ART (within 2-4 weeks of TB treatment) on several treatment outcomes among TB/HIV-coinfected patients.
Method:
A systematic search of clinical trials was performed in PubMed, Embase, Google Scholar, Science Direct, Medscape, and the Cochrane library. Clinical trials which were published in any language before the last date of search (March 31, 2015) were included. The qualities of the studies were assessed using criteria from the Cochrane Library. Heterogeneity test was conducted to assess the variations among study outcomes. For each study outcome, the risk ratio (RR) with 95% confidence interval (CI) was calculated as a measure of intervention effect. The Mantel-Haenszel method was used to estimate the RR using a fixed-effects model.
Findings:
A total of 2272 study participants from 6 trials were included in the meta-analysis. Early ART initiation during TB treatment was associated with reduced all-cause mortality (RR = 0.78; 95% CI = 0.63-0.98) and increased rate of TB-associated immune reconstitution inflammatory syndrome (TB-IRIS; RR = 2.19; 95% CI = 1.77- 2.70) and death related to TB-IRIS (RR = 6.94; 95% CI = 1.26-38.22). However, the time of ART initiation has no association with TB cure rate (RR = 0.99; 95% CI = 0.81-1.07), rate of drug toxicity (RR = 1.00; 95% CI = 0.93-1.08), death associated with drug toxicity (RR = 0.40; 95% CI = 0.14- 1.16), rate of low viral load (less than 400 copies/mL; RR = 1.00; 95% CI = 0.96-1.04), and rate of new AIDS-defining illness (RR = 0.84; 95% CI = 0.60-1.18). Immunological response in early ART arms of study participant in different trials showed a greater or equal response compared with late ART arms.
Conclusion:
This systematic review presents conclusive evidence on the reduction of all-cause mortality as a result of early initiation of ART. However, this study also confirms the high rate of TB-IRIS and death associated with it. Operational and implementation research are required to maintain the benefit of early ART initiation and proper management of TB-IRIS. Studies on the timing of ART in extrapulmonary and multidrug-resistant TB are recommended.
Keywords
Introduction
In 2012, there were an estimated 8.6 million new cases of tuberculosis (TB) globally and 13% of these cases were HIV positive. 1 There were an estimated 0.3 million HIV-associated TB deaths in the same year. 1 Concerted efforts are needed to identify and treat TB in HIV-infected persons. 2 Timely provision of antiretroviral treatment (ART) is the main intervention to reduce morbidities and mortalities associated with opportunistic infections such as TB. 3 Reports also indicate that up to 30% of patients die within the first 2 months of TB treatment in the absence of ART. 4
While TB is the most common cause of death in HIV-infected patients, many questions remain unanswered with regard to the treatment of concomitant HIV and TB infections. Current guidelines recommend institution of rifampicin-based treatment for TB regardless of HIV-coinfection status. 5 Among patients with HIV infection, ART is started within 2 to 8 weeks after initiation of anti-TB treatment. 6 However, the issue of timing of ART in patients presenting with TB has been a controversial. 7,8 Early or simultaneous initiation of ART may lead to high pill burden, clinical deterioration due to immune reconstitution inflammatory syndrome (IRIS), toxicity of drugs, reduced adherence, deterioration of illness, and death. On the other hand, delayed initiation of ART may lead to aggravation of illness and death. 7,8 However, there are also other modifying factors such as the site of the TB, the level of immune suppression, presence of other opportunistic infections, and resistance to anti-TB or anti-retroviral drugs. 7 –9 Therefore, the timing of treatment initiation is critically important for optimal treatment outcomes and patient care.
Despite the importance of optimal timing of TB/HIV co-treatment for clinicians and policy makers, currently, to the best of our knowledge, there is no such evidence of systematic review that clearly describes the effect of early initiation of ART in ART-naive people living with HIV-coinfected with TB. This study presents the effect of early initiation of ART among TB/HIV-coinfected patients on several treatment outcomes such as mortality, TB-IRIS, and drug toxicity.
Methods
Conduct of the Systematic Review and Search Strategy
The investigators conducted the current systematic review in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analyses statement (Supplemental Checklist S1). 10 A systematic electronic search of clinical trials in PubMed, Embase, Google Scholar, Science Direct, Medscape, and the Cochrane library were conducted by 3 authors without language, publication status, or date restrictions. Literature references were also scanned manually. The last date for electronic search was March 31, 2015.
The following topic-specific search terms including MeSH sub-headings and free text words were used (* stands for wild card symbol): TB, anti-TB, ART, anti-retrovi*, treat*, HAART, infect*, AIDS, HIV, opportunistic, start*, combin*, initiat*, antiviral*, agent*, acquired immun*, deficiency*, human immun*, early, treatment initiat*. Experts in the field were contacted via a standardized e-mail in order to retrieve unpublished studies.
Criteria for Considering Studies for This Systematic Review
Types of studies and participants
Randomized controlled clinical trials in which the participants consisted of HIV-infected persons with TB (pulmonary and extrapulmonary) enrolled for treatment of both diseases were included. The enrolled participants were treatment naive for both diseases initially.
Types of interventions
Trials that involve ART and TB treatment were included in the present systematic review. The participant’s time to initiate treatment for 1 or both of the infections might be at different level of care as defined by CD4 counts or World Health Organization clinical stages. The interventions involve early (within 2-4 weeks) and late (in 8-12 weeks) initiation of ART during TB treatment.
Outcome measures
Studies with the following outcomes were included in the systematic review: (1) success of TB treatment (cure from TB at the end of short-course therapy), (2) death from all causes, (3) responses to ART such as occurrence of new HIV-related events such as opportunistic infections other than TB, (4) virologic response, and (5) immunologic response such as changes in CD4 count.
Categorizing trials for quantitative and qualitative analyses
Data from trials with similar study design, outcome measures, intervention arms (early ART, within 2-4 weeks versus late ART, in 8-12 weeks), and patients characteristics were appropriate for pooled analysis and they were considered for quantitative analysis. When data were obtained from trials with selected patient characteristics (eg, HIV/AIDS with only TB meningitis) 8 and nature of data cannot be pooled for meta-analysis (eg, comparing integrated ART-TB treatment versus sequential ART-TB treatment), 11 the authors used the data from these trials for qualitative analysis.
Data Extraction
Based on title, key words, and abstract, 3 reviewers (S.M.A., A.A.R., and K.D.) selected the studies by applying the inclusion criteria. When there was any doubt whether to select a study or not, it was discussed to resolve it. The 3 reviewers (S.M.A., A.A.R., and K.D.) assessed the full versions of the selected articles. When disagreements about inclusion were not resolved by consensus, it was arranged to consult additional reviewer (A.D.). Figure 1 outlines the identification of studies for this systematic review and meta-analysis.
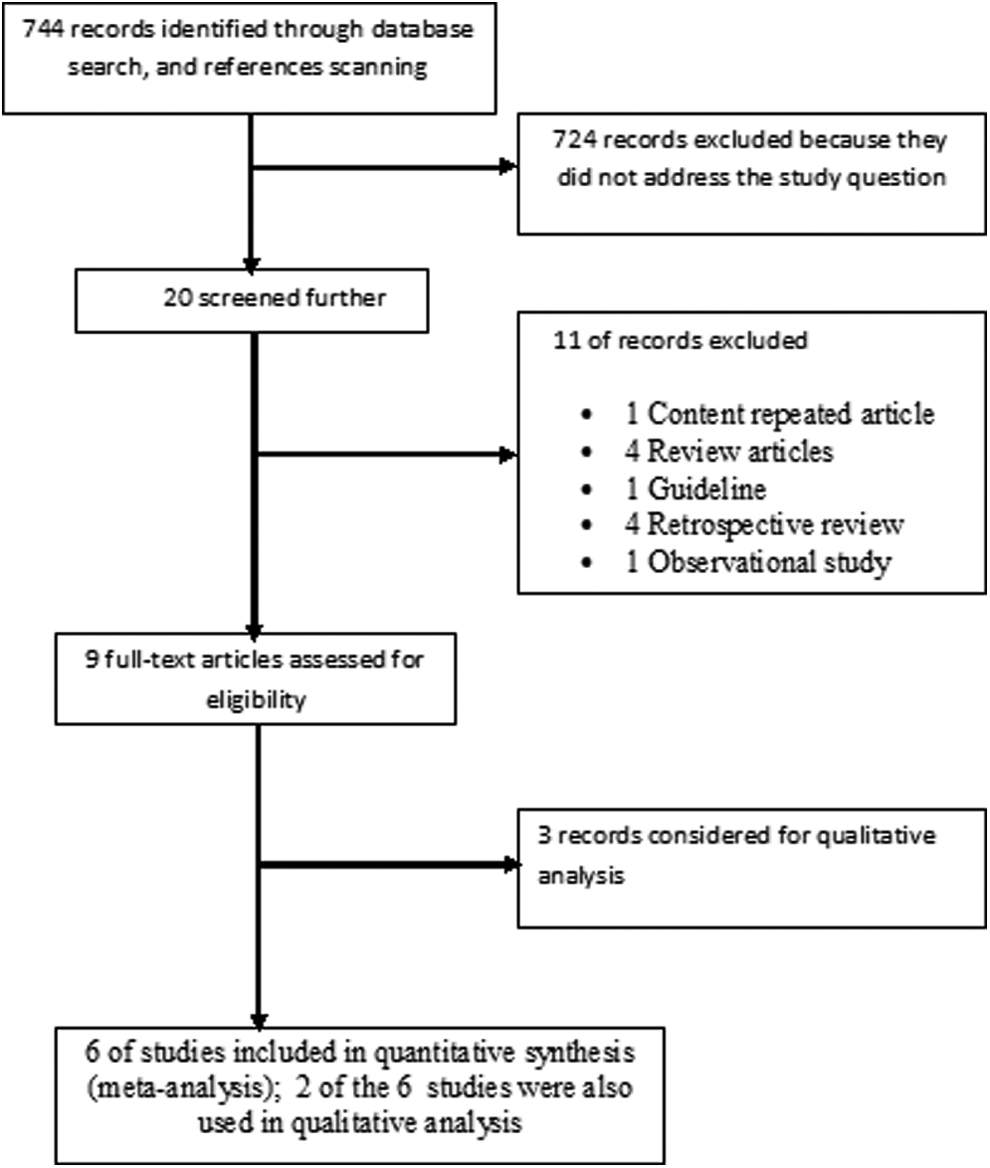
Flow diagram for study selection.
We did not blind the articles for authors, institution, or journal title. Three reviewers (S.M.A., A.A.R., and K.D.) independently rated the studies for methodological quality. Using a consensus meeting, disagreements between the 3 reviewers were discussed and resolved. Whenever a study did not contain sufficient information on methodological criteria or the information was unclear, the authors were contacted for additional information.
Three reviewers extracted data from the included studies. Any differences between the decisions of the reviewers were resolved by consensus. For each article, the following data were extracted using Excel 2007 spreadsheet: methods (design and settings), participants (age, sex, inclusion and exclusion criteria, status of infection), description of the interventions, outcome measures (primary and secondary outcomes), and findings.
Assessment of Risk of Bias in the Included Studies
We assessed the methodological quality of the included studies by using the criteria from the Cochrane Library included in the Review Manager software. 12 The criteria consists of 3 levels (unclear, low, and high risk of bias) and the rating on each item could be complemented by using quotes or paraphrasing of the study report as well as inclusion of the raters’ comments. We examined and rated the studies using the following criteria: generation of allocation sequence, allocation concealment, blinding of intervention, issues of incomplete data and loss to follow up, and other miscellaneous bias identified by the reviewers.
Assessment of Heterogeneity and Publication Bias
Heterogeneity, the variations among study outcomes, was checked by τ2 statistic, χ2 based Q test,
Publication bias was not assessed as tests for funnel plot asymmetry could not be used when there are fewer than 10 studies in the meta-analysis because test power is usually too low to distinguish chance from real asymmetry. 14
Sensitivity Analyses
Sensitivity test was performed to check the robustness of the main findings. It was done by excluding and including small studies and outliers (very different results from the rest of the studies) and by changing the pooled analysis between fixed and random effects models.
Statistical Analysis
All measures of effect were analyzed on an intention-to-treat bases. For each study outcome (all-cause mortality, TB cure rate, TB-IRIS, death related with TB-IRIS, drug toxicity, immunologic, and virologic response), we calculated the risk ratio (RR) and the corresponding 95% confidence interval (95% CI) as a measure of intervention effect. Since the outcomes, death, and development of adverse events, were unfavorable, a ratio less than unity indicates an outcome favoring the intervention. Where pooled effects were calculated, the Mantel-Haenszel method was used to estimate the RR using a fixed-effects model. When at least 2 trials assessed the effects of interventions and reported data on the similar outcome measures, pooling was considered. We conducted a formal statistical test for between studies variance and assessed whether the observed variability in effect sizes is greater than would be expected to occur by chance (sampling error). When the events in 1 of the arms (early or late ART) was 0, the software adjusts frequency of the event by adding 0.5. However, if the events in both arms are 0, the RR is not estimable.
Results
Description of Included Studies
Our electronic and reference scanning search yielded 744 abstracts and citations. Seven hundred and thirty-five studies were excluded and 9 articles were included. Six studies fulfilled the inclusion criteria for the meta-analysis 15 –20 (Table 1) and 5 for qualitative analysis. 8,11,15,17,21 All of the trials selected in the current systematic review had 2 or more outcomes—all-cause mortality rate, TB cure rate, TB-IRIS rate, drug toxicity, CD4 count, viral load, AIDS-defining illness, and death due to drug toxicity—that can be pooled for quantitative analysis.
Characteristics of Trials Included in the Meta-Analysis.
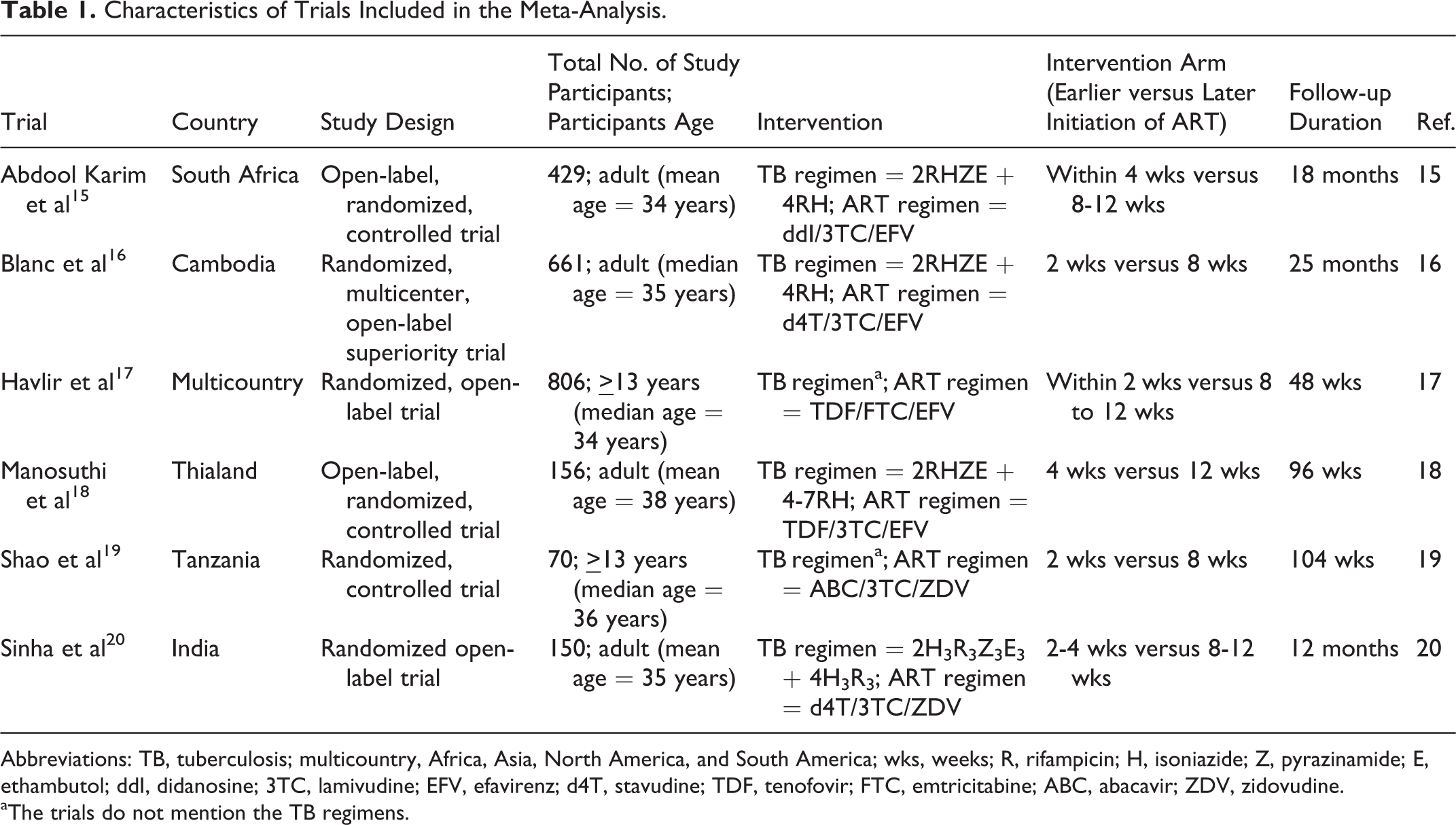
Abbreviations: TB, tuberculosis; multicountry, Africa, Asia, North America, and South America; wks, weeks; R, rifampicin; H, isoniazide; Z, pyrazinamide; E, ethambutol; ddI, didanosine; 3TC, lamivudine; EFV, efavirenz; d4T, stavudine; TDF, tenofovir; FTC, emtricitabine; ABC, abacavir; ZDV, zidovudine.
aThe trials do not mention the TB regimens.
The studies used for the quantitative analysis were 2 from African and 3 from Asian countries. One study was conducted in a multicountry settings (Africa, Asia, North America, and South America). A total of 2272 study participants were recruited in the 6 studies included in the current systematic review where each trial on average recruited 378 study participants (interquartile range [IQR] = 151-603). Five of the trials recruited patients diagnosed with both HIV/AIDS and TB (predominantly pulmonary) in both the early and later initiation arms of ART, whereas 1 trial recruited only pulmonary TB and HIV-coinfected individuals. The mean or median age of the study participants in the trials varied from 34 to 38 years. The study participants were followed-up for an average of 78 weeks (IQR = 54-99).
Quality Assessment of Trials
A summary of the risk of bias assessment of the controlled trials is found in Table 2. The extent of risk of bias was similar across the studies. In general, the trials had a low risk of bias in most areas: random sequence generation, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, and selective reporting. Unclear risk of bias was observed in allocation concealment of 2 trials. 15,17
Risk of Bias Assessment within the Trials.

aSelection bias.
bPerformance bias.
cDetection bias.
dAttrition bias.
eReporting bias.
Heterogeneity Assessment
Heterogeneity among the selected published trials was examined and the outcomes are presented in Table 3. Heterogeneity among studies was not observed in the trials used for the meta-analyses. The pooled RRs were calculated by the fixed-effects model, which assumes that there is no heterogeneity among the studies.
Statistics to Test Heterogeneity in the Meta-Analysis.

Abbreviations: TB, tuberculosis; IRIS, immune reconstitution inflammatory syndrome.
All-Cause Mortality in Early versus Later Initiation of ART
The pooled analysis of data provided by the 6 studies showed that all-cause mortality associated with early initiation of ART (within 2-4 weeks) in TB coinfected patients is lower than those who get ART later. The total number of cases in early and later initiation arms of the trials were 1153 and 1119, respectively. The RR was 0.78 (95% CI = 0.63-0.98), which favors the early initiation of ART in reducing all-cause mortality (Figure 2).

Risk of mortality: early versus late initiation of antiretroviral therapy (ART).
Among patients with CD4 counts of less than 50 cells/mm3, the rate of death or AIDS-defining illness in early ART was less than the late ART group: 8.5 cases per 100 person-years versus 26.3 per 100 person-years,
A study, not included in the pooled analysis (forest plot), compared the extent of all-cause mortality in earlier (76/127) versus later (70/126) initiation of ART in TB meningitis. The trial revealed that the early ART was not associated with improved 9-month survival (hazard ratio = 1.12; 95% CI = 0.81-1.55). 8 A recently published study compared the mortality rate of early ART initiation during TB treatment and delayed ART that involved the initiation of ART after completion of TB treatment. The research finding did not show the advantage of integrating ART and TB treatment in reducing all-cause mortality (3.0% in early ART versus 2.2% in delayed ART, relative risk 1.4, 95% CI = 0.8-2.3). 11
Tuberculosis Cure Rate in Early versus Later Initiation of ART
Three studies evaluate the TB response in the earlier versus later initiation of ART. The total number of cases in the early and later ART initiation arms of the trials were 634 and 606, respectively. There was no association between the time of ART initiation and TB cure rate (RR = 0.99, 95% CI = 0.81-1.07; Figure 3).

Tuberculosis cure rate: early versus late initiation of antiretroviral therapy (ART).
Tuberculosis IRIS in Earlier versus Later Initiation of ART
Rate of TB-IRIS
Six trials evaluate the association between TB-IRIS and the time of ART initiation during TB treatment. The total numbers of cases in the follow-up for IRIS were 1153 in earlier ART initiation groups and 1119 in later ART initiation groups. The RR was 2.19 (95% CI = 1.77-2.70), indicating low TB-IRIS among the later initiation of ART group (Figure 4).

Risk of tuberculosis-associated immune reconstitution inflammatory syndrome (TB-IRIS): early versus late initiation of antiretroviral therapy (ART).
Two trials evaluated the frequency of TB-IRIS in earlier versus later initiation of ART after stratifying study participants based on entry CD4 count (below or above 50/mm3). In both trials, earlier ART initiation in patients with entry CD4 count below 50/mm3 resulted in greater rate of TB-IRIS than in later ART initiation arms, 37.9% versus 11.4% (RR = 3.31; 95% CI:1.21-9.11), 15 and 18.8% versus 4.3% (RR = 4.41; 95% CI = 1.88-10.35). 21 The risk of developing TB-IRIS in earlier and later initiation arms was reduced in patients with entry CD4 level of greater than 50/mm3: earlier versus later initiation TB-IRIS rate of 16.4% versus 7.8% (RR = 2.11; 95% CI = 1.15-3.85) 15 and 5.8% versus 5% (RR = 1.15; 95% CI = 0.56-2.37; Figure 5). 21
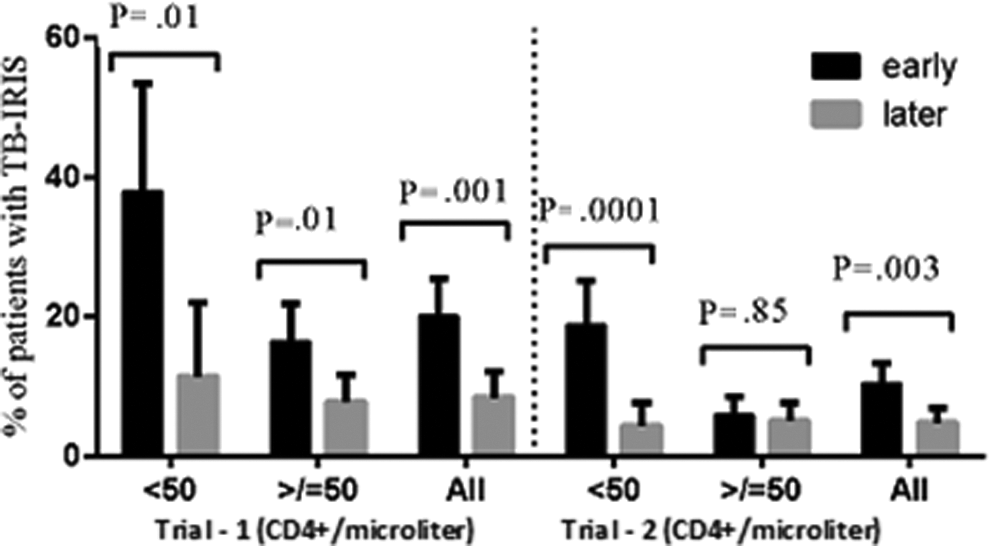
Percentage of patients developing tuberculosis-associated immune reconstitution inflammatory syndrome (TB-IRIS) categorized based on their entry CD4 count. Trial 1
14
and trial 2.
20
The error bars refer to the 95% confidence interval. *Pairs of bar graphs in CD4 count < 50 cells/mm3 of the earlier antiretroviral therapy (ART) initiation during TB treatment show higher rate of TB-IRIS (
A5221 STRIDE trial evaluated the time to TB-IRIS occurrence and its resolution in patients starting ART earlier and later during the TB treatment period. The median days of IRIS development in post-ART initiation in patients categorized under earlier and later ART arms were not significantly different—18 days (IQR = 10-59) in earlier ART versus 15 days (IQR: 7-23) in later ART groups (
Death due to TB-IRIS
Six trials assess the association between death due to IRIS and ART initiation time in TB/HIV-coinfected individuals. There were no any death reports due to IRIS in the 3 trials of both arms (earlier and later initiation of ART). 17,19,20 However, the pooled analysis showed that early initiation of ART was associated with higher risk of TB-IRIS-related death than later initiation of ART (RR = 6.94, 95% CI = 1.26-38.22), that is, 0.78% of study participants (9/1153) in earlier versus none of the study participants (0/1119) died in later ART initiation groups (Figure 6).

Risk of death due to tuberculosis-associated immune reconstitution inflammatory syndrome (TB-IRIS): early versus late initiation of antiretroviral therapy (ART).
Drug Safety in Earlier versus Later Initiation of ART
Rate of drug toxicity
Six trials compared the frequency of serious (grade 3 or 4) adverse drug events in their study arms: earlier and later initiation of ART in TB/HIV-coinfected patients during TB treatment. There was no association between ART initiation time and the rate of drug toxicity in TB/HIV-coinfected patients on TB treatment, RR 1.00 (95% CI = 0.93-1.08; Figure 7).

Risk of drug toxicity (3rd/4th grade/serious adverse event): early versus late initiation of antiretroviral therapy (ART).
A trial, not included in the pooled analysis, compared the severe adverse events in the earlier and later initiation of ART in TB meningitis. The percentage of patients with severe (grade 3 or 4) adverse events was high in both arms (90% in the earlier ART group and 89% in the deferred ART group; RR = 1.11; 95% CI = 0.94-1.28). 8
Death due to drug toxicity
Death due to drug toxicity related with the time of ART initiation was evaluated in 2 trials. There was no death report in 1 trial of both arms. 20 The other trial showed lack of association between the ART initiation time and death related with drug toxicity, RR = 0.40 (95% CI = 0.14-1.16). 16
Virological, Immunological, and Clinical Responses in Earlier versus Later Initiation of ART
Rate of undetectable viral load
The association between the time of ART initiation in patients who are on TB treatment and the rate of undetectable viral load was assessed in 2 trials. Earlier initiation of ART during TB treatment slightly favors in achieving higher rate of undetectable viral load compared with the later ART initiation, RR was 1.11 (95% CI = 1.02-1.20; Figure 8).

Undetectable viral load: early versus late initiation of antiretroviral therapy (ART).
Rate of viral load (less than 400 copies/mL)
Three trials assess the impact of earlier initiation of ART on lowering viral load to less than 400 copies/mL. There was no association between the time of ART initiation during TB treatment and achieving low viral load (less than 400 copies/mL; RR = 1.00, 95% CI = 0.96-1.04; Figure 9).

Viral load (less than 400 copies/mL): early versus late initiation of antiretroviral therapy (ART).
Rate of new AIDS-defining illness
The association between the time of ART initiation in patients who are on TB treatment and new AIDS-defining illness was assessed in 3 trials. 15,17,20 There was no association between the ART initiation time and the rate of developing new AIDS-defining illness (RR = 0.84, 95% CI = 0.60-1.18; Figure 10).

Risk of new AIDS-defining illness: early versus late initiation of antiretroviral therapy (ART).
Immunologic response
Five trials evaluated the immunologic response in relation to the time of ART initiation in TB/HIV-coinfected individuals. The response in 3 trials showed the absence of association between the immunologic response and the time of ART initiation, 16,17,19 but 2 studies showed an association between the immunologic response and the time of ART initiation 15,20 (Table 4).
Immunologic Response in Earlier versus Later Initiation of Antiretroviral Therapy.

Abbreviations: ns, not significant (
Sensitivity Analyses
Sensitivity analyses were conducted to examine the effect of including small studies and trials with different results from the rest (outliers), and the pooled RR estimates were stable. Sensitivity analyses for the pooled RR of outcomes are presented in Table 5.
Sensitivity Analyses for Pooled Outcomes.

Abbreviations: TB, tuberculosis; IRIS, immune reconstitution syndrome; CI, confidence interval; RR, risk ratio.
aOutlier was defined as study estimate out of the 95% CI of the pooled estimate.
bSmall study.
Discussion
Current treatment guidelines advocate earlier initiation of ART among TB/HIV-coinfected patients. There are however continued debates about the benefit and the risk of earlier and later initiation of the treatment in TB/HIV-coinfected patients. 22 There are no previous systematic reviews or meta-analyses to examine the benefits and risks of the 2 approaches of ART. We did a systematic review and meta-analysis on randomized control trials. The findings indicated a benefit of earlier initiation of ART on reduction of all-cause mortality. However, there is a clear evidence of increased incidence of TB-IRIS and death associated with it among the group that initiated ART earlier. These findings will have implications for clinical practice and policy.
The current analysis of published articles compared the treatment outcomes (the benefits and risks) of earlier initiation of ART during TB treatment with the later initiation. The main implication of our results is that ART needs to be initiated during TB treatment in such a time that can maximize survival with minimal risks, that is, within 2 to 4 weeks of the TB treatment initiation.
In the present meta-analysis, the risk of all-cause mortality in the later initiation of ART during TB treatment was higher than the earlier initiation arms. All-cause mortality risk in later ART initiation was 1.3 times more than early initiation: a relative risk reduction of 22% among patients at all CD4 levels starting ART earlier (Figure 2). The reduction of all-cause mortality in early ART initiation would be more than the figure mentioned if the RR was estimated in study participants with less than 50 CD4 cells/mm3. 15,17 In line with our analysis, retrospective chart reviews support the earlier initiation of ART in decreasing mortality among patients with TB and HIV. 23,24 Furthermore, our finding supports previous report warranting earlier ART in order to reduce the duration that patients have profound immunodeficiency, which results in a low baseline CD4 count–driven mortality in TB/HIV-coinfected patients. 4,25
In a previous systematic review, pooled analysis of outcomes (TB failure rate, relapse, and death during treatment) from trials having 2 arms—in patients treated with anti-TB medication and those patients on ART during TB treatment—showed that ART during TB treatment is associated with significantly lower rates of TB failure and relapse. 26,27 This evidence on improved TB outcomes by integrating ART and TB treatment is conclusive and imperative. Although trials compare treatment outcome in earlier versus later initiation of ART during TB treatments, in the period of TB treatment during which the ART needs to be initiated is not yet analyzed to identify the right time for optimum outcomes.
Apparently, ART during TB treatment reduces the rates of TB failure and relapse. 27 The current systematic review assessed TB cure rate in the earlier versus later ART during TB treatment and showed no statistically significant difference in the 2 arms. All the studies included in the pooled analysis did not show any difference in TB cure rate separately as well.
A systematic review using 16 studies reported the pooled cumulative incidence of TB-associated IRIS to be 15.7% out of 3459 patients, and 3.2% of those with TB-associated IRIS died. The same study 28 reflect the association between the risk of IRIS and CD4 count at the start of ART, where the risk is higher among patients with less than 50 cells/mm3. In the present study, the rate of TB-associated IRIS in earlier versus later ART during TB treatment was evaluated. The patients in earlier ART during TB treatment had 2.19 times more risk of developing TB-associated IRIS than those patients who started ART later during TB treatment (Figure 4) irrespective of their CD4 count. In low CD4 count at baseline, it has been evident that the rate of TB-associated IRIS was higher in patients starting ART earlier (Figure 5), and then the difference disappears in those patients with greater than 50 CD4 cells/mm3 during recruitment time. The interaction between TB-associated IRIS and CD4 count reported in the current systematic review is in agreement with the report by Müller and his colleague of pooled analysis. 28
The present study demonstrated that death due to TB-IRIS was higher in earlier ART during TB treatment (9 out of 1153 patients) compared with the later initiation (0 out of 1119 patient). The all-cause mortality in the earlier ART group (117 out of 1153 patients) was lower than the later ART group (145 out of 1119 patients). Tuberculosis-associated IRIS contributed to 7.7% death (9/117) in earlier ART arms. Hence, our quantitative and qualitative analyses support the earlier initiation of ART in patients on TB treatment by the net reduction in all-cause mortality.
Antiretroviral and anti-TB drugs have overlapping toxicity profiles—drug-induced liver injury, cutaneous reactions, renal impairment, neuropathy, and neuropsychiatric adverse effects—and drug interactions. 29,30 Had the advantage of integrated ART and anti-TB treatment in reducing mortality not outweighed adverse effects and risks of drug interaction, sequential therapy of TB and ART would have been recommended in avoiding drug interaction and overlapping adverse effects of antiretroviral and anti-TB drugs. 15 With this notion in mind, it is necessary to identify the time point to initiate ART during TB treatment for optimal outcomes. The current systematic review makes a pooled analysis of adverse events in earlier versus later ART during TB treatment and found that the rate of serious (grade 3 or 4) adverse was not different between earlier and later ART initiation during TB treatment, that is, equal risk (Figure 7). Death due to drug toxicity showed similar risk in earlier and later ART group, confirming that earlier initiation of ART benefits patients by reducing all-cause mortality without increasing the risk of adverse effects.
The current systematic review shows that immune response (change in CD4 count) during follow-up is improved in earlier ART during TB treatment than later ART group in 2 trials. 15,20 Whereas in the other trials, 16,17,19 the immune response is not different during follow-up among TB/HIV-coinfected patients initiating ART at different times. Hence, the association between immune response and the time of ART initiation during TB treatment among the overall study participants could not be well established (Table 4). This might be related with the difference in patient characteristics in each trial that might influence immunologic response after ART—for instance, low CD4 reconstitution in old age. 31
The current systematic review may have limitations. Firstly, we compared the outcomes in the earlier versus later ART groups during TB treatment with a heterogeneous array of initiation time of ART, that is, 2 to 4 weeks as earlier versus 8 to 12 weeks as later ART groups. The longer period in each category possibly obscure some important differences in the rate of outcome measure between the categories. We would like to declare that 1 trial from Ethiopia is under the process of publication and the details of the results were not disclosed by authors. All in all, there were few studies because of which assessment on publication bias was not done. 14 However, the authors ran a sensitivity analyses to determine whether the studies introduce bias into the result of the systematic analysis (Table 5).
The current systematic review reflected the benefits and risks of time of ART initiation during TB treatment in HIV/AIDS patients developing predominantly pulmonary TB. It appears that initiating ART earlier during TB treatment reduces all-cause mortality and outweighs the risks of TB-IRIS. The review synthesizes the data from trials conducted in both resource-limited setting and developed countries, and its findings can be informative for TB/HIV co-treatment globally. Specifically, the study suggested the earlier initiation of ART in patients with low CD4 to reduce mortality though TB-IRIS proportionally increases. All the findings call for the importance of clinical judgment as to the importance of initiating ART earlier against the risk of TB-IRIS. This demands health service in resource-limited countries to be equipped with facilities assessing immunologic status of patients and health care providers able to diagnose and manage TB-IRIS properly via appropriate training and continual medical update and education.
Our study pooled trials based on study participants composed mainly of pulmonary TB/HIV-coinfection treatment. 15 –20 Therefore, the study findings could be informative only about the benefits and risk of earlier initiation of ART in the aforementioned cases to practitioners and TB/HIV control officers. To extend the reduced all-cause mortality report from the current study toward the routine clinical service, authors recommend operational and implementation research. In addition, studies on time of ART during TB treatment in patients with extrapulmonary TB/HIV, multidrug- or extensively drug-resistant TB/HIV coinfection are warranted before extending the findings of the current systematic review to these populations.
Conclusion
The present systematic review presents conclusive evidence on reduction of all-cause mortality by early initiation of ART during TB treatment and increased risk of TB-IRIS and death associated with TB-IRIS in patients with TB/HIV coinfection. The TB cure rate and achieving lower viral load (<400 copies/mL) and development of new AIDS-defining illness were not associated with the time of ART initiation, and the change in immunologic response is inconclusive. Health care facilities are recommended to initiate ART earlier in TB/HIV-coinfected patients with a better IRIS management practice to maintain the aforementioned benefits with minimal TB-IRIS risks.
Footnotes
Acknowledgments
The authors would like to thank the librarians of Addis Ababa University for providing articles.
Authors’ Note
KD conceived the idea and designed the study; SMA, KD, and AAR performed data extraction and analysis; KD and SMA wrote the first draft of the manuscript; and SB, DD, TA, MT, and AD critically reviewed the manuscript. SMA and KD equally contributed to this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors declare that there is no financial support for the current study. KD is supported by a Wellcome Trust Research Training Fellowship in Public Health and Tropical Medicine (grant number 099876).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
