Abstract
Tumor necrosis factor-alpha inhibitors (TNF-αi’s) such as Infliximab are effective treatments for various autoimmune diseases. However, in rare instances, they have been implicated in central nervous system demyelination. Typically, this side effect is seen within the first year of medication exposure. This case reports on the rare occurrence of a central demyelinating lesion secondary to Infliximab after several years of uncomplicated use. A 39-year-old, right-handed male with a history of ulcerative colitis (UC) was being maintained on infliximab therapy for several years before presenting to the emergency room. He reported weeks of intermittent, worsening left-sided extremity paresthesia and weakness. Initial workup ruled-out a stroke, but subsequent MRI revealed a strange right-sided cortical enhancement that anatomically corresponded to the patients’ complaints. Ruling out other possibilities, the patient’s TNF-αi medication became a concern. Treatment with corticosteroids and suspension of the patient’s infliximab enabled a swift improvement and gradual full recovery. The patient’s UC medication was subsequently changed as he returned to baseline. This case demonstrates a rare complication of a commonly prescribed medication that appeared well outside the timeframe of similar other case reports, highlighting the importance of considering TNF-αi side-effects as possible contributory factors to disease states even after many years of relative stability.
Introduction
Infliximab is a chimeric IgG1 monoclonal antibody that neutralizes tumor necrosis factor-alpha (TNF-α), a key pro-inflammatory cytokine. By blocking TNF-α’s interaction with its receptors, Infliximab suppresses a cascade of inflammatory cytokines and subsequent immune cell recruitment, thereby tempering autoimmune-mediated tissue damage. 1 It has become an important therapy for conditions such as rheumatoid arthritis, Crohn’s disease, and ulcerative colitis (UC). Common adverse effects of Infliximab include infusion reactions, infections, cytopenia, and hepatotoxicity. 1 Rarer but important complications include autoimmune phenomena, such as lupus-like syndromes, psoriasis, and demyelinating disorder like multiple sclerosis (MS). 2 These demyelinating adverse events are particularly significant because they can mimic more common auto-immune conditions in clinical and radiologic presentation as well as potential severity, delaying appropriate diagnosis, and treatment if not included among clinicians’ initial differentials.
The relationship between TNF-αi’s and demyelination is complex. Paradoxically, although TNF-α is pro-inflammatory, blocking TNF-α can still precipitate central nervous system (CNS) demyelination via inflammatory signaling in alternative pathways for susceptible individuals.3,4 Sporadic instances have been reported of MS and optic neuritis demyelinating syndromes emerging during anti-TNF therapy in patients with no prior neurologic disease.2,5-7 There are other published reports detailing these instances related to the specific TNF-α inhibitor infliximab, but most describe the typical side-effects from infliximab occurring within the first year of treatment, with the average onset reported to be within ~5 months of use, but with some reports extending up to 6 years.8-11
This pro-inflammatory phenomenon—termed TNF-α Inhibitor-Associated Demyelinating Disease—although rare, poses a potentially serious concern given the widespread use of TNF-αi’s like infliximab, and this disease state mimicking more common pathologies. Rapid recognition of disease enables prompt treatment, thus increased awareness of the potential for this rare-side effect even years into therapy is important to understand. Herein, we detail a case of a 39-year-old male with a history of UC managed with infliximab who reports several weeks of progressive, lateralized numbness and tingling in his extremities that eventually acutely worsened and led to his presentation in the emergency department. Ruling out other possible causes, it is concluded that he has developed demyelinating CNS disease secondary to his long-term infliximab therapy. Suspending this medication and treating his symptoms with steroids led to resolution of his symptoms.
Case Presentation
A 39-year-old, right-handed male with a history of UC, depression, anxiety, hyperlipidemia, obesity, and chronic low back pain presented to the emergency department reporting an acute worsening of left-hand weakness and a near-syncopal episode while at work. He explained that in the process of stocking shelves, he felt a sudden onset of weakness in his left hand, which quickly progressed such that he was unable to move his left fingers to grip anything. Thereafter, a wave of light-headedness came over him and he lowered himself to the ground before briefly losing consciousness. He did not hit his head.
Upon further interview, the patient recalled a gradual onset of left leg numbness and weakness that initially began ~3 to 4 weeks prior to his presentation. He sought treatment in the interim through an online appointment with his primary care physician (PCP) and was told his symptoms were likely due to sciatica. However, his symptoms continued to worsen and, in the days leading up to his presentation in the Emergency Department (ED), he developed additional clumsiness and weakness in his left hand. He denied any visual disturbances, vertigo, or incontinence during this time.
His presenting vitals were stable. Physical exam revealed a 4/5 weakness in the left lower extremity. He was otherwise negative for any other focal deficits or upper motor neuron signs. An acute stroke was initially considered; however, head Computed Tomography (CT) showed no acute hemorrhage or infarct. An MRI of the brain (Figure 1) was then obtained that revealed a small Fluid-Attenuated Inversion Recovery (FLAIR) (Figure 2) hyperintense lesion in the right frontal lobe cortex with surrounding edema and faint enhancement, but no diffusion restriction to suggest acute ischemia. This lesion was located around the right central sulcus, corresponding to the patient’s left-sided symptoms. No additional lesions were seen elsewhere in the brain. The radiology impression was that this focal cortical lesion could represent a demyelinating process like MS, an evolving infection, a postinfectious demyelinating encephalomyelitis (ie, Acute Disseminated Encephalomyelitis (ADEM)).
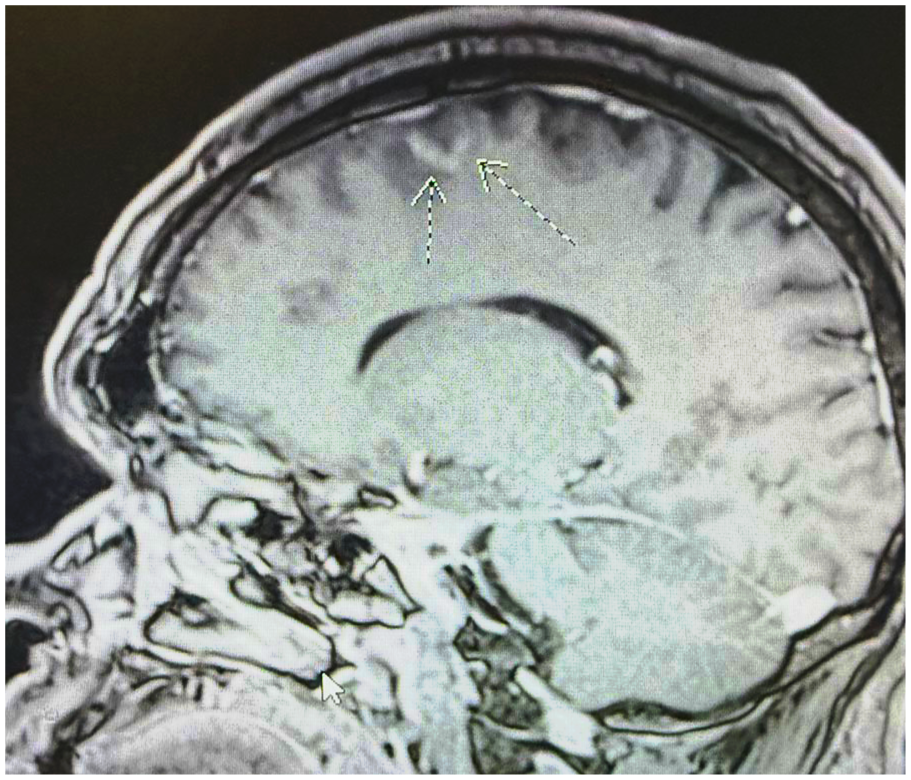
Lesion on MRI: T1 sagittal view. Focal slightly expansile T1/FLAIR hyperintensity involving cortex and juxtacortical white matter at the anterior aspect of right superior frontal gyrus with associated curvilinear juxtacortical enhancement.
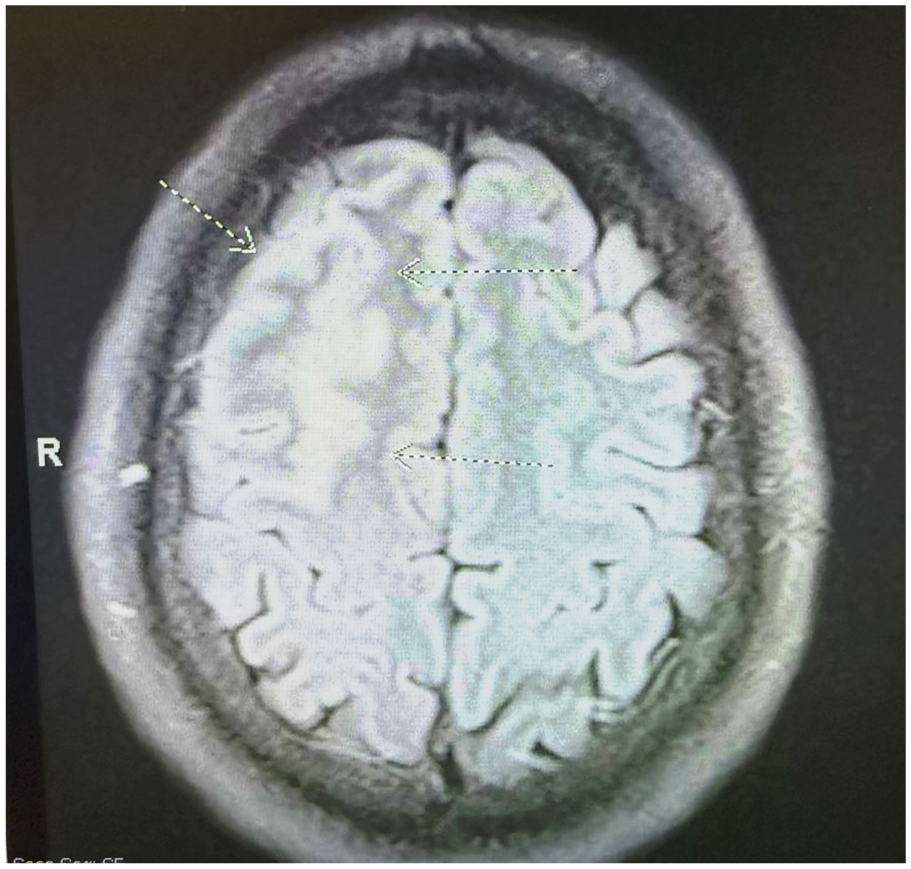
Lesion on MRI: T2 FLAIR view. Focal slightly expansile T1/FLAIR hyperintensity involving cortex and juxtacortical white matter at the anterior aspect of right superior frontal gyrus with associated curvilinear juxtacortical enhancement.
A lumbar puncture was performed to further distinguish between central inflammation or infection. Cerebrospinal fluid analysis demonstrated a normal opening pressure, an absence of white blood cells, normal protein and glucose levels, and was negative for infectious pathogens (including HSV, VZV, JC virus). Oligoclonal bands were not detected (Table 1). These results made an infectious encephalitis or typical MS less likely. Given the context and the patient’s long-term TNF-α inhibitor therapy, a rare infliximab-induced demyelinating syndrome became a leading consideration.
Laboratory Values.

Reactive Leukocytosis from the steroids started the previous day.
is indicating Relevant Labs for Diagnosis.
The patients UC had been in remission for 5+ years on maintenance infliximab infusions (5 mg/kg every 8 weeks) with no prior side effects. Interestingly, he was initially trialed on the first-line drug mesalamine but experienced side effects such as rash and worsened symptoms which led to him starting the second-line agent infliximab. Additionally, the patient reported that 2 of his children had been recently sick with upper respiratory infections—1 several weeks ago and the other still currently sick at home. A Covid-19 test was negative. Although he never felt ill himself, he reported more frequent sneezing in the last few days. He also reported episodic dizziness for the past 1+ month, as well as a long-term history of occasional headaches without nausea or sensitivity to light or sound. No recent travel or recent insect bites were reported.
Following neurology consultation, 5 days of intravenous methylprednisolone (1 g daily) was started to address possible autoimmune demyelination. The patient’s regular infliximab infusions were held considering the possible pathological relationship and taking into account his lack of UC flares for several years.
By the second day, the patient experienced significant improvement of his symptoms. His ambulatory ability was improved enough for him to walk to the bathroom unassisted. He was found to still have slight 4+/5 LLE weakness in knee flexion, but was otherwise normal. Repeat CBC demonstrated an increased WBC to 12.2, all other labs were still normal. His CSF fluid culture was negative, and CSF protein and glucose were normal. Notably, IgG levels in the CSF were elevated suggesting an active inflammatory process with a demyelinating lesion suspected as the primary cause.
Over the next 2 to 3 days on steroid treatment, the patient’s neurologic status stabilized further—strength in the left hand and leg fully improved and maintained at 5/5 strength, and his subjective sensory symptoms diminished. Home steroid dosing was planned for completion of his 5-day course. Before discharge, the case was thoroughly reviewed with multiple consulting physicians. It was felt that the demyelinating CNS lesion was likely precipitated by infliximab given the temporal association of improvement with cessation and exclusion of other causes. Following discussion with a consulting gastroenterologist, Infliximab was discontinued indefinitely and the alternative Vedolizumab was recommended. Given his improvement on steroids, the patient was discharged home with a cortisone taper and scheduled for outpatient follow-up.
At the 10-day follow-up visit, the patient reported a slight rebound of his symptoms similar to those that he first presented with in the ED. He continued to experience left foot and hand numbness and tingling, slight left-sided weakness, and reported minor speech problems, and intermittent lightheadedness. He had made lifestyle changes including more frequent hydration and taking positional changes and had not experienced another near-syncopal episode.
At day 14, the patient reached out to a telehealth physician regarding a pounding, constant 9/10 headache that he had reportedly been experiencing ever since his hospital discharge. He had a history of migraines, but this one was unrelenting. However, he confirmed resolution of his prior symptoms of left-sided weakness and numbness and was prescribed sumatriptan and ondansetron for his headache.
The 3-week in-patient follow-up visit demonstrated a normal neurological exam. He reported only mild residual, intermittent tingling in his left foot. Follow-up MRI of the brain (~6 weeks after the initial) showed reduction of the prior lesion’s edema and no new demyelinating lesions. A full auto-immune encephalitis panel returned completely negative for any known disease markers, consistent with the working diagnosis of a TNF-α inhibitor-induced demyelinating syndrome. The patient was therefore advised to avoid TNF-αi’s in the future. For his UC, gastroenterology initiated a switch to vedolizumab as an alternative therapy. At this last visit, the patient had near-fully returned to his baseline and remained stable with no relapse of demyelination or colitis.
Discussion
TNF-α plays a dual role in the body not only contributing to inflammation but also modulating complex pathways of immune response. Blocking TNF-α does not simply stop inflammation altogether, and is paradoxically known to trigger rare autoimmune-mediated demyelination by an unknown mechanism. Proposed theories include
Altered Cytokine Balance: TNF-α inhibition skews the immune environment, possibly upregulating other cytokines (like interferon gamma) that drive autoimmunity.
Enhanced Autoreactive T-Cells: Prolonged TNF-α blockade may remove a constraint on autoreactive T-lymphocytes, allowing them to attack myelin in the CNS.
Mimicry and Unintended Targets: The anti-TNF agent or the immune complexes formed could expose or mimic CNS antigens, precipitating an MS-like pathogenic process. Notably, TNF-αi’s are contraindicated in patients with MS, as studies have shown they can exacerbate demyelination.2-12
This patient had no prior neurologic disease and developed a demyelinating lesion while on long-term infliximab, was not positive for MS, and had full resolution of symptoms with medication cessation and steroid treatment, leading to the diagnosis of TNF-α inhibitor associated demyelination. This phenomenon of TNF-αi’s simultaneously being highly effective for systemic inflammation yet capable of inducing neurologic autoimmunity has been described as a “double-edged sword” of anti-TNF therapy. 11 It is unclear why only a small subset of patients develops this complication; etiology is likely multivariate and related to developed or genetic susceptibility and the interplay of TNF-α in maintaining self-tolerance in the CNS.
Another intriguing possibility is the facilitation of viral illness to this patient’s rare auto-immune process. Among the scant literature, Stüve and Zamvil noted a common temporal relationship between the onset of TNF-αi side effects and either infection or vaccination. 13 In line with this thinking, because it was noted that this patient had sick kids at home during the course of his symptom presentation, viral exposure could have facilitated an immune over-activation into an auto-inflammatory process. This etiology is conjecture but correlates with the sudden emergence of symptoms after total stability for several years. 13 Acknowledgment of this association in time may lend support to the theory of infectious molecular mimicry contributing to the risk of developed autoimmunity while on TNF-α inhibitors.
Cases of TNF-α inhibitor-associated demyelination have been reported with various agents in this class besides Infliximab, including etanercept and adalimumab. The clinical presentations range from isolated optic neuritis, to transverse myelitis, to full-blown MS. 12 In a case series by Gharib et al, 3 patients on TNF blockers developed MS-like demyelination. Interestingly, 1 case involved a patient also on infliximab who presented with limb numbness and weakness very similar to our patient. In this case series, discontinuation of the TNF-αi led to symptom improvement in all cases. However, 2 patients had recurrent demyelinating attacks afterward, suggesting that in some individuals TNF-αi’s may unleash an underlying propensity for demyelinating syndromes. 12
There are no formal guidelines for treating TNF inhibitor-induced demyelination, so management is derived from MS relapse treatment protocols and case reports. The first step is to immediately discontinue the offending TNF-αi once an adverse event is suspected. In many cases, this alone leads to stabilization or improvement of symptoms. 12 High-dose corticosteroids are also commonly employed, analogous to treating an MS flare or ADEM. Our patient responded well to IV methylprednisolone, which likely hastened the recovery by reducing CNS inflammation. In instances where there is evidence of ongoing disease, to minimize delays in treatment, MS-directed therapies such as steroids or even plasma exchange can be considered while a definitive diagnosis is sought.
Two different meta-analysis have concluded that the increased risk for developing an inflammatory CNS disease due to TNF-α therapy is between 36% and 38%.7,14 Given the increasing use of TNF-αi’s, increased awareness of this risk is important for early detection and treatment of this rare side effect. The prognosis of TNF-αi demyelination is variable but generally favorable if recognized promptly. From a systemic standpoint, cessation of TNF-α therapy can risk flare of the underlying disease (eg, colitis), but this can be managed by switching to a different class of immunomodulator. In our case, vedolizumab was chosen for UC, as it targets gut lymphocyte trafficking and has not been associated with similar demyelination events. Patients should be counseled that re-challenge with a TNF-αi is not advised after a demyelinating event, given the high likelihood of symptom recurrence with continued TNF-α suppression.
Limitations of this case study include the single sample in a patient with various other comorbidities. Considering the association of this case with familial respiratory infections, acknowledgment of the possible contribution of viral illness in the pathophysiology is necessary. Further research is needed to elucidate whether medication exposure, genetics, or viral insults are mediating factors for developing TNF-αi demyelinating disease. This case contributes to the scant case-reports of such events and may prove valuable for such investigations.
Conclusion
This case details a rare neurological side-effect of infliximab. Although uncommon, TNF-α inhibitor-associated demyelinating disease is an important adverse effect that clinicians should be aware of. This case highlights how even stable patients maintained on TNF-αi’s like infliximab for many years can develop the diagnosis of a TNF-α demyelinating syndrome. Early recognition and intervention is critical to achieve a favorable neurological outcome. This report adds to evidence that TNF-α plays a nuanced role in CNS immune homeostasis, demonstrating how aberrant auto-immune activation can be triggered by TNF-α inhibitors while on long-term therapy.
Footnotes
Acknowledgements
I would like to thank Kaiser Permanente San Diego Medical Center for providing access to and support for this case report.
Authors’ Note
No prior presentations, abstracts, or publications have been made involving this case.
Ethics Considerations
Ethical approval to report this case series was obtained from Western University of Health Sciences IRB (25RFD023).
Consent for Publication
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
