Abstract
This report describes a case of facial hyperpigmentation in a patient with Crohn’s disease receiving adalimumab, a tumor necrosis factor (TNF)-alpha inhibitor. The onset of hyperpigmentation coincided with adalimumab administration, and its discontinuation resulted in significant improvement. Histopathological findings suggest a postinflammatory process at the dermo-epidermal junction. However, the precise mechanism remains unclear.
Keywords
Introduction
Adalimumab is an anti-tumor necrosis factor (TNF)-alpha agent, commonly used to treat inflammatory diseases, including Crohn’s disease. Tumor necrosis factor-alpha inhibitors are associated with a high incidence of cutaneous adverse events. However, facial hyperpigmentation secondary to adalimumab is not a documented adverse effect. 1 This report describes a case of facial hyperpigmentation in a patient with Crohn’s disease, receiving adalimumab therapy.
Case Presentation
A 50-year-old man with a 20-year history of Crohn’s disease had been on azathioprine therapy for 14 years. He underwent ileocolic resection surgery 2 years ago. Adalimumab therapy was initiated 7 months ago, in conjunction with azathioprine, due to uncontrolled bowel disease. He reported persistent hyperpigmentation of the face, which had been progressively worsening over the past 3 months. Importantly, he denied any history of applying topical products to his face. The physical examination revealed diffuse, symmetrical, dark brown to grayish hyperpigmentation of the face, except for the infraorbital region and the nose (Figure 1A). No oral, nail, or other skin lesions were noted. No evidence of hyperpigmentation was observed beyond the face, particularly on other photo-exposed areas such as the neck and hands. The standard biological investigation yielded normal results: antinuclear antibodies were negative (<1: 80), endocrine workup including vitamin B12 was 320 pg/ml (normal range >200 pg/ml), and cortisol at 8 a.m. was at 450 nmol/l (250-700 nmol/l). A skin biopsy taken from the hyperpigmented area showed a focal dyskeratotic epidermis with spongiosis and keratinocyte necrosis. The dermis showed a perivascular and peri-annexal lymphocytic infiltrate with pigment incontinence (Figure 1B).

(A) Symmetrical dark-brown to grayish hyperpigmentation of the face; note the sparing of the infraorbital region and the nose. (B) Focal dyskeratotic epidermis with spongiosis and keratinocyte necrosis. Perivascular and peri-annexal lymphocytic infiltrate in the dermis with pigment incontinence (Hematoxylin and Eosin, ×10). (C) Improvement in the face after the discontinuation of adalimumab.
After discontinuing both adalimumab and azathioprine, following the achievement of clinical and endoscopic remission of Crohn’s disease, and starting the application of topical steroids along with photoprotection measures, a marked regression of hyperpigmentation was observed after a 1-year interruption (Figure 1C). The total score of the Naranjo algorithm, which corresponds to the probability of an adverse drug reaction, was between 5 and 8, indicating a probable reaction. There was no discernible worsening or recurrence of hyperpigmentation 2 years after the reintroduction of azathioprine, which enhances the imputability of adalumimab.
Discussion
Tumor necrosis factor-alpha inhibitors are known to cause a range of cutaneous adverse events, affecting approximately 25% of patients. 1 The most commonly reported adverse events include xerosis, eczema, psoriasis, palmoplantar pustulosis, skin infections, alopecia, and skin tumors. 1 Adalimumab, a fully human immunoglobulin (IgG1) monoclonal antibody, is purportedly associated with fewer adverse effects compared to other agents in its class.2,3 However, facial hyperpigmentation as a side effect of adalimumab is particularly rare.3,4
In the present case, a strong temporal relationship was observed between the administration of adalimumab and the onset of facial hyperpigmentation. In addition, discontinuation of adalimumab led to regression of the hyperpigmentation, further supporting a causal relationship. The likelihood of azathioprine being the cause of hyperpigmentation is low, as it had been consistently administered for several years prior to the incident, and its reintroduction did not coincide with a recurrence of hyperpigmentation.
There are only a few reports in the medical literature of isolated facial pigmentation associated with adalimumab. This has been documented in three patients with psoriasis (two of whom had previously used methotrexate)2 -4 and one patient with Crohn’s disease. 5 None of these patients had pre-existing facial lesions. Interestingly, two cases also documented sparing of the infraorbital region similar to our patient.3,5 However, the hyperpigmentation remained unchanged following the withdrawal of adalimumab. Table 1 summarizes the epidemiological, histopathological, and clinical features of these cases.
Epidemiological, Histological, and Clinical Features of Adalimumab-Induced Hyperpigmentation.
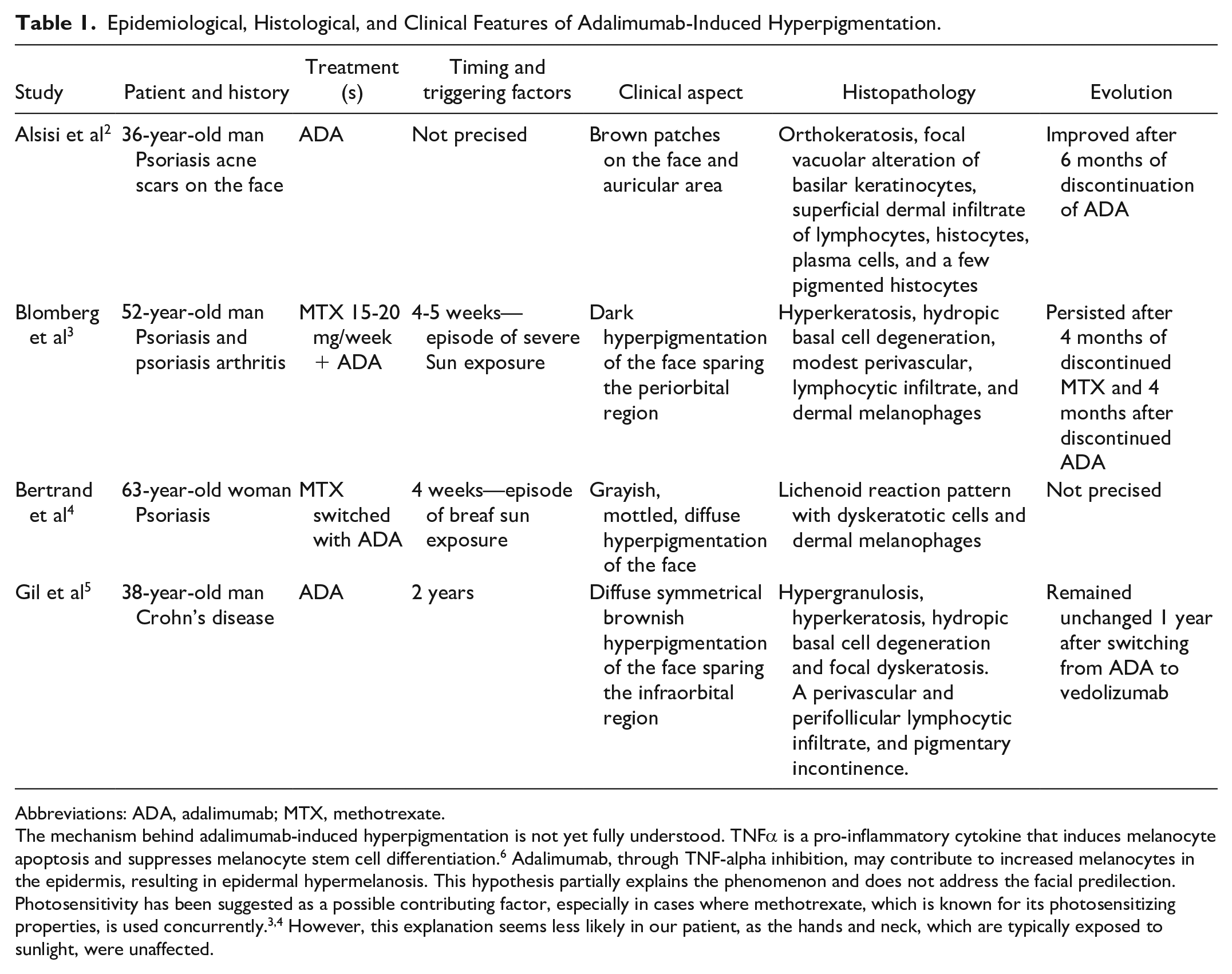
Abbreviations: ADA, adalimumab; MTX, methotrexate.
The mechanism behind adalimumab-induced hyperpigmentation is not yet fully understood. TNFα is a pro-inflammatory cytokine that induces melanocyte apoptosis and suppresses melanocyte stem cell differentiation. 6 Adalimumab, through TNF-alpha inhibition, may contribute to increased melanocytes in the epidermis, resulting in epidermal hypermelanosis. This hypothesis partially explains the phenomenon and does not address the facial predilection. Photosensitivity has been suggested as a possible contributing factor, especially in cases where methotrexate, which is known for its photosensitizing properties, is used concurrently.3,4 However, this explanation seems less likely in our patient, as the hands and neck, which are typically exposed to sunlight, were unaffected.
All cases, including the present one, show a common histopathological pattern characterized by the presence of interface dermatitis and inflammatory infiltrates within the dermis.2,3,5 A lichenoid pattern has been reported in one case. 4 These findings suggest the occurrence of an inflammatory process affecting the dermo-epidermal junction, resulting in pigment incontinence, which is more consistent with a mechanism of post-inflammatory hyperpigmentation. However, the specific involvement of the face remains to be elucidated.
This report presents a rare case of isolated hyperpigmentation of the face as a cutaneous adverse reaction to adalimumab. The significant regression of hyperpigmentation following the discontinuation of adalimumab was a distinctive feature of this case. Further investigations are needed to elucidate the underlying mechanisms of such reactions and to accurately assess their clinical significance.
Footnotes
Author Contributions
The authors confirm contribution to the paper as follows: study conception and design MT and AS; data collection: MM; analysis and interpretation of results: AS, IC, and MM; draft manuscript preparation: MT and AS. All authors contributed equally to the manuscript and read and approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Approval was obtained from the local ethics committee
Informed Consent
The patient included in the article gave informed consent to publish his case details and pictures.
Data Availability Statement
Authors agree to make data and materials supporting the results or analyses presented in their paper available upon reasonable request from the corresponding author
