Abstract
Ulcerative colitis (UC) pathogenesis includes the altered gut microbiota, environmental factors, and human immune and genetic predisposition. Recently, its association with reduced bifidobacteria quantity in the microbiota is reported. Xyloglucan, a plant-based prebiotic oligosaccharide, causes increase in bifidobacteria quantity. In this article, we share the results of our UC cases treated by intracolonic single-dose administration of Bifidobacterium animalis subsp. lactis and xyloglucan combination. Intracolonic single-dose administration of 200 billion colony-forming units (CFUs) of B. animalis subsp. lactis and 4 g of xyloglucan combination was administrated to 10 severe UC patients, who were either unresponsive or had inadequate response to treatment. All patients continued treatment after the procedure. Treatment responses were evaluated by colonoscopic, laboratory, and clinical examination after 6 weeks. Intracolonic single-dose administration of B. animalis subsp. lactis and xyloglucan was found effective in the mucosal healing and resolution of colonic symptoms in UC patients. Intracolonic administration of B. animalis subsp. lactis and xyloglucan in UC is a new single-strain and strain-specific prebiotic combination method. It is easy to apply and has no observable side effect. Its effectiveness on mucosal healing could be attributed to the enhancement of non-stimulatory status and biodiversity in colonic mucosa. Nonetheless, it is still necessary to develop diagnostic strategies to determine the patients to whom this method would be the most applicable.
Introduction
Alterations in gut microbiota (dysbiosis) and reduced bifidobacteria levels in the gastrointestinal tract of ulcerative colitis (UC) patients were reported in the literature. 1 Despite the existence of dysbiosis in the intestinal microbiota of UC patients, it is unclear whether it is the cause or the effect of UC. The transfer of bifidobacteria by breast feeding and oral probiotic may contribute to normal mucosal homeostasis. 2 Oral probiotic administration might also have a complementary role in treating and preventing UC flares. 3 Recent studies show the association between inflammatory bowel disease (IBD) activity and low diversity of gut microbiome.2,3 Fecal microbiota transplantation (FMT) was developed in this context for IBD patients. 4 However, because of the high degree of uncertainty on what gut microbiome profiles may emerge in the short and long terms, FMT may have some potential adverse effects. 5 Intracolonic single-dose administration of Bifidobacterium animalis subsp. lactis BB-12 and xyloglucan combination (ICB) is a new single-strain microbial and strain-specific prebiotic application that has no adverse events for IBD patients (Figures 1 and 2).6,7 Xyloglucan is a plant polymer that consists of cellulose, which carries xylose and galactosyl–xylose contents. 8 Xyloglucan provides a physical barrier that protects the stability of mucosal cells against various damaging agents, such as microorganisms, allergens, and pro-inflammatory compounds in human gut system. 8 B. animalis subsp. lactis was shown to increase up to 30 times in the presence of xylo-oligosaccharides (XOSs). 9 Our work reports the results of UC patients who were treated by single-dose colonoscopic application of B. animalis subsp. lactis and xyloglucan combination. Our patients were unresponsive or had inadequate responses to the prior treatment regimens.

Mucosal healing (left) 1 month after ICB and xyloglucan administration in treatment-unresponsive ulcerative left-sided colitis (right; patient 7).
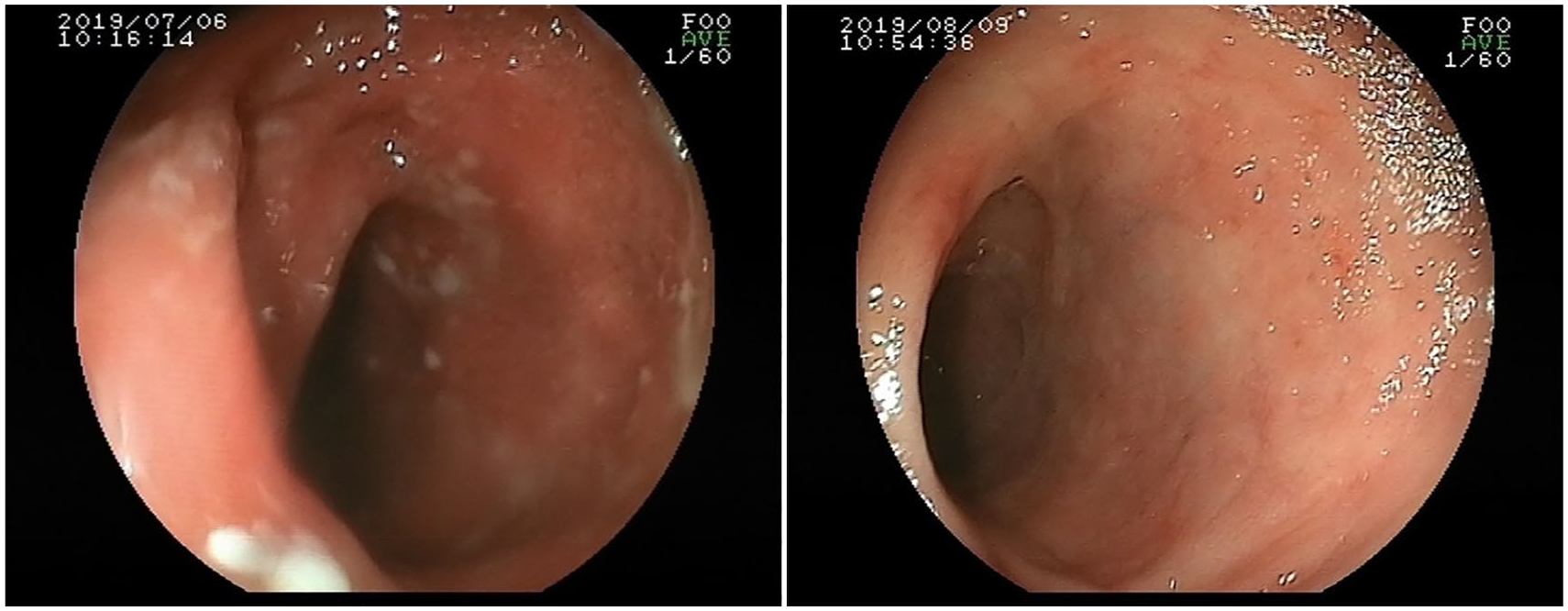
Mucosal healing (right) 1 month after ICB and xyloglucan administration in treatment-unresponsive ulcerative proctosigmoid colitis (left; patient 6).
Methods
Patients
The research was conducted with a single-center retrospective case series in 2019. We report the results of 10 UC patients treated by single-dose ICB administration. The symptoms and clinical and endoscopic evaluations were assessed after 6 weeks of intervention. The age range was between 11 and 56 years (30 ± 11.723). Six patients were female. All the patients had continued medical therapy after the administration (seven patients took 5-acetylsalicylic acid (5-ASA) + Azathiopurine and three took vedolizumab treatment).
Clinical material
Before the colonoscopy procedure, 200 billion colony-forming units (CFUs) of B. animalis subsp. lactis BB-12 (Chr. Hansen) and 4 g of xyloglucan were dissolved in a 250-mL saline physiologic fluid. This prepared mixture was sprinkled on colon wall during colonoscopy. Meanwhile, the existing medical treatments of patients were continued.
Results
Clinical results
The clinical and demographic data of the patients are summarized in Table 1. All cases had clinical and symptomatic active disease until at least 3 months before administration. The Mayo score was used to assess the endoscopic severity of UC. 10 According to the Montreal classification, 11 two cases had E3S2 colitis and eight patients had E2S2 colitis. All the patients had higher blood C-reactive protein (CRP) level (50–80 mg/L) and fecal calprotectin (200–300 μg/mg) before the administration. After 6 weeks of the administration, mucosal healing and resolution of colonic symptoms were determined and blood CRP (<10 mg/L) and fecal calprotectin (<60 μg/mg) levels were measured normally. Two patients (case 6 and 8) achieved steroid-free remission. After the administration, all patients had continued clinical remission for 4 months. These results are summarized in Table 2. Of the 10 cases, 7 were treated with 5-ASA + Azathiopurine for at least 1 year and 3 were treated with vedolizumab for at least 6 months before endoscopic administration. No adverse events were developed after ICB administration. This study does not include a control group. Comparison of Mayo scores before and after the administration was performed using the Wilcoxon signed-rank test (Table 3).
Patients’ characteristics.

M: male; F: female; ASA: 5-acetylsalicylic acid; FMT: fecal microbiota transplantation.
Post-treatment outcomes at sixth week.

Comparison of Mayo scores before and after administration was performed using the Wilcoxon signed-rank test.

After adm.mayo < Before adm.mayo.
After adm.mayo > Before adm.mayo.
After adm.mayo = Before adm.mayo.
Wilcoxon signed-rank test.
Based on positive ranks.
According to the results of the analysis, there was a significant difference between before and after administration. (z = −2.879, P = 0.004).
Discussion
IBDs are chronic diseases characterized by pathogenic mechanisms including changes in intestinal microbiota. 12 UC is characterized by uncontrolled innate and adaptive immune responses toward the gut microbiota. Bifidobacteria levels are reduced in the gastrointestinal tract of IBDs. 13 Bifidobacteria provide protection against impaired gut epithelial cell injury. 13 Bifidobacterial exopolysaccharide (EPS) structure was shown to play a crucial role in this protective effect.14,15 It has been shown that EPS structure of Bifidobacterium longum subsp. longum 35624 could prevent aggravated pro-inflammatory responses. 16 B. animalis subsp. lactis proteins are able to increase human gut IgA levels. 16 Also, B. animalis subsp. lactis promotes regulatory T cell (Tregs) and Th1 responses. 16 In an experimental study, co-culture of B. animalis subsp. lactis BB-12 with intestinal dendritic cells reduces pro-inflammatory cytokine IL-10 levels. 17 Meng et al. 18 showed that BB-12 could provide a significant reduction in the percentage of CD14 + HLA-DR + cells that expressed TLR-2 in peripheral blood samples. Although Vila et al. 19 have cited no definitive claim of whether Bifidobacteria are actually lower in UC, oral administration of BB-12 markedly ameliorated dextran sodium sulfate (DSS)-induced colitis, and this effect was attributed to suppressing tumor necrosis factor (TNF)-α-mediated gut epithelial cell apoptosis. 20 Chae et al. 20 also reported that BB-12 could protect against the development of colitis in a DSS-induced mouse model of UC. Although B. animalis subsp. lactis BB-12 does not belong to the human gut microbiota system, it has been shown to be effective and safe for human intestinal microbiota. 21
Mucosal-associated invariant T (MAIT) cells are innate T cells which express aVα7.2-Jα33 TCRα chain T cell receptor (TCR). MAIT cells play an important role in gut anti-bacterial immunity and recognize bacterial riboflavin metabolites presented by the major histocompatibility complex (MHC) class Ib molecule.22,23 Recent studies have revealed that MAIT cells might be related to IBDs.24–26 Mortier et al. 25 showed that the expression of TCR is increased in intestinal tissue samples collected from IBD patients. Also, decreased levels of TCR were shown in peripheral blood samples of IBD patients. 25 Ruiz et al. 16 reported that B. animalis strain exhibits significant induction of Th1 response that provides anti-inflammatory status in IBD patients. B. animalis strain causes non-stimulator status for MAIT cells, and it can be hypothesized that colonoscopic single-strain B. animalis subsp. lactis administration provides enhancement of mucosal healing in IBD patients.
FMT has been used primarily in the treatment of UC with refractory Clostridium difficile infections. 27 Although some beneficial effects of FMT have been shown in UC patients, 27 there are still controversial findings about FMT effect on Bifidobacterium levels in IBD patients.28,29 In a recent analysis, Papanicolas et al. 29 revealed that during the FMT procedures, Bifidobacterium levels might be reduced in the donor stool. Also, they cited that the depletion of beneficial bacteria in stool and the relative abundance of potentially pathogenic species (Escherichia coli and other gram-negative bacteria) during the FMT procedures were two important issues about safety of FMT. Interestingly, Mizuno et al. 30 have shown that the transplantation of stool rich in bifidobacteria may be important for success of FMT. Accord-ing to their study, donor stool with abundant Bifidobacterium transplantation alleviated symptoms in IBS patients. So, it can be concluded that the modulation of Bifidobacterium levels on gut flora might play a key role in the management of bowel disorders and endoscopic single-strain B. animalis subsp. lactis application might be developed in this context for IBD patients.
Prebiotics are commonly non-digestible oligosaccharides which are classified as fructo-oligosaccharides, galacto-oligosaccharides, and XOSs. XOSs are polymers of the sugar xylose and are obtained by hydrolysis of xylans which contain β-(1 → 4) glycosidic bonds. 9 XOSs act as a prebiotic within the digestive tract. 31 Xyloglucan is a highly water-soluble plant polymer that contains XOSs. 8 The configuration of XOSs gives the xyloglucan a “mucin-like” structure, thus providing optimal adhesive properties. 8 Bifidobacteria contain intracellular xylosidases degrading XOS to D-xylose which is metabolized via the fructose-6-P pathway. 31 XOS prebiotics enhance the growth of B. animalis subsp. lactis.31,32 Ansell et al. 33 reported that XOS substrates provide significant induction of Bifidobacterium levels. Viborg et al. 9 defined a novel β-d-xylosidase BXA43 enzyme family 34 that was highly upregulated in B. animalis subsp. lactis BB-12 cultures grown in the presence of XOSs. They also showed that B. animalis subsp. lactis BB-12 gene BIF_00092 is responsible for encoding β-d-xylosidase. Viborg et al. also cited that the BB-12 strain-specific β-d-xylosidase BXA43 enzyme family provides highly upregulated in BB-12 cultures grown in the presence of XOSs, so it can be proposed that B. animalis subsp. lactis BB-12 levels are significantly increased in the presence of XOSs. However, this study does not rule out the possibility of XOSs acting by increasing the resident microbiota of the study volunteers, since no metagenomic analysis was conducted.
In our study, out of the 10 cases, 7 took 5-ASA + Azathiopurine for at least 1 year and 3 took vedolizumab for at least 6 months before endoscopic administration. No adverse events were developed after ICB administration; we determined that ICB as a single-strain and prebiotic-specific administration might provide mucosal and clinical improvement in unresponsive mild to moderate UC patients. However, it should also be cited that our study group was small and our initial results must further be supported by large randomized studies.
Conclusion
In this retrospective study, we observed that an intracolonic single-dose administration of B. animalis subsp. lactis and xyloglucan combination was effective in the mucosal healing and resolution of colonic symptoms in unresponsive UC patients. It is observed that Bifidobacterial use of XOSs leads to rapid recovery of IBDs. However, these clinical data need to be supported by cohort studies with a larger pool of patients.
Footnotes
Acknowledgements
The authors would like to thank Assistant Professor Dr. Ömer Bilen from Bursa Technical University for his support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and informed consent
Our institution does not require ethical approval for reporting case series. Written informed consent was obtained from the patients for their anonymized information to be published in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
