Abstract
Granulomatosis with polyangiitis (GPA) and eosinophilic GPA (EGPA) are distinct yet overlapping forms of antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, each with unique clinical features. GPA typically involves necrotizing granulomatous inflammation of the upper and lower respiratory tracts and is strongly associated with cytoplasmic ANCA (c-ANCA) and PR3-ANCA positivity, while EGPA is characterized by eosinophilia, asthma, and vasculitis. Rare cases of GPA can present with eosinophilia, complicating the distinction between these entities and raising the possibility of a GPA/EGPA overlap syndrome. We present the case of a 40-year-old female with a history of rheumatoid arthritis and recurrent pneumonia, admitted with worsening dyspnea, peripheral eosinophilia, and high titers of anti-PR3-ANCA. Bronchoalveolar lavage confirmed eosinophilic infiltration, leading to an initial diagnosis of GPA with eosinophilia. The patient responded well to corticosteroids, rituximab, and avacopan, with significant clinical improvement. This case underscores the importance of integrating clinical, serologic, and histopathologic findings when diagnosing ANCA-associated vasculitis, particularly in patients with eosinophilia and asthma-like symptoms. Recognizing GPA with eosinophilia as distinct from true GPA/EGPA overlap is crucial for prognosis and treatment decisions.
Introduction
Granulomatosis with polyangiitis (GPA) and eosinophilic GPA (EGPA) are distinct forms of antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis characterized by systemic small vessel inflammation. 1 GPA typically involves necrotizing granulomatous inflammation affecting the upper and lower respiratory tracts, while EGPA is defined by eosinophilic and granulomatous inflammation, commonly associated with asthma and peripheral eosinophilia. 2 While eosinophilia and asthma are hallmark features of EGPA, rare cases of GPA can also present with eosinophilia, leading to potential misdiagnosis as EGPA or GPA/EGPA overlap syndrome. 3 This highlights the importance of a thorough evaluation and the application of strict diagnostic criteria in patients presenting with cytoplasmic ANCA (c-ANCA)-associated vasculitis, particularly when eosinophilia and asthma are part of the clinical picture.
Here, we present a case of c-ANCA-associated vasculitis with overlapping clinical features of both GPA and EGPA, initially raising concern for an overlap syndrome. The patient exhibited peripheral eosinophilia, nasal obstruction, chronic sinusitis, cavitary pulmonary lesions, and positive anti-PR3 antibodies. This case highlights the complexity of diagnosing and managing c-ANCA-associated vasculitis with overlapping syndrome and underscores the importance of a thorough evaluation and tailored therapeutic strategies to improve patient outcomes.
Case Presentation
A 40-year-old woman with a history of rheumatoid arthritis, chronic sinusitis with nasal septal ulceration, chronic otitis media, mononeuritis multiplex, peripheral neuropathy, asthma-like symptoms, and chronic hypoxic respiratory failure on 2 l nasal cannula oxygen presented with worsening cough and shortness of breath, progressing to acute hypoxic respiratory failure requiring intubation.
Her medical history revealed a prior diagnosis of rheumatoid arthritis managed with methotrexate, prednisone, and etanercept, though she had not followed up with a rheumatologist due to frequent hospitalizations for recurrent respiratory failure. Four months prior, she was hospitalized at an outside facility for acute-on-chronic respiratory failure requiring intubation. Bronchoscopy at that time showed white plaques, inflamed mucosa, and significant eosinophilia, leading to a diagnosis of acute eosinophilic pneumonia (AEP). She was treated with high-dose glucocorticoids and antibiotics. However, the progression of pulmonary infiltrates contradicted the diagnosis of AEP or chronic eosinophilic pneumonia (CEP). Bronchoalveolar lavage (BAL) demonstrated reactive bronchial epithelial cells, and lung biopsy revealed mixed inflammation with no significant eosinophils or malignancy. Cultures for Acid Fast Bacillus (AFB) testing for Tuberculosis and fungi were negative, and treatment with antifungals and antibiotics failed to resolve her symptoms. Subsequent imaging identified bilateral cavitary pulmonary lesions (Figures 1 and 2, black arrows), prompting prolonged antimicrobial treatment.
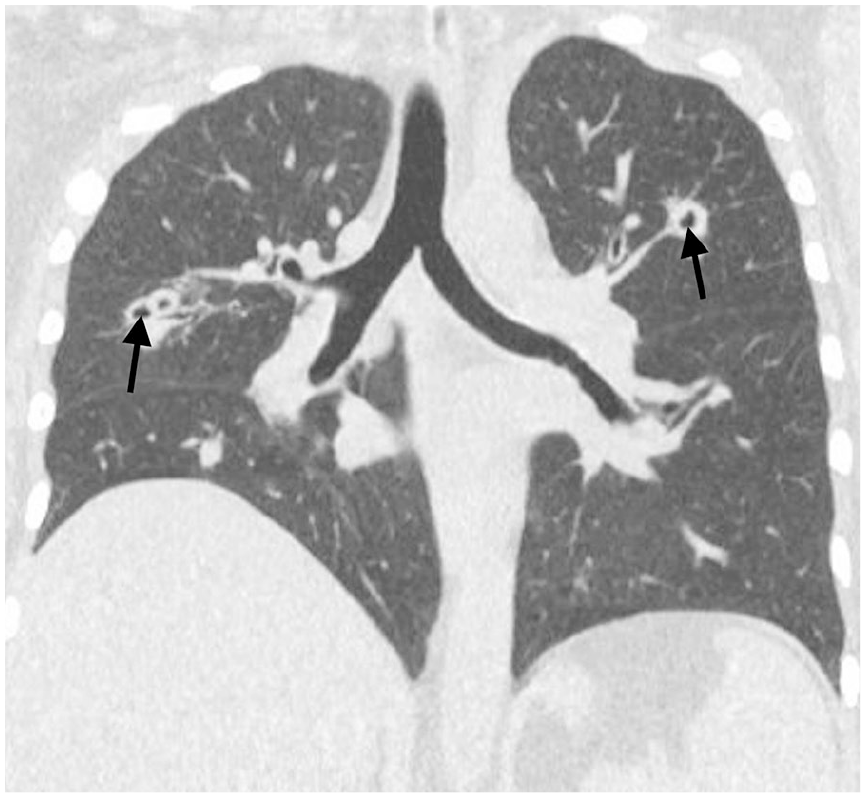
Computed Tomography Scan (CT) chest showing peribronchial cavitary lung lesion (black arrows).

CT chest showing cavitary lung lesion (black arrow).
During the current admission, repeat bronchoscopy revealed reactive bronchial epithelial cells and acute inflammation without malignancy, with a BAL eosinophil count of 1500. Laboratory findings included positive c-ANCA (1:160) and anti-PR3 antibodies (1.5), while anti-myeloperoxidase (MPO), p-ANCA, ANA, anti-dsDNA, anti-scleroderma, and other autoimmune markers were negative (Table 1). Imaging demonstrated extensive paranasal sinus mucosal disease (Figure 3, black arrow) and bilateral cavitary lung nodules. ENT evaluation resulted in myringotomy with tympanostomy tube placement, and a left maxillary sinus biopsy confirmed necrotizing granulomatous inflammation (Figures 4 and 5).
Comprehensive Immunological, Autoimmune, and Infectious Disease Testing Results.

Abbreviations: Ab, antibody; ANA, antinuclear antibody; Anti-dsDNA, anti-double-stranded DNA; Anti-MPO, anti-myeloperoxidase; Anti-PR3, anti-proteinase 3; c-ANCA, cytoplasmic antineutrophil cytoplasmic antibody; CMV, cytomegalovirus; IgA, immunoglobulin A; IgG, immunoglobulin G; IgM, immunoglobulin M; p-ANCA, perinuclear anti-neutrophil cytoplasmic antibody; PCR, polymerase chain reaction; WBC, white blood count; k/cmm, Thousand cells per cubic millimeter.

CT head showing fluid bilateral and mucosal thickening of maxillary sinuses (arrows).
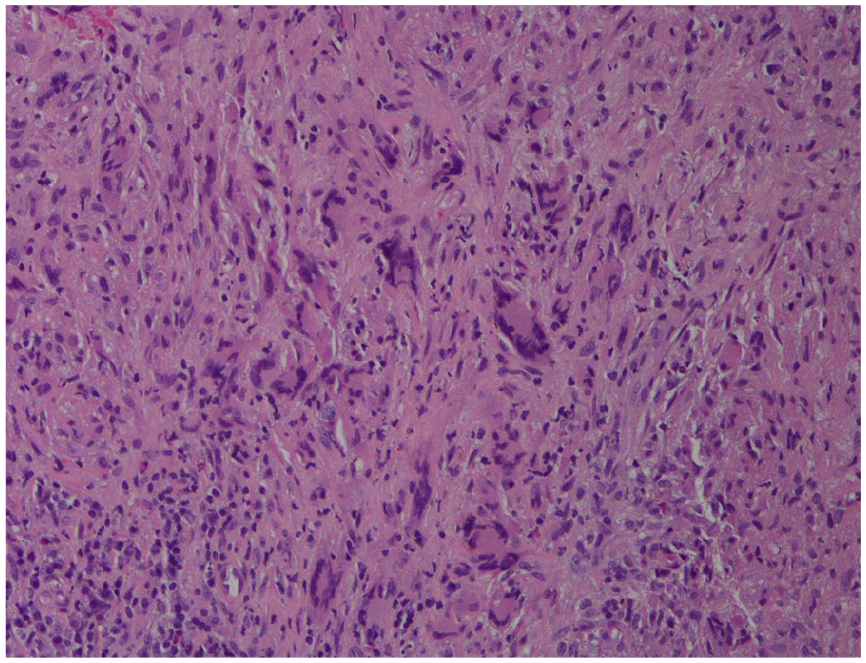
Maxillary sinus biopsy showing giant cells.
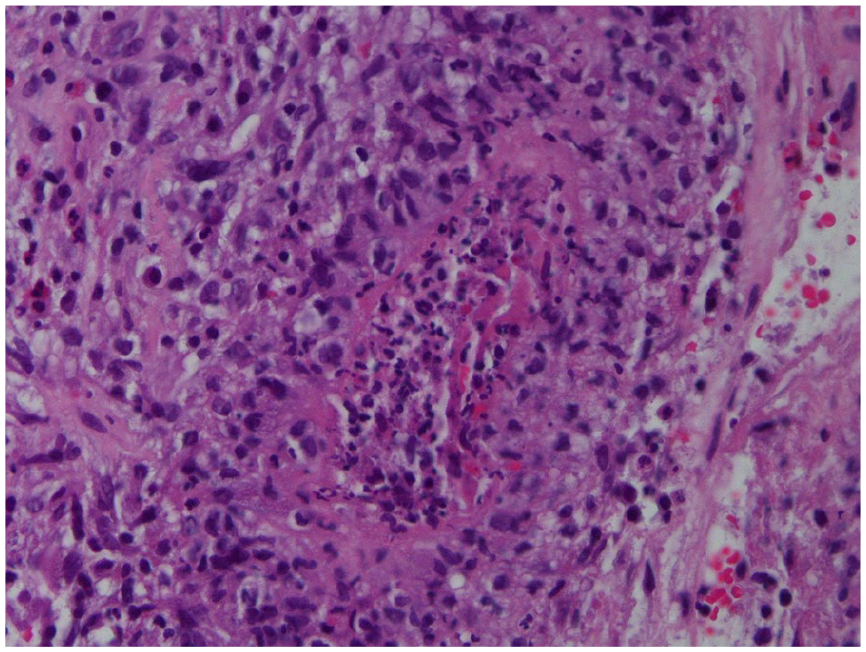
Maxillary sinus biopsy showing necrotizing granuloma.
Given her constellation of findings, including chronic hypoxic respiratory failure, cavitary lung lesions, sinus involvement, peripheral eosinophilia, and positive anti-PR3 antibodies, she was diagnosed with a GPA/EGPA overlap syndrome. She was initially treated with intravenous solumedrol, transitioned to oral prednisone (60 mg daily), and started on rituximab infusions. Her outpatient management included azathioprine and methotrexate, though her regimen was later switched to avacopan alongside prednisone, which effectively controlled her symptoms by addressing the underlying inflammation.
Discussion
Differentiating EGPA from GPA can be particularly challenging in patients exhibiting overlapping clinical features. While eosinophilia and asthma are hallmark features of EGPA, 2 rare cases of GPA can also present with eosinophilia, potentially leading to misclassification as EGPA or as a GPA/EGPA overlap syndrome. 3
A review of the literature highlights several cases with mixed clinical features ultimately diagnosed as GPA/EGPA overlap syndromes. Uematsu et al described a patient with PR3-ANCA positivity, peripheral eosinophilia, asthma, and necrotizing vasculitis, along with classic GPA manifestations such as nasal obstruction and microscopic hematuria. 4 Surendran et al similarly reported a case with polyarthritis, c-ANCA positivity, nasal septal perforation, and eosinophilia. 5 In that patient, asthma and tissue eosinophilia suggested EGPA, while nasal destruction and proptosis favored GPA. Quan et al diagnosed an overlap syndrome based on compelling clinical, radiographic, laboratory, and pathological findings, including cavitary lung lesions, migratory pulmonary infiltrates, alveolar hemorrhage, eosinophilia, and elevated immunoglobulin E (IgE) levels. 6 Shoda et al also reported a patient with PR3-ANCA positivity, granulomatous inflammation, and eosinophilia—despite eosinophilia being atypical for GPA, histopathology and other features supported a GPA diagnosis. 7 These reports underscore the importance of integrating clinical, serologic, and histopathologic findings when diagnosing ANCA-associated vasculitis. Notably, the presence of asthma and eosinophilia does not preclude a GPA diagnosis, and misclassifying these cases as overlap syndromes can overestimate disease complexity.
EGPA typically presents with peripheral eosinophilia, asthma, neuropathy, lung infiltrates, and chronic rhinosinusitis, with tissue biopsies showing eosinophilic infiltration. 1 Diagnosis requires fulfilling at least 4 of 6 criteria, with high sensitivity (85%) and specificity (99.7%). 8 Eosinophilia—defined as either >10% on differential WBC count or >1500 cells/µl—is a defining feature, present in nearly all EGPA cases, often alongside elevated IgE. 2 Approximately 40% of EGPA patients are ANCA-positive, most often with a perinuclear (MPO) pattern.2,9
Conversely, GPA generally presents with pulmonary nodules (often cavitating), necrotizing granulomatous inflammation, and prominent renal disease, typically associated with c-ANCA and anti-PR3 antibodies. Diagnostic criteria include abnormal urinary sediment, abnormal chest imaging, oral ulcers or nasal discharge, and granulomatous inflammation on biopsy, with at least 2 criteria yielding a sensitivity of 88% and specificity of 92%. 8
Turning now to our patient, the constellation of late-onset asthma, chronic sinusitis, arthralgia, peripheral neuropathy, cavitary lung lesions, peripheral eosinophilia (32% on Complete Blood Count [CBC], 1500 cells/µl on BAL), and cutaneous rash initially suggested EGPA. However, the presence of high-titer c-ANCA and anti-PR3 antibodies, cavitary lung lesions, and maxillary sinus biopsy findings strongly favored GPA. Alternative causes of eosinophilia—including neoplastic, parasitic, hematologic, and autoimmune conditions—were thoroughly excluded.
Notably, a bronchoscopy performed 4 months earlier showed white plaques, mucosal inflammation, and marked eosinophilia on BAL, leading to a provisional diagnosis of AEP. Despite high-dose corticosteroids and antibiotics, the patient’s persistent eosinophilia and progressive pulmonary infiltrates made AEP and CEP unlikely. Furthermore, the presence of high-titer PR3-ANCA and peribronchial cavitary lesions—features uncharacteristic for CEP—argued against this diagnosis.
Typically, EGPA manifests on CT with peripheral, asymmetric, migratory ground-glass infiltrates, often without cavitation. 8 Sinus CT in EGPA reveals paranasal sinus thickening without bony destruction. 10 In contrast, our patient’s cavitary lung nodules and extensive paranasal sinus disease with bony involvement were more consistent with GPA.
The absence of eosinophilic infiltration on maxillary sinus and lung biopsy was a pivotal finding that ultimately argued against EGPA. Although eosinophilia is rare in GPA, it has been described in severe disease, likely reflecting an intense inflammatory response rather than an overlap syndrome. Indeed, similar cases have been reported by Potter et al, who described GPA with marked peripheral and tissue eosinophilia, achieving remission with corticosteroids, methotrexate, and cyclophosphamide. 3
Management of EGPA and GPA varies based on disease severity, though key differences reflect their underlying biology. For non-severe EGPA, glucocorticoids plus mepolizumab are recommended, with methotrexate, azathioprine, or mycophenolate as alternatives.11-13 In contrast, non-severe GPA glucocorticoids plus methotrexate or mycophenolate is used for induction therapy.11,14,15 For severe disease, both conditions require glucocorticoids with either cyclophosphamide or rituximab, though rituximab is more widely used in GPA, including for maintenance.11,16 In relapsing or refractory EGPA, adding or switching to mepolizumab is recommended if not previously used.11,12 Maintenance in both diseases may include azathioprine, methotrexate, or mycophenolate, with mepolizumab additionally used in EGPA.17,18 Treatment strategies should be personalized, often requiring a combination or stepwise approach based on the organs affected, prior therapies received, and the patient’s overall health status. Avacopan, a C5a receptor antagonist, may be considered as a glucocorticoid-sparing agent in select cases, particularly in patients at high risk for glucocorticoid toxicity in EGPA. 19
In summary, despite the initial suspicion of EGPA due to the presence of asthma and eosinophilia, the clinical, radiographic, serologic, and histopathologic findings in our case favored a diagnosis of GPA with eosinophilia rather than a true overlap syndrome.
Conclusion
GPA with eosinophilia is rare but recognized in the literature, posing diagnostic challenges that require a high index of suspicion and a multidisciplinary approach. The lack of standardized diagnostic criteria highlights the need for further research. Treatment of EGPA and GPA is guided by disease severity, with mepolizumab recommended for non-severe EGPA and methotrexate or mycophenolate used in non-severe GPA. Severe disease in both conditions typically treated with glucocorticoids combined with cyclophosphamide or rituximab, the latter being more commonly used in GPA. Maintenance regimens overlap, though mepolizumab remains specific to EGPA, and avacopan may serve as a steroid-sparing option in select cases. This case underscores the importance of distinguishing GPA with eosinophilia from EGPA or overlap syndromes, as accurate classification is essential for guiding treatment and improving patient outcomes.
Footnotes
Acknowledgements
I would like to express my heartfelt gratitude to Dr Saroj Sigdel, MD, Associate Professor of Pathology, for generously providing the histology slides.
Authors’ Note
This case report Abstract was presented in poster format at the American Thoracic Society (ATS) International Conference 2025 in San Francisco, California, on May 20, 2025.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
