Abstract
Intermediate-risk pulmonary embolism describes hemodynamically stable patients with evidence of right ventricular strain, whereas high-risk pulmonary embolism comprises hemodynamic decompensation resulting in cardiogenic shock. The clinical manifestations of the two aforementioned conditions are challenging to treat in the setting of heparin-induced thrombocytopenia (HIT). Without immediate therapeutic anticoagulation, patients can subsequently deteriorate and require additional advanced therapies, with the end-goal of restoring pulmonary artery perfusion. We present the case of a male with bilateral intermediate-risk pulmonary emboli who developed type 2 HIT prompting different selection of appropriate management strategies.
Introduction
Acute pulmonary thromboembolism is a life-threatening cardiovascular condition with high mortality. In intermediate-risk pulmonary embolism (PE), management strategy may include catheter-directed thrombolysis in addition to systemic anticoagulation. Immediate systemic thrombolysis should also be considered if the patient’s condition deteriorates and shock ensues. When hemodynamically stable, heparin infusion is the mainstay of therapeutic anticoagulation with potential complications. Heparin-induced thrombocytopenia (HIT) is a rare but potentially fatal, antibody-mediated adverse drug reaction of heparin therapy. It occurs among 1% of hospitalized patients receiving heparin, and the mortality rate is as high as 20%. 1 Here we present the unusual clinical course a patient with acute intermediate-risk bilateral pulmonary thromboemboli who developed HIT and required additional management strategies due to rapid conditional change.
Case Presentation
A 59-year-old man with a history of hypertension and moderate, persistent asthma presented with 5-day history of worsening dyspnea on exertion and substernal chest pain. Initial vital signs were blood pressure of 159/105 mmHg, pulse 104 beats/min and pulse oximetry of 99% on room air. Physical examination was unremarkable. Admission electrocardiogram showed right axis deviation and T-wave inversions in the anterolateral leads but no ST depressions or elevations (Figure 1). It also demonstrated S1Q3T3 pattern that was suggestive PE. Chest x-ray was notable for prominence of pulmonary arteries, typically seen in pulmonary hypertension (Figure 2). Laboratory studies revealed elevated NT pro-BNP 2737 pg/mL, high-sensitivity troponin 86 ng/L, and elevated D-dimer 55.45 mg/L. His initial hemoglobin and platelet count were found to be within normal limits at 15.2 gm/dL and 193,000/μL, respectively. His urinalysis and toxicology screen were negative. Autoimmune panel, including Lupus anticoagulant, was also negative.

Electrocardiogram demonstrates right axis deviation, S1Q3T3, and T-wave inversions in the anterolateral leads.
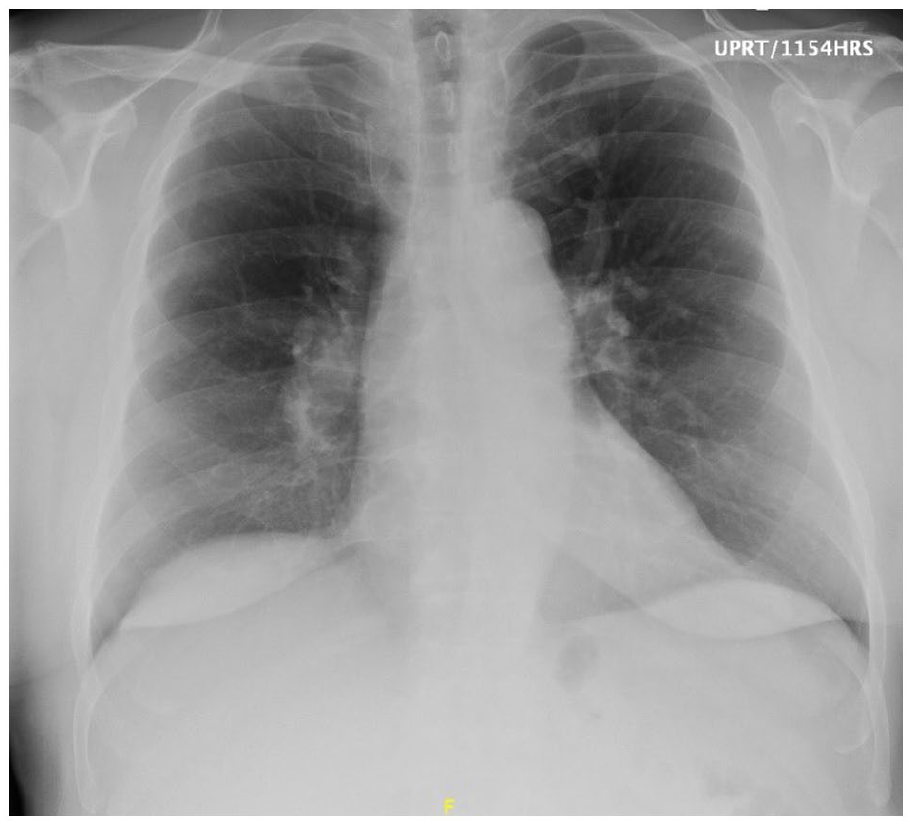
Stable cardiomediastinal silhouette with tortuous and/or ectatic thoracic aorta. Redemonstration of prominence of the pulmonary arteries which can be seen in the setting of pulmonary arterial hypertension.
Chest computed tomography (CT) angiography showed acute large central bilateral PE which extended into the upper and lower lobe pulmonary arteries (Figure 3). Venous Doppler of the lower extremities demonstrated occlusive deep venous thrombosis of the left popliteal vein. An urgent transthoracic echocardiogram (TTE) demonstrated left ventricular ejection fraction 60% to 65% without evidence of right ventricular (RV) strain. The patient was treated with heparin infusion, sublingual nitroglycerin, and as-needed morphine for chest pain relief.

(a) Coronal and (b) axial view of chest computed tomography angiography show bilateral pulmonary embolism extending into main pulmonary arteries. Reflux of contrast into inferior vena cava and hepatic veins secondary to elevated right heart pressure.
On hospital day 2, the patient endorsed worsening dyspnea, and repeat TTE showed unchanged LVEF now with evidence of RV strain. The right atrium and ventricle were noted to be moderately dilated with reduced systolic function and an estimated RV systolic pressure (RVSP) of 28 mmHg (Figure 4). The inferior vena cava was also noted to be moderately dilated. At this time, the patient developed worsening frequent chest pain with increasing oxygen requirement. As a result, his treatment team decided to consult interventional radiology (IR) for catheter-directed thrombolysis due to worsening clinical status. However, due to a significant decrease in platelet count to 62,000/μL, catheter-directed thrombolysis was postponed, and an inferior vena cava catheter was placed. In addition, immediate argatroban infusion was initiated under the direction of hematology for suspected type 2 HIT, which did later become positive with optical density of 1.011. Although the patient denied prior heparin use and was on therapeutic anticoagulation for only 48 hours, type 2 HIT was the likely diagnosis given a drop greater than 50% of basal value platelet count from admission.
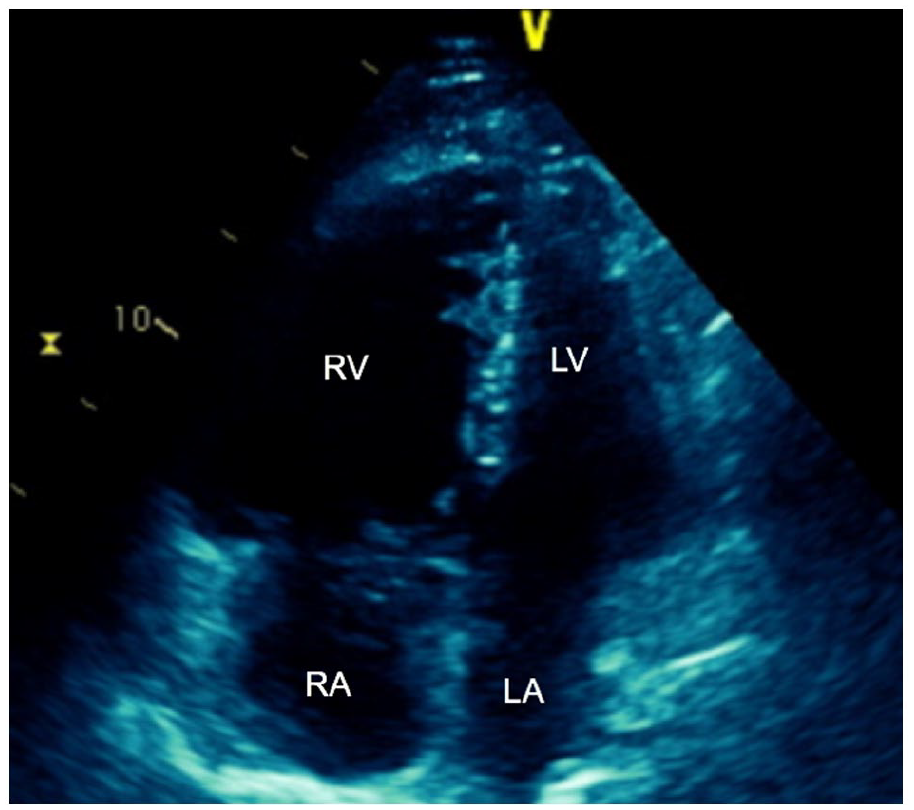
Four chamber view of transthoracic echocardiogram demonstrates dilated RV with RV/LV ratio >1.0.
By hospital day 12, the patient’s platelet count had recovered to 169,000/μL. CT chest showed improvement of clot burden in the left pulmonary artery (PA) but increasing in the right PA with bilateral pulmonary infarct. TTE showed worsening RVSP of 59 mmHg. At this time, the patient’s treatment team consulted IR for catheter-directed thrombectomy and thrombolysis, which were successful. Two days later, the patient was feeling less dyspneic with ambulation while argatroban infusion was continued after thrombolysis. Cardio thoracic surgery was consulted for surgical evaluation due to incomplete lysis of right sided PE and recommended transfer to tertiary center. Repeat chest CT angiography showed reduction of the subsegmental clot burden by 30%. Follow-up TTE to assess RV strain now showed RVSP down to 30 mmHg. Due to his significant and sustained clinical improvement, surgical thrombectomy referral was made on an outpatient basis. The patient was discharged on oral anticoagulation, torsemide, and triamterene.
Discussion
Mortality of high- and intermediate-risk PE remains exceedingly high despite thrombolytic and anticoagulation therapy. At present, clinical effectiveness of fibrinolysis on mortality has not been clearly established beyond 90 days. 2 Despite initial encouraging results, thrombectomy and catheter-directed thrombolysis have not been considered as the first choice of treatment in the current European Guidelines for high-risk PE, even in cases of major contraindication to thrombolysis. 3 Given the variability in PE mortality, risk stratification of low-, intermediate-, and high-risk PE has been adopted by all major guideline committees including the American College of Cardiology. Risk stratification is used to navigate treatment modalities. Patients with low-risk PE (normotensive, normal biomarkers) are typically treated with direct oral anticoagulants in the outpatient setting. High-risk PE (hypotension with systolic blood pressure <90 mmHg for >15 minutes, syncope, cardiac arrest) warrants immediate thrombolytic therapy, with or without mechanical hemodynamic support. 4 Intermediate-risk PEs can present in a normotensive patient with imaging indicative of RV strain, elevated biomarkers, Pulmonary Embolism Severity Index (PESI) class III-IV and its simplified version, sPESI >1 (Table 1). Our patient lacked risk factors for PE, and it was hypothesized part of the lower extremity DVT might have broken off and traveled to the lungs. To maintain hemodynamic stability, systemic thrombolysis, catheter-directed therapy, and surgical embolectomy with mechanical support plus anticoagulation were all considered in the treatments of our patient’s intermediate-risk PE.
Criteria for Low-, Intermediate-, and High-Risk PEs.

Abbreviation: PE, pulmonary embolism; RV, right ventricular; BNP, brain natriuretic peptide; PESI, Pulmonary Embolism Severity Index.
Unfractionated heparin is the preferred anticoagulant for those who are candidates for thrombolysis or further advanced therapies as it allows more flexibility for procedures. 5 Systemic thrombolysis in patients with intermediate-risk PE has been shown to reverse hemodynamic compromise by improving RV dilatation, pulmonary artery pressure, and pulmonary perfusion. 6 Unlike high-risk PE, systemic thrombolytic therapy in intermediate-risk PE has not shown to reduce mortality and recurrence. 7 Given the risks of systemic thrombolysis including major bleeding and intracranial hemorrhage (ICH), catheter-directed approaches are used in patients with relative contraindications to tissue plasminogen activator (tPA). Catheter-directed delivery of the fibrinolytic agents or mechanical fragmentation of a thrombus with a standard pulmonary artery catheter have a lower risk of ICH (0.35%) when compared with systemic thrombolysis (3%). 8 Surgical embolectomy is another management strategy suited for patients with intermediate-risk PE who have failed thrombolytic therapy or with absolute contraindications to thrombolytics (Figure 5). In recent years, the perioperative mortality for patients undergoing surgical embolectomy has decreased to a rate between 3.6% and 29%. 9 In patients with cardiogenic shock or refractory PE, mechanical circulatory support is considered. Venoarterial extracorporeal membrane oxygenation and RV assist devices bypass the pulmonary circulation and decrease RV afterload, improving RV function. 10

Pulmonary embolism treatment algorithm. 11
Systemic thrombolysis such as recombinant tPA has been suggested as the first-line treatment in patients with high-risk PE presenting with very few contraindications. 12 On average, more than 90% of patients will respond within 36 hours. 13 Good clinical outcome can be achieved if the thrombolysis is initiated within 48 hours of onset. 14 In comparison, catheter-directed thrombolysis also showed a reduction in the mean RV distension at 48 hours post thrombolysis. 15 For catheter-directed therapy, the rates of major bleeding ranged from 0% to 4%, and less than 1% of patients experienced ICH. 16 The rate of major bleeding in patients who received systemic thrombolysis was 19%, and 5% was intracranial. 17 In general, bleeding rates vary among studies, and the rates comparing interventional bleeding risk after 48 hours are lacking in literature. Considering our patient presented 5 days after symptom onset, systemic heparinization was initiated as the benefits and risks were similar to systemic thrombolysis after 1 week of onset. 18 In addition, the patient’s PESI score was 69 (Class II low risk) and sPESI was 0, which further supported the use of unfractionated heparin over thrombolysis in the setting of intermediate-risk PE. Our patient remained severely dyspneic despite anticoagulation with non-alarming vital signs. Considering his deterioration while on anticoagulant therapy, catheter-directed thrombolysis was performed after his platelet count recovered. In the ULTIMA trial comparing catheter-directed thrombolysis plus anticoagulation and anticoagulation alone, there was significant improvement of RV distension in the former, thus supporting a hemodynamic benefit in the catheter-directed therapy group. 19
HIT antibodies may develop in up to 8% of heparinized patients, and approximately 1% to 5% have thrombocytopenia. 20 Although thrombocytopenia is the most common presenting feature of HIT, in up to 25% of patients with HIT, the development of thrombosis precedes that of thrombocytopenia. 21 Our patient’s elevated heparin-induced platelet antibody of 1.011 and a decrease of more than 50% in platelet count warranted cessation of heparin and initiation of argatroban. For his acute intermediate-risk PE, a platelet count of 100,000/μL is generally considered adequate for thrombolysis as indicated by Srinivias et al in a study comparing thrombolysis and anticoagulation in management of lower limb venous thromboembolism. 22 Therefore, our patient received thrombolysis when his platelets recovered, which provided him with immediate relief of symptoms.
There were many small studies, which showed success with catheter-directed thrombectomy with or without localized thrombolytics with minimal complication and good efficacy for high-risk PE. 23 One common theme that these studies shared with the management of intermediate-risk PE was that catheter-directed thrombectomy was a treatment option only when systemic thrombolysis was contraindicated. 24 With more advancement in the field, catheter-directed therapy could be considered as the first-line treatment for intermediate-risk PE as it can provide both mechanical and pharmacological treatment and rapid relieve of RV strain. 25
Conclusion
Intermediate-risk PE remains a challenging clinical scenario due to potential clinical deterioration as a result of treatment failure, medication side effects, and relative contraindication to life-saving procedures. Both medical and procedural treatment options are available and should be considered. The current evidence to date does not support the use of systemic thrombolysis in this patient group. Thrombectomy and catheter-directed thrombolysis remain as invaluable tools and can be life-saving when immediate relieve of RV strain is desired.
Footnotes
Authors’ Note
This study was presented in abstract form at CHEST in Nashville, October 2022 (Asllanaj B, Benge E, Bhatia S, Mcwhorter Y. Managing acute intermediate risk pulmonary thromboembolism in a patient who developed heparin-induced thrombocytopenia: Review of current guidelines and literature. Chest. October 1, 2022;162(4): A977).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in whole or in part) by HCA Healthcare and/or an HCA Healthcare affiliated entity. The views expressed in this publication represent those of the authors and do not necessarily represent the official views of HCA Healthcare or any of its affiliated entities.
Ethics Approval
Our institution did not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
