Abstract
Diffuse alveolar hemorrhage (DAH) syndrome is characterized by bleeding into the alveolar spaces of the lungs and occurs when there is an injury to the alveolar microcirculation that leads to hemorrhage from the alveolar capillaries. We report a case of an 82-year-old woman who presented with acute respiratory distress. The patient had a history of rheumatoid arthritis (RA) and was on amiodarone for atrial fibrillation therapy. Initial diagnostic workup, including bronchoscopy and imaging studies, revealed diffuse opacities and the bronchoalveolar lavage fluid consistent with DAH. The patient required aggressive management with supportive care and corticosteroids. Laboratory work showed a synergistic effect between amiodarone and RA in inducing DAH. This makes the report unique as no reports in the literature described a synergic effect of amiodarone and RA in inducing DAH. The primary objective of this report is to guide physicians and remind them to keep DAH at the top of their differential diagnosis in the setting of an RA patient taking amiodarone.
Keywords
Background
Diffuse alveolar hemorrhage (DAH) syndrome is characterized by bleeding into the alveolar spaces of the lungs and occurs when there is an injury to the alveolar microcirculation that leads to hemorrhage from the alveolar capillaries. The cardinal symptom in DAH is hemoptysis. 1 Diffuse alveolar hemorrhage has been associated with multiple systematic inflammatory diseases and medications.2-4
Rheumatoid arthritis (RA) is a chronic multisystem autoimmune inflammatory disease with a global prevalence of 0.46%, the highest being in North America, with a prevalence of 0.70%. 5
Amiodarone is a class III anti-arrhythmic agent commonly used to manage supraventricular and ventricular arrhythmias. However, it has been implicated with many adverse effects and caution must be used in clinical practice. 6
This report describes the case of an 82-year-old patient with RA, who developed DAH shortly after initiating treatment with amiodarone. The extensive literature review revealed few reports that have described amiodarone-induced DAH (Table 1)7,8 and even fewer had described DAH associated with RA (Table 2).9-11 Although the previous reports have described DAH because of RA or amiodarone toxicity, no cases have suggested amiodarone augmenting the exacerbation of DAH in an RA patient, which makes our case worth writing about.
Literature Review Showcasing DAH Associated With Amiodarone.

Abbreviations: DAH, diffuse alveolar hemorrhage; BAL, bronchioalveolar lavage; NA, not applicable.
Literature Review Showcasing DAH Associated With RA.

Abbreviations: DAH, diffuse alveolar hemorrhage; RA, rheumatoid arthritis; NA, not applicable; BAL, bronchioalveolar lavage.
Case Presentation
An 82-year-old woman presented with a past medical history significant for atrial fibrillation, congestive heart failure, RA, iron deficiency anemia, asthma, liver cirrhosis, type 2 diabetes mellitus, gastroesophageal reflux disease, and deep vein thrombosis status post–inferior vena cava (IVC) filter placement. The patient was diagnosed with RA in 2019 and was managed effectively using hydroxychloroquine.
One month prior to the current admission, the patient was admitted due to chest pain and shortness of breath. The patient underwent esophagogastroduodenoscopy (EGD) in which no bleeding was noted and there was only mild gastritis; a colonoscopy the following day showed no active bleeding. In addition, both chest computed tomography (CT) and chest X-ray were negative for signs of consolidation. The patient was diagnosed with atrial fibrillation with a rapid ventricular response and was placed on amiodarone 200 mg twice daily to be doubled.
This patient’s admission was related to fatigue, dizziness, and multiple falls upon standing up; the patient’s blood pressure was 129/54 mm Hg, a pulse of 81 bpm with an irregularly irregular rhythm, respiratory rate of 24 breaths/min, SpO2 of 89%, and temperature of 98.6 °F. She also complained of having dark brown stools with positive fecal occult blood. Anemia workup showed low Hgb with iron deficiency. The patient was noticed to have an increased oxygen requirement and was transferred to the intensive care unit.
Imaging showed bilateral diffuse pulmonary infiltrates on chest X-ray and extensive ground-glass opacity and crazy paving pattern were noted bilaterally on chest CT angiogram with and without intravenous (IV) contrast, as shown in Figures 1 and 2.

Chest computed tomography angiogram showing extensive ground-glass opacity and crazy paving pattern noted bilaterally: (A) Sagittal view, (B) coronal view, and (C) axial view.
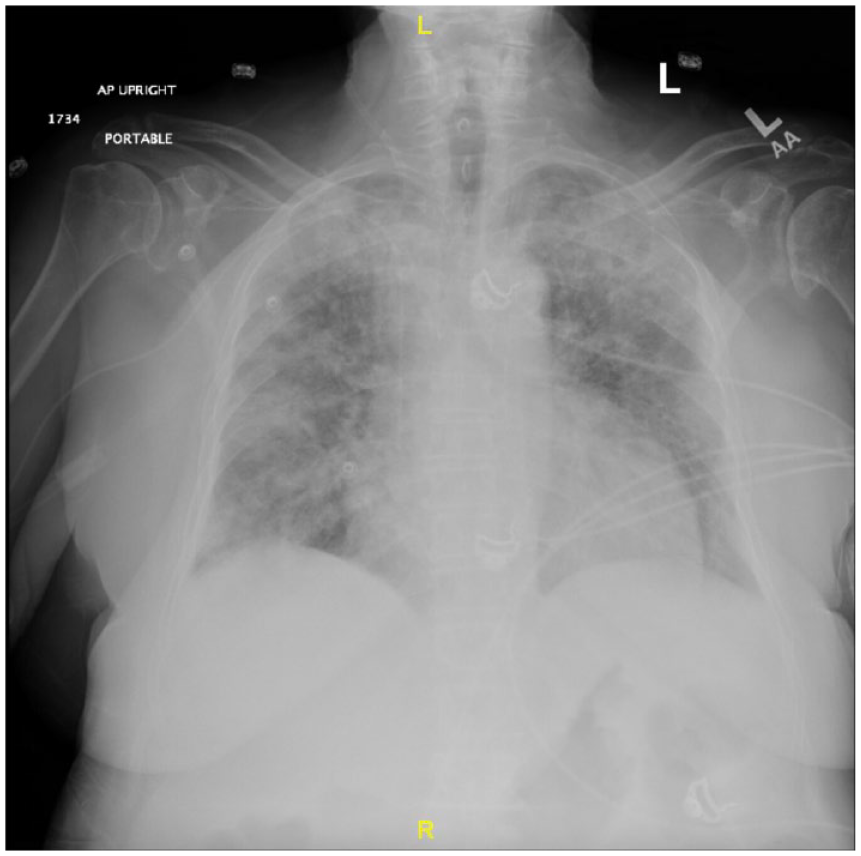
Chest X-Ray showing bilateral diffuse pulmonary infiltrates.
Based on the laboratory findings and imaging, bronchoscopy was performed to rule out the concerns of DAH. Serial bronchioalveolar lavage (BAL) was progressively bloody, consistent with alveolar hemorrhage. Analysis of the fluid showed 3471 red blood cells (RBCs), cultures were found negative, and cytology showed alveolar macrophages and inflammatory cells. Malignancy was ruled out.
The autoimmune panel showed anti-nuclear antibody 1:640 (positive), cyclic citrulline peptide antibody 76 units (positive), and rheumatoid factor 49.1 IU/mL (positive). The antineutrophil cytoplasmic antibody (C-ANCA and P-ANCA), glomerular basement membrane antibody, myeloperoxidase antibody, Serine proteinase 3, and anti-Smith antibodies were all negative. The C3 and C4 complement levels were within normal ranges.
The patient’s coagulation profile was abnormal, with elevated PT at 18.9 and elevated INR at 1.61, which were attributed to secondary to synthetic liver dysfunction in NASH cirrhosis.
Discussion
The patient likely experienced DAH secondary to amiodarone-induced pulmonary toxicity due to the recent initiation of amiodarone. Amiodarone was discontinued and replaced with sotalol to manage atrial fibrillation. In addition, the patient was placed on high-dose methylprednisolone, starting with 1000 mg daily, with a slow taper 5 days later. Our patient’s clinical history was unique, given that she was diagnosed with RA in 2019 with rheumatoid factor (RF) titers of 36.8 IU/mL. After a thorough review of the patient’s previous hospital records, it was noticed that her anti-nucleotide antibodies (ANAs) titers were negative during her first admission. During her most recent admission, the patient’s ANA titers became positive with a value of 1:640, and the RF titers rose to 49.1 IU/mL. This elevation in inflammatory markers suggests that the patient was experiencing an acute RA flare. This concurrent flare in the setting of recent anti-arrhythmic initiation indicates a likely synergistic effect of both amiodarone and RA in developing DAH.
Our recommendation from this case report is that clinicians maintain DAH high on their differential in RA patients on amiodarone presenting with respiratory distress. Further studies should be done on the effect of amiodarone on RA pulmonary manifestations as a likely synergistic relationship exists.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
The abstract statement has not been presented earlier.
