Abstract
A 54-year-old female with history of underlying asthma and 10 pack-year smoking history was seen in interventional pulmonology clinic for evaluation of multiple scattered pulmonary nodules incidentally found on chest computed tomography (CT). Given the central location of the dominant left upper lobe (LUL) nodule and its proximity to an airway, bronchoscopic biopsy was felt to be the right approach. The IonTM Endoluminal System robotic-assisted navigational bronchoscope (Intuitive Surgical, Sunnyvale, California) was used to sample the LUL nodule under fluoroscopic guidance. Together with clinical and radiological findings, the histological and immunophenotypic findings are supportive for Diffuse Idiopathic Pulmonary Neuroendocrine Cell Hyperplasia (DIPNECH). The DIPNECH is a rare condition first described in a case series published in cancer in 1953. This highly atypical condition highlights the utility of modern navigational bronchoscopy in safely securing a diagnostic bronchoscopic biopsy in locations not previously reachable. This is especially relevant given the challenge and risk to percutaneous CT-guided biopsy. Complications are known to scale with depth from skin site, emphasizing benefits of the bronchoscopic approach in obese patients.
Keywords
Case Report
A 54-year-old female with history of underlying asthma and 10 pack-year smoking history was seen in interventional pulmonology clinic for evaluation of multiple scattered pulmonary nodules incidentally found on chest computed tomography (CT). She was born in the Midwest and had lived in Arkansas, Washington, and California.
At the time of presentation to interventional pulmonology clinic, her only reported symptom was exertional dyspnea. She denied cough, wheezing, fevers, chills, night sweats, or weight loss. Physical exam was unremarkable other than for obesity.
Review of her previous chest imaging revealed scattered bilateral subcentimetric solid pulmonary nodules that have been fairly stable over a year and half period except for mild increase in size of left upper lobe (LUL) nodule measuring up to 1 cm in size. Nodules are mostly round/ovoid and were PET negative.
Serologic testing for Mycobacterium tuberculosis, Histoplasma, Coccidioides, and Cryptococcus was negative. Antinuclear antigen and rheumatoid factor were also negative. Spirometry was notable for mild airflow obstruction with no significant bronchodilator response.
Given the central location of the dominant LUL nodule and its proximity to an airway, bronchoscopic biopsy was felt to be the right approach. The Ion Endoluminal System robotic-assisted navigational bronchoscope (Intuitive Surgical, Sunnyvale, California) was used to sample the LUL nodule under fluoroscopic guidance (Figure 1).
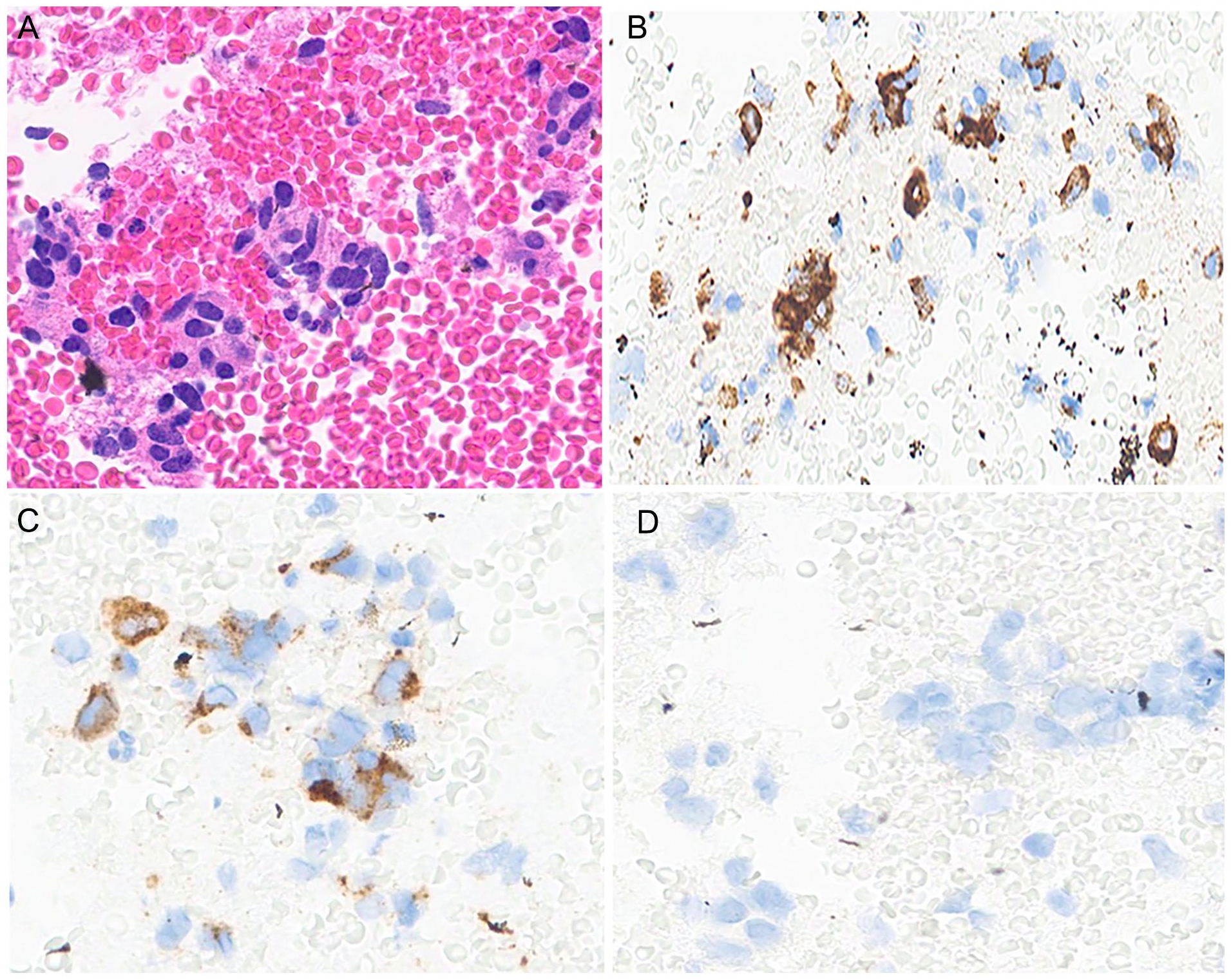
The histological section of the cell block obtained from the robotic fluoroscopy–guided transbronchial fine needle aspiration of the left upper lung lobe shows clusters of crowded cells with ovoid nuclei, increased nuclear to cytoplasmic ratio, and salt and pepper chromatin (A, hematoxylin & eosin, 400×); imparting a neuroendocrine morphology. These neoplastic cells show expression of synaptophysin (B, immunostaining, 400×), and chromogranin (C, immunostaining, 400×). Given patient’s past medical history of pancreatic neuroendocrine neoplasm, the possibility of a metastatic neoplastic process is ruled out with negative expression of PAX8 (D, immunostaining, 400×), and positive PAX8 expression in the pancreatic neuroendocrine neoplastic cells (not shown in this article).
Together with clinical and radiological findings, the histological and immunophenotypic findings are supportive for Diffuse Idiopathic Pulmonary Neuroendocrine Cell Hyperplasia (DIPNECH).
Given the patient’s low symptom burden, the treatment team and patient agreed on a strategy of symptom control with inhaled corticosteroid and long-acting beta agonist. Her symptom control since that time has been such that no further treatment has been needed.
Discussion
Pulmonary neuroendocrine cells (PNECs) are rare epithelial stem cells lining the mammalian respiratory tract in clustered neuroepithelial bodies. The PNECs are prevalent in the fetal and neonatal lung and appear to play a role in lung development. By adulthood, they make up less than 1% of total lung cells. They possess both afferent and efferent neuronal connections which transmit information such as partial pressure of O2, CO2, and mechanical stresses to the central nervous system (CNS). The PNECs have been implicated in the pathophysiology of asthma; in a mouse model, knockout of PNEC-unique genes eliminates allergen-induced asthmatic response. Likewise, overactivity of PNECs is linked to worsening asthma, as well as to bronchopulmonary dysplasia and chronic obstructive pulmonary disease (COPD).1-3
The DIPNECH is a rare condition first described in a case series published in Cancer in 1953. This report described 4 cases with widely variable acuity at presentation, wherein histologic features of hyperplastic neuroendocrine neoplasm were correctly identified in diffusely distributed diminutive noninvasive pulmonary nodules. However, without molecular techniques which have since been developed, neoplastic tissue could not be classified as native pulmonary or metastatic in origin. The authors assigned the nonspecific name of “peripheral and multiple bronchial adenomas” pending further elucidation. 4
The DIPNECH origin from PNECs was confirmed in a case series of 6 patients in the New England Journal of Medicine in 1992. This group manifested with cough, exertional dyspnea, and obstructive or mixed obstructive/restrictive pulmonary function tests (PFTs), leading to characterization of multiple nodules composed of hyperplastic neuroendocrine cells confirmed to be of pulmonary origin. Peribronchiolar fibrosis was also documented which had not previously been identified. This is now thought to be a critical part of pathogenesis in DIPNECH.2,5-7
Pulmonary carcinoid (PC) is an uncommon malignancy, representing 2% of all lung cancers. The DIPNECH is considered a pre-malignant condition, but there are no strong data to determine the rate of progression to PC. Interestingly, DIPNECH has been confirmed to co-occur with or precede development of PC, but PC appears to occur without a DIPNECH stage in most cases.8,9
Presentation
The diagnosis of DIPNECH is most common in middle aged females and not associated with tobacco smoking. Mean age at diagnosis is 58 years with a female-to-male ratio of about 10:1. Owing to its rarity and requirement for invasive workup, exploration in medical literature has been limited to case series and small trials. 3
The DIPNECH patients often carry a diagnosis of asthma, COPD, or gastroesophageal reflux disease (GERD) at the time of presentation. These diagnoses are applied due to complaints of chronic experience exertional dyspnea and/or chronic nonproductive cough. Pulmonary function tests tend to demonstrate airflow obstruction or mixed obstruction/restriction pattern, although normal airflow patterns are not uncommon. The average duration of symptoms prior to diagnosis was 8.6 years in 1 case series of 19 patients diagnosed between 1992 and 2006.10,11
More than 60% of cases will already have multiple round-to-ovoid nodules at the time of first CT. Mosaic attenuation with air trapping is the next most reliable finding secondary to variable inflation of neighboring secondary lobules resulting from diffuse constrictive bronchiolitis. Bronchiectasis has also been documented. These findings are intensified on expiratory high-resolution CT.6,12
Of note, detection of DIPNECH nodules on plain chest radiography is virtually impossible. 13
Workup
Serum chromogranin and urine 5-HIAA levels have been assessed as diagnostic tools. Unfortunately, these are not reliably elevated in DIPNECH, and occasional elevated levels do not correlate with symptom burden nor progression. There is no known serologic test for DIPNECH.8,11,12
Biopsy and tissue evaluation remain necessary for the diagnosis of DIPNECH. 8 It is critical that metastatic, infectious, and rheumatologic disease be excluded as part of the differential diagnosis given the extremely low baseline prevalence of DIPNECH. 9
Pathology demonstrates neuroendocrine cell hyperplasia along the basement membrane and stain positive for neuroendocrine markers, most typically chromogranin A, synaptophysin, and CD56. Early studies relied on surgical biopsies; however, modern standard of care is divided between endobronchial ultrasound-guided (EBUS) and CT-guided percutaneous needle biopsy (CT-PNB). Bronchoscopic approach is preferred for lesions near large airways and percutaneous approach for more peripheral lesions. Percutaneous approach poses a greater risk of complications, namely pneumothorax and pulmonary hemorrhage. In 1 head-to-head comparison of CT-PNB to EBUS for solitary pulmonary nodule, incidence of pneumothorax was 17.5% (14/80) compared to 1.25% (1/80) respectively. This risk increased with depth of nodule from the pleura. 14
A typical bronchoscope with EBUS and biopsy capability has a diameter of 6.5 mm and only modest flex capability, limiting the depth of endobronchial navigation. Furthermore, manually operated bronchoscopy has significant precision limits. New-generation robotic-assisted bronchoscopy tools have been developed to address this problem. The operator uses route-planning software superimposed over navigational CT images to create a path to a target lesion, and then guides a thin, finely controllable device with computer assistance rather than under direct visualization.8,14-16
Management
Owing to the rarity of this condition, clinical trials and prognostication data are limited. Patients frequently present with prior diagnosis of COPD or asthma and thus are often already using inhaled bronchodilators and corticosteroids. There do not exist rigorous trials demonstrating efficacy of these agents, but they appear to be effective in symptom control.7,9
As in other types of neuroendocrine neoplasm, synthetic somatostatin analogues (SSAs) have been employed with success. In an open-label multicenter trial including 42 patients with either biopsy-confirmed DIPNECH or lung neuroendocrine neoplasm with clinical and radiographic findings consistent with DIPNECH, 33 (76%) of patients treated with SSAs had some degree of symptom improvement. Furthermore, out of 15 patients with pre-treatment PFTs, 14 (93%) experienced improvement in forced expiratory volume in 1 second (FEV1).3,11,17-19
The mammalian target of rapamycin (mTOR) inhibitor sirolimus was also assessed in a small case series of 3 patients with moderate or severe obstruction on PFTs and significant dyspnea. They experienced durable improvement of FEV1 and symptom burden, although patients did suffer side effects of painful mouth ulcers, anorexia, and headache which resolved with dose reduction. 18
Current management recommendations of the National Comprehensive Cancer Network are to attempt control of bothersome cough and dyspnea with inhaled agents, reserving SSAs as second line. Surveillance chest CT is recommended every 12 to 24 months or as clinically indicated. If symptoms continue to be unmanageable, multidisciplinary discussion is suggested to consider cytoreductive surgery, systemic chemotherapy, or management of metastatic disease to the liver if present.8,9
Prognosis
Most patients have stable symptoms indefinitely or slow progressive functional decline. In rare cases, constrictive bronchiolitis can progress to critical airflow obstruction. Lung transplant can be offered, although this has been done only a handful of times in reported literature. Zhou et al 20 reported 2 cases of bilateral lung transplant between the years of 2010 and 2013 who did not suffer recurrence or complication as of 2014.
Conclusion
This highly atypical condition highlights the utility of modern navigational bronchoscopy in safely securing a diagnostic bronchoscopic biopsy in locations not previously reachable. This is especially relevant given the challenge and risk to percutaneous CT-guided biopsy. Complications are known to scale with depth from skin site, emphasizing benefits of the bronchoscopic approach in obese patients.
A reasonable approach to the management of DIPNECH is to attempt symptom control first with inhaled medications using a similar approach to asthma. If respiratory symptoms persist, SSAs are an effective second-line treatment. In uncommon cases which fail this level of management, it is reasonable to seek expert multidisciplinary opinion regarding chemotherapy or lung transplant.
Footnotes
Acknowledgements
None.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Self-funded by authors through University of California Davis.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
This has not be presented anywhere else.
