Abstract
Cocaine is an indirect-acting sympathomimetic drug that inhibits norepinephrine and dopamine reuptake in the adrenergic presynaptic cleft. Cocaine use has been associated with strokes, angina, arrhythmias, and agitation. Data on gastrointestinal complications such as mesenteric ischemia, bowel necrosis, ulceration, and perforation are scarce. Here, we present a rare case of cocaine-induced esophageal, gastric, and small bowel necrosis that contributes to the limited literature on this subject. Diagnosis of cocaine-induced gastrointestinal complications involves a combination of imaging studies, laboratory assessments, and histopathological examinations. Timely surgical resection, supported by intravenous fluids, antibiotics, and pain management, is the mainstay of treatment. The prognosis varies but is significantly influenced by the promptness and effectiveness of the intervention, underscoring the importance of vigilant clinical care in such cases.
Keywords
Introduction
Cocaine is an indirect-acting sympathomimetic drug that works by inhibiting catecholamine reuptake in adrenergic presynaptic neurons.1,2 Although approved by the Food and Drug Administration (FDA) as a topical anesthetic for mucous membranes, cocaine has a US box warning for its high potential of abuse and dependence due to its stimulatory effects. 3 Cocaine has been implicated in various systemic adverse effects, such as acute coronary syndrome, stroke, renal infarcts, and death.1,2,4-12 Many of these adverse effects are thought to be a result of direct toxicity, vasoconstrictive, and prothrombotic effects of cocaine. 5 There is a paucity of data on the gastrointestinal complications of cocaine use due to its rarity and vague presentation. Consequently, there is often a delay in diagnosis and intervention. Herein, we describe a unique case of cocaine-induced esophageal, gastric, and small bowel necrosis in an adult female with a history of bipolar disorder and chronic intranasal cocaine use. The patient admitted to using crack-cocaine a couple of hours before the emergency room (ER) visit. We urge clinicians to consider cocaine-induced bowel ischemia and necrosis when evaluating cocaine users for abdominal pain.
Case Report
A 59-year-old woman with a medical history of bipolar disorder, polysubstance use disorder, and cannabis hyperemesis syndrome presented to the ER after a syncopal episode at home. This incident was preceded by several episodes of melenic stools and coffee-ground emesis. In the ER, the patient complained of diffuse abdominal pain and a tingling sensation in the left lower extremity. She denied experiencing chest pain, palpitations, or shortness of breath. The history was limited because of the patient’s clinical status. The initial vital signs were significant for tachycardia (116 bpm) and elevated blood pressure (162/82 mm Hg). The patient appeared in acute distress due to pain, but she saturated well on ambient air. On physical examination, the patient had a soft, obese abdomen, which was tender to palpation in the epigastrium and the left lower quadrant. There was no evidence of guarding, rigidity, or rebound tenderness. She had normoactive bowel sounds throughout, and the digital rectal examination was negative for fresh blood. The patient’s mentation was intact, and she had a normal respiratory effort. The remainder of the examination was unremarkable.
Laboratory values were significant for electrolyte and metabolic derangements, including hyponatremia, hypochloremia, and low bicarbonate with an anion gap of 33 (Table 1). Bloodwork was also notable for acute kidney injury, lactic acidosis, transaminitis, leukocytosis, and thrombocytosis (Table 1). The patient was administered 2 liters of lactated Ringer’s (LR) solution, and the intensivist service was consulted for an evaluation. Following the administration of fluids, the patient’s lactic acid trended down from 7.3 to 1.3 mmol/L, kidney function improved, and the anion gap improved to 14. The patient’s repeat hemoglobin dropped from 11.7 to 8.5 g/dL and she was transfused with 1 unit of packed red blood cells (PRBCs). Bedside point-of-care ultrasound showed a hyperdynamic left ventricle without any effusion or volume overload and a barely visible inferior vena cava. An additional 3 liters of LR solution was given as boluses. The patient was kept nil per os (NPO) and started on pantoprazole 40 mg, intravenously (IV), twice daily (BID), and antiemetics as needed. The patient was deemed stable for admission to regular medical floors with critical care staying on for co-management.
Pertinent Admission Laboratory Values Compared to the Reference Ranges.

Overnight, the patient had additional episodes of hematemesis despite symptomatic treatment with fluids, antiemetics, pantoprazole, and thiamine. On re-evaluation, she was tachycardic (109 bpm) and hypoxic. Repeat bloodwork showed a lactic acid of 6.3 mmol/L, and the patient was intubated at the bedside for airway protection. Computed tomography (CT) angiography of the chest, abdomen, and pelvis revealed a small-bowel obstruction and associated pneumatosis and portal venous gas, suggestive of ischemia (Figure 1). The CT scan also demonstrated diffuse colonic wall thickening, suggestive of non-specific colitis. The patient was started on norepinephrine due to borderline hypotension, and the surgical team was consulted for small bowel ischemia.
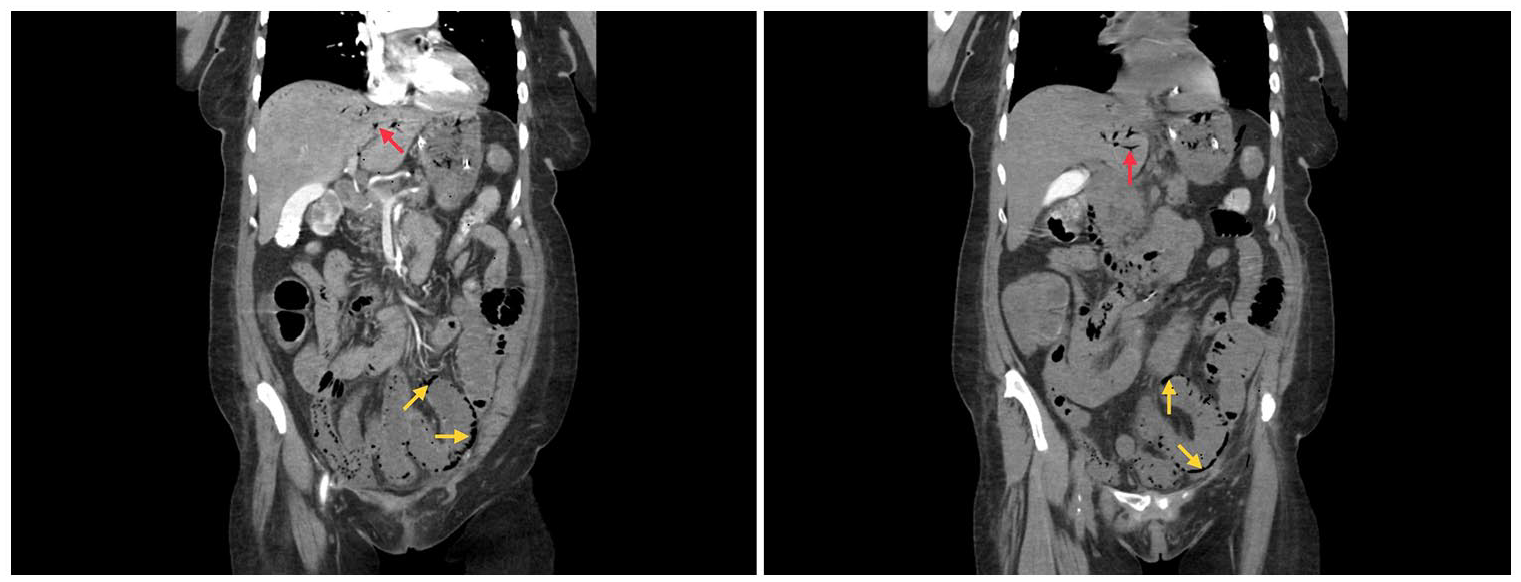
CT angiography of the chest, abdomen, and pelvis showing small-bowel obstruction and associated pneumatosis (Yellow arrow) and portal venous gas (red arrow), suggestive of ischemia. CT angiography is also notable for diffuse colonic wall thickening indicative of non-specific colitis.
Given the emergent nature of the situation, a 2-physician consent was obtained. The patient underwent an emergent open exploratory laparotomy, which showed portal venous gas and necrotic small bowel. An intraoperative esophagogastroduodenoscopy (EGD) revealed a necrotic distal esophagus and gastric body. No ulcers were seen on bowel exploration. The surgeons performed a small bowel resection with primary anastomosis, and the patient tolerated the procedure well. Due to significant intraoperative acidosis, the patient remained intubated and was admitted to the surgical intensive care unit (SICU) for postoperative care.
The resected portion of the small bowel measured 48.0 cm in length, and gross examination showed a serosa with dusky brown and multiple skipped necrotic areas. A tan-brown, focally necrotic, hemorrhagic, and partially congested mucosa was noted on further examination. The skipped necrotic areas showed a paper-thin wall but without any gross evidence of perforation. The histopathology revealed a small intestine with acute inflammation, mucosal sloughing, necrosis, and a thin wall, consistent with intestinal ischemia (Figure 2). One of the resections showed acute inflammation with mucosal sloughing. The patient’s urine drug screen was positive for cocaine and tetrahydrocannabinol (THC), and she later admitted to chronic intranasal cocaine use. The patient admitted to using crack-cocaine a couple of hours before the ER visit. She denied a personal or family history of clotting disorders and the hypercoagulable state workup was unremarkable. The patient’s presentation was consistent with cocaine-induced esophageal, gastric, and small bowel necrosis.

Histomicrographs stained with H&E showing cocaine-induced microscopic changes on the resected segment of the small intestine. The histopathologic features include acute inflammation, mucosal sloughing, necrosis, and a thin wall consistent with intestinal ischemia.
The patient’s SICU stay was uneventful, and she was continued on intravenous pantoprazole 40 mg, BID and sucralfate 1 g, daily. She was extubated on postoperative day 3 and she was successfully transitioned to oxygen therapy with a nasal cannula. Her hospital course was complicated by multiple episodes of watery diarrhea and electrolyte derangements, both of which resolved with electrolyte repletions and fluid therapy. Infectious stool workup including Clostridium difficile was negative. The patient was counseled on substance use and medication compliance and discharged to a shelter. At the time of the discharge, her mentation was intact, and she tolerated a regular diet with mechanical soft consistency. She reported mild abdominal pain, which improved with acetaminophen. The patient will undergo a follow-up EGD in 8 to 12 weeks after discharge to assess for any postprocedural complications.
Discussion
Cocaine is a drug in the amphetamine class and has numerous documented complications, mostly cardiovascular and cerebrovascular in nature.9,10,12 Common side effects of cocaine use include hemorrhagic strokes, myocardial infarctions, chest pain, agitation, and cardiac arrhythmias.9,10,12,13 In the United States, there are estimates that up to 14.5 million people, or 6% of the adult population, have used cocaine. 10 With the increased availability of cocaine, coupled with the misconception that cocaine is a mostly harmless drug, the prevalence of cocaine users and unique complications will likely continue to increase. We present a peculiar case of cocaine-induced esophageal, gastric, and small bowel necrosis in an adult woman with a history of chronic intranasal cocaine use. To the best of our knowledge, this is the first reported case of cocaine-induced esophageal, gastric, and small bowel necrosis.
Mesenteric vessels have a high burden of alpha-adrenergic receptors and cocaine use inhibits norepinephrine and dopamine reuptake, leading to significant vasospasm and vasoconstriction of the microvasculature.6,7,14,15 Cocaine also increases intracellular calcium, causing further vasoconstriction, which may evolve into bowel ischemia.7,15 In addition to non-occlusive mesenteric ischemia, cocaine can also cause platelet aggregation, localized endothelial injury, and decreased fibrinolytic activity.4,7 Prolonged bowel ischemia can progress to necrosis, ulceration, hemorrhage, infarction, perforation, or retroperitoneal fibrosis.8,16 Because of its rarity and ambiguous presentation, cocaine-induced bowel ischemia is often misdiagnosed, leading to grave patient outcomes.
Cocaine-induced gastrointestinal complications include small bowel hematomas, intussusception, bowel ischemia, thrombosis, necrosis, hemorrhage, acute pancreatitis, and bowel perforation.5,12,17,18 Despite limited data, temporality and a dose-response relationship between cocaine use and mesenteric ischemia have been established. Bowel ischemia typically presents with sudden-onset sharp abdominal pain that is disproportionate to the physical findings.9,12 The presence of peritoneal signs on examination may be indicative of bowel perforation. Hemodynamic changes may accompany the symptoms, although hypotension may be masked by the hypertensive effects of cocaine.
Cocaine-induced mesenteric ischemia can also manifest as anorexia, bloody diarrhea, melena, hematochezia, bowel obstruction, fever, or shock.1,7,8,10 Patients with esophageal involvement often present with nausea, vomiting, hematemesis, dysphagia, or odynophagia. Our patient did not tolerate oral intake at presentation, and she was later found to have distal esophageal necrosis. Strzepka et al 12 presented a unique case of acute pancreatitis in a male patient with a history of crack-cocaine use. The patient was treated with fluids, antiemetics, and analgesics and was discharged on day 4 after clinical improvement. In young adults, cocaine-induced acute abdomen can mimic other conditions, including acute pancreatitis, acute appendicitis, inflammatory bowel disease, ovarian torsion, peptic ulcer disease, and abdominal migraine.10,19 The prognosis for this condition is extremely poor; mortality is high as identification of bowel ischemia often occurs late in the disease course. 6 Colonic ischemia irrespective of the route of cocaine administration has been associated with worse outcomes.8,15
Imaging plays a crucial role in the diagnosis of cocaine-induced gastrointestinal complications. Contrast-enhanced CT scans can detect pneumatosis intestinalis, or signs suggestive of bowel ischemia, such as mucosal enhancement and continuous bowel wall thickening.7,9 Gupta et al 9 described an interesting case of cocaine-induced gastric ischemia that was diagnosed on imaging. The patient was noted to have hemorrhagic gastritis but without perforation. Similarly, Leth et al 11 presented a case of cocaine-induced ischemic colitis, which was detected on a CT scan as thickened and dilated bowel wall. The patient further underwent a colonoscopy to confirm the diagnosis and rule out Crohn’s disease. Endoscopic modalities aid clinicians in visualizing the extent of damage caused by cocaine-induced mesenteric ischemia 13 and ruling out other potential causes of an acute abdomen. Endoscopies performed in patients with mesenteric ischemia patients often show edema, diffuse erythema, and subepithelial hemorrhage.13,20 Our patient was noted to have distal esophageal and gastric necrosis during an intraoperative EGD.
Laboratory findings, including white blood cell (WBC) count and lactate levels, are important diagnostic markers of acute mesenteric ischemia. Leukocytosis, often observed in patients with acute abdominal symptoms, suggests an inflammatory response. 7 In addition, lactate levels can indicate tissue hypoxia and are commonly elevated in cases of mesenteric ischemia, although a normal lactate level does not entirely rule out bowel ischemia.7,12,21,22 Elevated serum lactate levels are typically indicative of late-stage ischemia and transmural bowel necrosis. 7 On presentation, our patient had an elevated lactate level which was consistent with her shock state and suggestive of mesenteric ischemia. A pathological examination of the resected bowel specimens provides definitive evidence of ischemic damage. On gross examination, ischemic bowel segments tend to have mucosal necrosis, congested mucosa, ulceration, perforation, or transmural necrosis with inflammatory infiltration. 19 Histopathology usually shows submucosal edema, vascular congestion, mucosal sloughing and ulceration, or full-thickness necrosis.7,19
The management of cocaine-induced mesenteric ischemia involves a comprehensive approach that integrates surgical intervention and supportive care. Surgical removal of necrotic tissue is imperative to prevent further ischemic damages and to improve the patient’s overall prognosis.2,7-10,12,15,21,22 Intravenous fluids (IVFs) are administered to maintain hydration levels, lower lactate levels, and replace lost blood. 23 Antibiotics are prescribed to control and prevent infections, a critical aspect of postoperative care. 19 Pain management techniques, encompassing various pharmacological and non-pharmacological interventions, are crucial for providing relief to patients, promoting comfort, and aiding in the recovery process. The prognosis of patients with cocaine-induced gastrointestinal complications depends on the extent of the damage, timeliness of intervention, and overall patient status. Early diagnosis and intervention are associated with improved patient outcomes.7,19
Conclusion
The use of cocaine in the United States has been increasing owing to its low cost and ease of access. Traditionally associated with hemorrhagic strokes, angina, and agitation, cocaine can also cause gastrointestinal complications such as bowel ischemia, mesenteric infarctions, and ulceration. Due to its rarity and vague presentation, cocaine-induced bowel ischemia is often misdiagnosed leading to grave patient outcomes. We present a rare case of cocaine-induced esophageal, gastric, and small bowel necrosis.
Footnotes
Author Contributions
L.B. and S.S.O. conceptualized the idea for this case report. R.B., T.W., and P.P. wrote some sections of the case report. R.Y., G.M., and J.E. fact-checked, edited, and proofread the final version of the case report. S.Q. prepared the pathology slides and helped with the interpretations.
Data Availability Statement
Further enquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require IRB approval/waiver for case reports.
Informed Consent
We obtained consent from the patient for the publication of this case report.
