Abstract
Acute esophageal necrosis (AEN), also known as Gurvits syndrome, is a rare and potentially life-threatening condition characterized by necrosis of the esophageal mucosa. Acute esophageal necrosis is often associated with critical conditions, such as myocardial infarction, diabetic ketoacidosis (DKA), coronavirus disease 2019 (COVID-19) infection, or post-surgical complications. Patients typically present with nausea, hematemesis, acute dysphagia, and melena. Given its high mortality rate, prompt detection with upper endoscopy and early initiation of treatment are crucial. Most cases of Gurvits syndrome are managed conservatively using intravenous fluids, proton pump inhibitors, and antibiotics. Herein, we present a case series of AEN in the setting of DKA. Both patients received supportive care and were discharged in a stable condition.
Keywords
Introduction
Acute esophageal necrosis (AEN), also known as “black esophagus,” Gurvits syndrome, or acute necrotizing esophagitis, is a rare condition characterized by necrosis of the esophageal mucosa, leading to a blackened appearance of the esophagus.1,2 Typically, AEN is associated with poor prognosis, with mortality rates ranging from 32% to 36% due to comorbidities. This condition is usually diagnosed through upper endoscopy, revealing a darkened esophagus, usually in the distal third.3-5 Although the exact cause of AEN remains unclear, it is believed to be multifactorial and primarily managed medically. The causes and risk factors include diabetic ketoacidosis (DKA), malnutrition, hypertension, malignancy, and antipsychotic medications.6-8
Patients with AEN may present with symptoms such as upper gastrointestinal bleeding (GIB), abdominal pain, dysphagia, nausea, vomiting, fever, and syncope.2,6,9,10 Early recognition and appropriate management are crucial for improving the outcomes of AEN patients. Supportive care is the mainstay of treatment for most AEN cases, with surgery reserved for acute complications such as esophageal perforation and mediastinitis. 6 Herein, we describe a case series of AEN in the setting of DKA. Both patients were successfully treated with proton pump inhibitors (PPIs), carafate suspension, intravenous (IV) fluids, and insulin infusion. Acute esophageal necrosis should be considered in the differential diagnosis of upper GIB in patients with DKA.
Case Presentations
Case 1
A 52-year-old male with a medical history of insulin-dependent diabetes mellitus (IDDM) and hypertension was brought to the emergency department (ED) for evaluation of bizarre behavior and vomiting. The patient stated that he was released from jail earlier in the day and that he missed his insulin doses. He denied having fever, chills, dysphagia, odynophagia, acid reflux, chest pain, shortness of breath, palpitations, abdominal pain, melena, hematochezia, recent weight loss, polyuria, polyphagia, polydipsia, or dysuria. In the ED, the patient appeared confused but followed verbal commands. The initial vital signs were notable for elevated blood pressure (140/72 mm Hg), tachycardia (125 beats per minute), and tachypnea (28 breaths per minute). On further examination, the patient was alert and oriented to self and place only. There was no facial asymmetry or tongue laceration, and the pupils were dilated but reactive to light and accommodation. The remainder of the examination was unremarkable.
Triage blood tests were significant for hyperkalemia, hyperglycemia, elevated creatinine and blood urea nitrogen, elevated alkaline phosphatase, elevated serum lipase, and leukocytosis (Table 1). Arterial blood gas analysis revealed a severe metabolic acidosis and elevated lactic acid. Additional tests revealed moderate ketones with an anion gap of 27.5. An electrocardiogram (EKG) revealed atrial fibrillation with a rapid ventricular rate but no acute ischemic pattern. The patient was treated for hyperkalemia and admitted to the medical intensive care unit (ICU) for the management of DKA. The heart rhythm converted to sinus rhythm after 10 mg of dose of IV diltiazem and 150 mg of amiodarone. The patient was managed with a DKA protocol comprising insulin infusions, IV fluids, and electrolyte fluid solutions.
Pertinent Admission Laboratory Values for Case 1 Compared With the Reference Ranges.
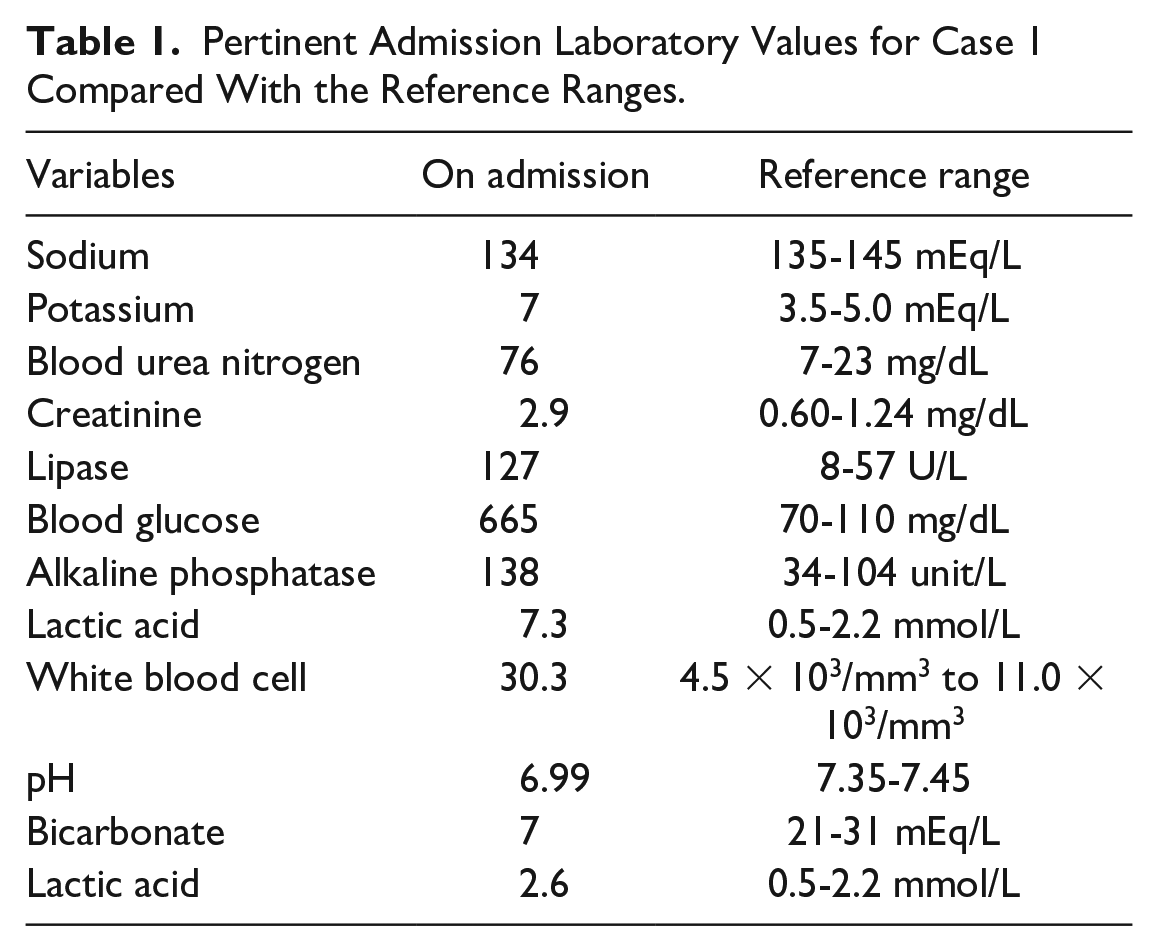
The patient’s ICU course was complicated by melena and acute blood loss anemia, necessitating a gastroenterology consultation. Bedside esophagogastroduodenoscopy (EGD) revealed diffuse esophageal plaques concerning for candidiasis, ulcerated esophageal necrosis, and a small hiatal hernia (Figure 1). The histopathology of random esophageal biopsies revealed necroinflammatory material and ulcer debris associated with abundant fungal microorganisms, which were morphologically compatible with Candida species. Overall, the findings were consistent with AEN in the setting of DKA and Candida esophagitis. Immunohistochemical staining for cytomegalovirus (CMV) and herpes simplex virus (HSV-1 and HSV-2) was negative. The patient received supportive care with buprenorphine, proton pump inhibitors (PPIs), carafate suspension, fluconazole, IV fluids, and insulin infusion. A relook endoscopy after 6 months showed normal esophageal mucosa (Figure 2).

Endoscopic images for case 1 (during hospitalization) showing diffuse esophageal plaques, concerning for candidiasis, and ulcerated esophageal necrosis. The stomach and duodenum appeared normal.

Endoscopic images for case 1 (6 months after discharge) showing normal esophageal mucosa and a small food bolus in the distal esophagus.
Case 2
A 60-year-old male with a history of type 2 diabetes mellitus, hypertension, chronic constipation, and Parkinson’s disease (on carbidopa/levodopa and pramipexole) presented to the ED complaining of acute-onset abdominal pain and distension. The abdominal pain was vaguely localized, sharp in nature, and rated 9/10 at its peak. It was associated with nausea, constipation, and shortness of breath. The patient denied recent illness, melena, hematochezia, chest pain, palpitations, polyuria, polyphagia, or recent hypoglycemic events. He denied alcohol, tobacco, or illicit substance use. The patient also reported compliance with diabetes mellitus medications (linagliptin/metformin and glipizide) and a hypocaloric diet.
In the ED, the patient was lethargic and in acute distress due to pain. The vital signs were significant for transient hypotension (99/57 mm Hg) and tachycardia (103 beats per minute). He was afebrile and saturated 97% in room air. Upon examination, the patient spoke in short sentences with Kussmaul breathing, but the lungs were clear on auscultation. The abdomen was distended and diffusely tender on palpation. The patient had diminished bowel sounds in the lower abdominal quadrants, but there were no signs of trauma, peritonism, flank ecchymosis, or palpable organomegaly. The patient was alert and oriented to self, time, and place. He had a resting tremor in the right upper extremity and cogwheel rigidity. The rest of the examination was unremarkable.
Owing to concerns for DKA, a venous blood gas was ordered, which showed low pH, elevated lactic acid, low bicarbonate level, and an anion gap of 24. Triage blood tests revealed leukocytosis with bandemia, thrombocytopenia, hyperglycemia, low serum bicarbonate level, moderate ketones, elevated creatinine and blood urea nitrogen, and hyperbilirubinemia (Table 2). The EKG showed sinus tachycardia and left axis deviation, with no evidence of acute ischemic changes. Contrast-enhanced computed tomography (CT) of the abdomen and pelvis revealed hepatic steatosis, fecal impaction in the rectosigmoid region, and focal colitis in the sigmoid colon. The patient was administered 3 L of IV fluids and the serum lactic level decreased to 6 mmol/L. The patient was admitted to the ICU for septic shock and DKA. The patient was treated with vasopressor support, insulin infusion, electrolyte solutions, and broad-spectrum antibiotics.
A View in the Dark: Acute Esophageal Necrosis in the Setting of Diabetic Ketoacidosis.
Overnight, the patient developed peritonitis and melenic stools. A nasogastric tube (NGT) was placed on low intermittent suction for gastric decompression, and a rectal tube was inserted because of persistent melenic stools. A repeat abdominal CT scan showed worsening colitis without signs of bowel perforation. The surgical service was consulted due to concerns for acute mesenteric ischemia, but they found no objective evidence of ischemia on imaging. They disimpacted the patient at the bedside and recommended an aggressive bowel regimen. Bedside EGD revealed a “black esophagus,” consistent with AEN (Figure 3), and the patient was managed conservatively with PPIs, carafate suspension, and insulin infusion. On day 4 of admission, the patient’s stool became yellowish, and the hemoglobin level remained stable. A clear liquid diet was initiated and advanced as tolerated, and the patient was discharged on day 11 of admission in stable condition. A relook endoscopy after 8 months showed normal esophageal mucosa (Figure 4).
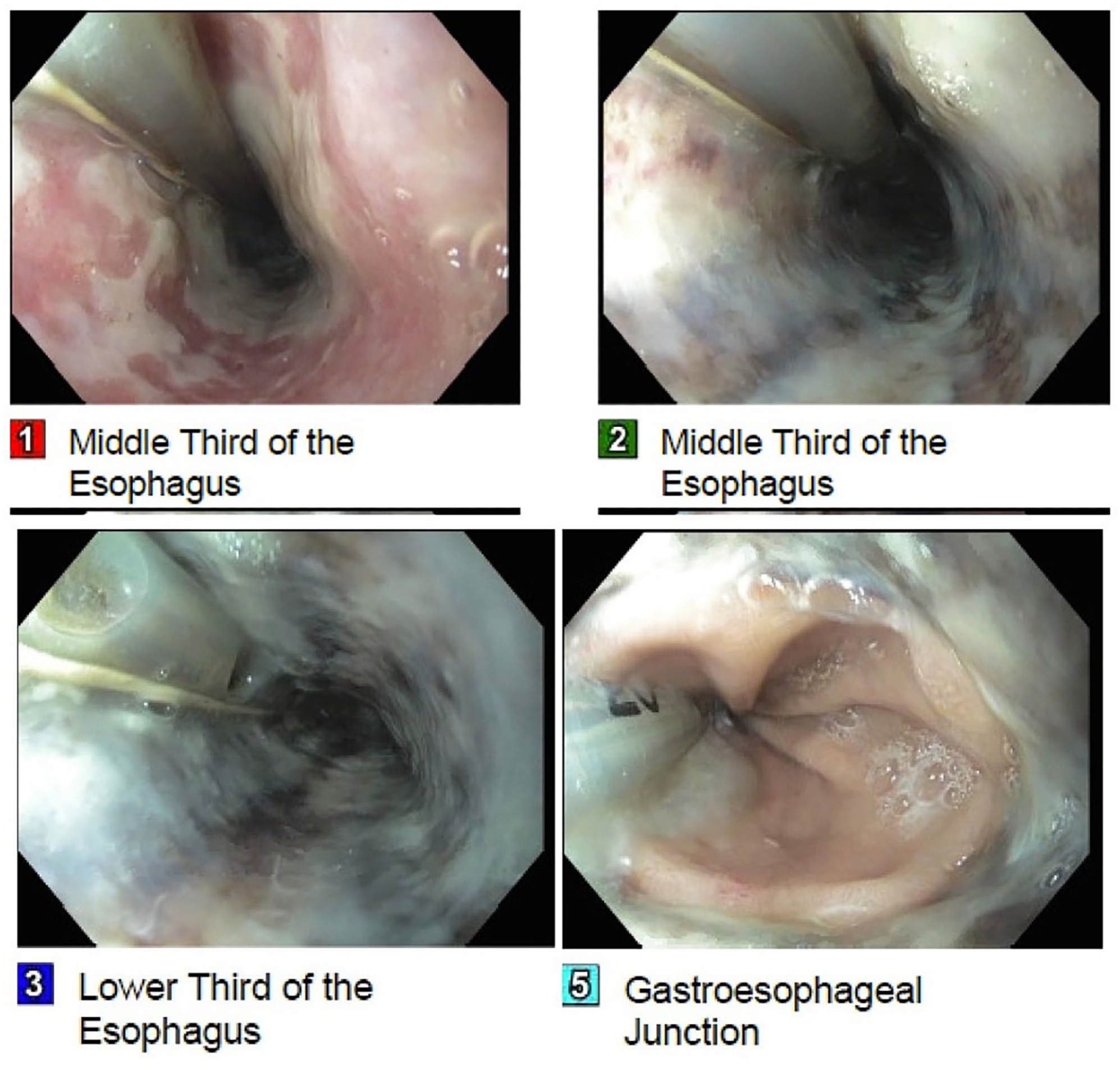
Endoscopic images for case 2 (during hospitalization) showing diffuse dark discoloration and ulceration of the upper esophagus. The cardia/fundus, duodenal bulb, and second portion of duodenum were normal.

Endoscopic images for case 2 (8 months after discharge) showing normal esophageal mucosa.
Discussion
Acute esophageal necrosis is a rare condition characterized by a striking black discoloration of the esophageal mucosa, typically seen during EGD. The necrosis usually starts in the distal esophagus and abruptly transitions to normal mucosa at the gastroesophageal junction (GEJ).1,2 Acute esophageal necrosis is more prevalent in critically ill male patients in their sixth decade of life with multiple comorbidities.8-11 The cause of AEN is multifactorial, with risk factors including DKA, sepsis, malnutrition, chronic renal disease, cardiovascular disease, alcohol abuse, and male gender.2,9 Acute esophageal necrosis is thought to result from a combination of esophageal ischemia, impaired mucosal barrier function, and reflux of gastric contents, leading to necrosis of the esophageal mucosa. 2 In this series, we share 2 cases of AEN in the setting of DKA managed with supportive care.
Acute esophageal necrosis is an extremely rare complication of DKA that may result from poor tissue perfusion, mucosal barrier dysfunction, and corrosive injury. The profound osmotic diuresis in DKA limits the blood supply to watershed areas such as the distal esophagus, predisposing them to ischemia and necrosis. 11 In addition, diabetes mellitus promotes atherosclerosis, which potentiates tissue ischemia in affected patients. 6 Signs and symptoms include nausea, vomiting, coffee-ground emesis, acute dysphagia, abdominal pain, and melena.2,6,9,11 Patients may also experience odynophagia, eructation, pyrosis, and regurgitation.2,9,12 Triage blood tests may show acute blood loss anemia, leukocytosis, and hyperlactatemia.5,13 In severe cases, physical examination may show signs of sepsis or shock, characterized by tachycardia, tachypnea, hypotension, and fever. 12 Abdominal guarding and tenderness may also be observed on examination. Complications of AEN include esophageal stenosis, strictures, perforation, mediastinitis, peristaltic abnormalities, and abscess formation.7,14
The diagnosis of AEN is usually based on endoscopic findings of circumferential black esophageal mucosa that abruptly terminates at the GEJ.11,13,15 Other possible endoscopic findings include different bleeding sites and presence of gastric and duodenal ulcers. 13 The CT scans will show thickening of the distal esophagus with or without esophageal perforation secondary to the associated inflammation. 11 However, it is important to note that CT may show nonspecific changes depending on the symptom onset, disease course, and involvement. 11 An important diagnostic finding to be aware of is signs of occlusions of the splenic or left gastric arteries which can also impact the blood supply to the distal esophagus.8,11,15 Although the causes of AEN vary, the fundamental pathophysiology and histological appearance are the same. The foundation is based on the hemodynamic instability that is caused by the underlying insult to the body, which compromises the blood flow to the distal esophagus. 1 Histologically, AEN presents with mucosal necrosis with the absence of viable epithelium and a brisk increase in inflammatory infiltrate. 1 Depending on the insult and the time frame of hypoperfusion, the necrosis can extend into the muscularis propria. Although helpful at times, a biopsy is not essential for diagnosing AEN.
The management of AEN is directed at correcting the underlying insult and hemodynamic resuscitation to ensure adequate blood flow to the distal esophagus.8,11,15 Supportive measures include nil per os (NPO) status, IV hydration, IV PPIs, and short-term parenteral nutrition.16,17 In addition, sucralfate reduces local insult to the esophagus from gastric acid, thus aiding the healing process. 16 Acute esophageal necrosis associated with DKA also requires electrolyte repletions and insulin infusions to achieve euglycemia. A prolonged course of oral PPIs even after symptom resolution prevents esophageal stricture and stenosis. 12 Enteral tube placement should be avoided, given the increased chances of perforation. 10 In case 2, an enteral tube was inadvertently placed and should have been avoided. Prophylactic antibiotics should be limited to patients with signs of sepsis, septic shock, esophageal perforation, or mediastinitis.7,16 In cases of co-existing infectious esophagitis, a specific antimicrobial therapy should be started to hasten healing. 6 The patient described in case 1 had comorbid Candida esophagitis, which was effectively treated with IV fluconazole.
Surgical intervention is usually reserved for patients with esophageal perforation, or mediastinitis. 10 In these cases, self-expandable stents (SEMS) or intrathoracic drainage systems have proved effective. 6 Adrenaline sclerotherapy can be employed in patients with active bleeding. 12 Balloon tamponade with a Sengstaken-Blakemore tube is contraindicated due to the increased risk of esophageal perforation.6,10,12 Repeat EGD is warranted to monitor for improvement within 1 month as the esophageal mucosal recovery occurs over a period.6,12 Our patients underwent relook endoscopies a few months after their hospitalization, and both studies were unremarkable. Endoscopic balloon dilation is the primary treatment for strictures, although some patients may require multiple dilation sessions and, in severe cases, esophagectomy and esophageal bypass may be necessary. 6
Conclusion
Acute esophageal necrosis is an infrequent complication of DKA and is characterized by extensive black discoloration of the esophageal mucosa. Acute esophageal necrosis is an endoscopic diagnosis, and prompt treatment of the underlying etiology can avert further complications such as esophageal perforation or mediastinitis. The patient described in case 1 was admitted for DKA requiring insulin therapy and aggressive hydration. This condition along with the atrial fibrillation potentially predisposed the patient to developing AEN. Similarly, the patient in case 2 was admitted for DKA and septic shock. Although DKA is the likely cause of his AEN, sepsis-induced hypotension may have also contributed to the limited blood supply to the distal esophagus.
Footnotes
Author Contributions
LB conceptualized the idea for this case series. RB, DP, TW, NM, and GM assisted with data curation, collection of pertinent patient data, and writing the manuscript. YC and WB edited, fact-checked, and proofread the final version of the case series.
Data Availability Statement
Further inquiries can be directed to the corresponding author.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patients for their anonymized information to be published in this article.
