Abstract
We present a case of Brugada syndrome in a 74-year-old patient who presented with urine retention and incidentally found to have non–sustained ventricular tachycardia (NSVT) on electrocardiogram (ECG) and telemetry. To reveal characteristic type 1 Brugada pattern, right-pericardial lead was placed in the third right intercostal space. No antiarrhythmics were started, a loop recorder was implanted, and on follow-up episodes of self-terminating sustained ventricular tachycardia (VT) were noted. The patient was started on quinidine with resolution of VT.
Introduction
Brugada syndrome (BrS) is a rare but serious genetic disorder that alters cardiac action potential, leading to an increased risk of ventricular arrhythmias and sudden death. It is named after the Spanish cardiologists who first described it in 1992 and is characterized by a distinctive pattern on an electrocardiogram (ECG) known as the “Brugada sign.” Symptoms of BrS can vary widely and may include syncope, palpitations, and sudden cardiac arrest. The condition is usually diagnosed through a combination of clinical evaluation, family history, and genetic testing. Treatment in patients with syncope is implantable cardioverter-defibrillator (ICD). The role of antiarrhythmics is limited, and quinidine is effective in suppressing ventricular arrhythmia episodes.
Case Report
A 74-year-old male with a history of diabetes, hypertension, and chronic obstructive pulmonary disease (COPD) presented to the emergency department with urinary retention. There was no personal or family history of syncope or sudden cardiac death (SCD), and he denied substance or alcohol use. During his hospitalization, he remained asymptomatic and hemodynamically stable. Electrolytes and cardiac biomarkers were normal. Echocardiogram showed normal systolic function and wall motion. The initial ECG showed non–sustained ventricular tachycardia (NSVT), and lead V1 demonstrated subtle ST elevation (see Figure 1).

Initial electrocardiogram (ECG). ECG 1 demonstrates non–sustained ventricular tachycardia (NSVT). Lead V1 also shows ST elevation, raising suspicion for Brugada pattern (BrP).
The differential diagnosis for NSVT with ST elevation in lead V1 includes Brugada pattern (BrP) ECG, left ventricular hypertrophy (LVH), left bundle branch block (LBBB), or NSVT related to ST segment elevation myocardial infarction in the left anterior descending territory or left main disease. In this case, due to the absence of typical chest pain, normal cardiac enzymes, lack of wall motion abnormalities or LVH on echocardiogram, and a narrow QRS complex on ECG, BrP was suspected.
The patient remained completely asymptomatic with runs of NSVT. A repeat ECG was performed after moving the right-pericardial lead to a higher point (third right intercostal space), which showed type 1 BrP (see Figure 2). Patient was initially treated by hospitalist team with intravenous amiodarone without decrease in episodes of NSVT. Recognizing BrP and asymptomatic status amiodarone was stopped.

Repeat electrocardiogram (ECG). ECG 2 demonstrates type 1 Brugada pattern (BrP) after moving the right, precordial leads to the third right intercostal space.
A defibrillator was also not indicated due to the absence of any malignant symptoms such as syncope or a family history of SCD. Instead, an implantable loop recorder (ILR) was implanted prior to discharge to monitor VT burden. During clinic follow-up, the ILR interrogation revealed runs of self-terminating sustained VT (see Figure 3). Quinidine therapy was initiated, which resulted in the resolution of VT (see Figure 4).
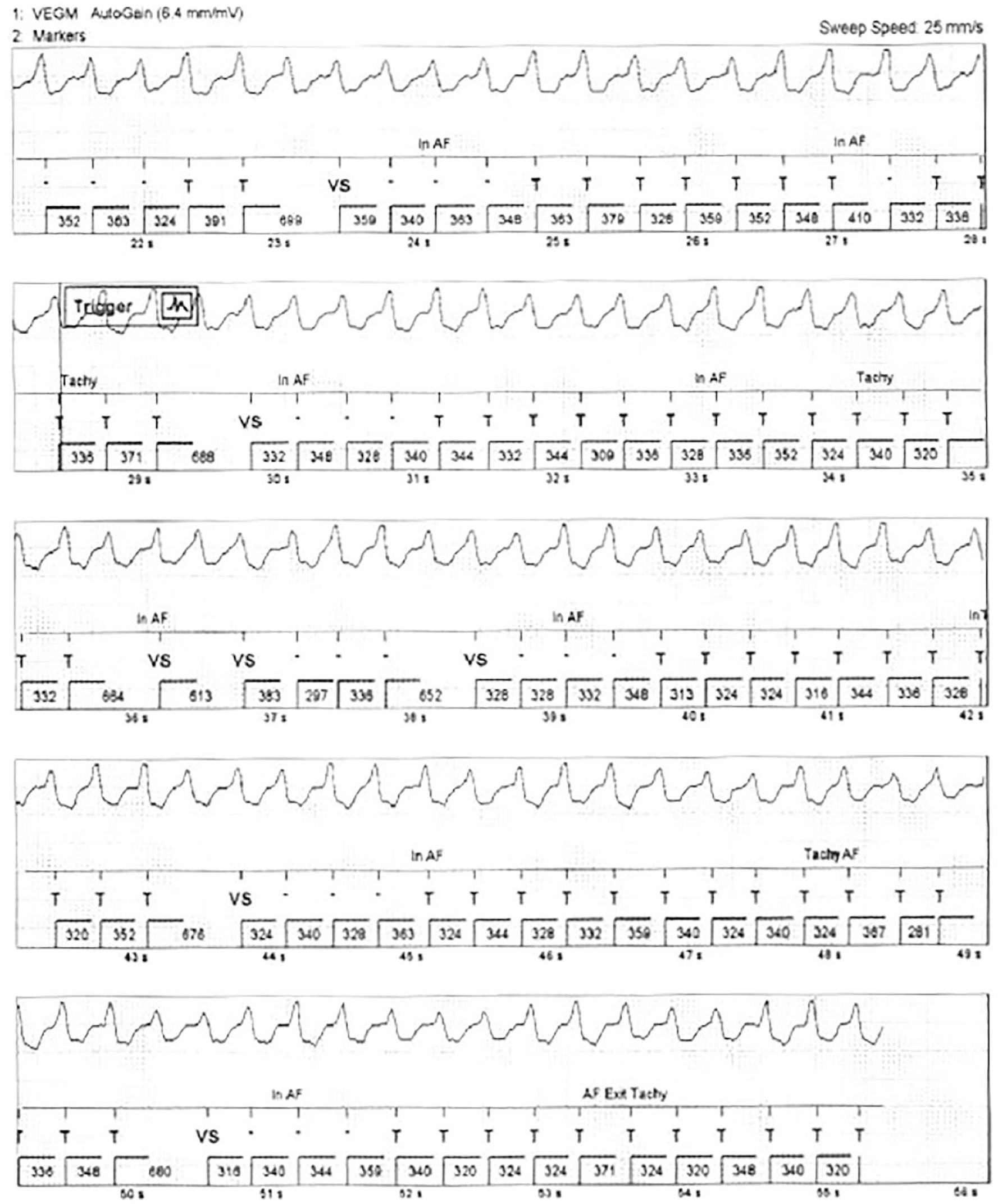
An ILR recording demonstrated sustained ventricular tachycardia (VT) before the initiation of quinidine therapy.

An ILR recording after quinidine therapy, which demonstrated a significant resolution of ventricular tachycardia (VT). It is worth noting that the ILR recording shows a sinus rhythm with premature atrial contractions (PACs) that were misinterpreted as atrial fibrillation (Afib).
Discussion
Brugada syndrome 1 type 1 is a rare autosomal-dominant loss-of-function mutation in the SCN5A gene that codes for cardiac Na+ channels, resulting in a characteristic ECG pattern in the right precordial leads known as the BrP. Most patients are asymptomatic. Brugada syndrome can cause ventricular tachycardia (VT), ventricular fibrillation (VF), and SCD. 2 Acute stressors or drugs can unmask the typical ECG features and precipitate arrhythmia.
Brugada pattern ECG and BrS are usually diagnosed in adulthood. In 2 larger series of patients with BrP ECGs and those with symptomatic BrS, the average age was 41 years.3,4
Brugada syndrome should be suspected when ST elevation in V1/V2 alone is present. If diagnostic criteria are unmet, a drug challenge (ajmaline, flecainide, and procainamide) or repositioning ECG leads may reveal a diagnosis. Brugada syndrome can remain undiagnosed until later in life as a significant percentage of patients remain asymptomatic. Defibrillator implantation is indicated for prevention of SCD in symptomatic patients. The use of conventional antiarrhythmics can exacerbate arrhythmias. Quinidine may benefit BrS patients with high VT burden. 5 Careful risk stratification and consideration of quinidine trial in select group of patients are warranted because excessive ICD shocks can occur with NSVT. 2
Quinidine works by blocking ion channels in the heart, including the sodium and potassium channels, which can help to mitigate atrial and ventricular arrhythmias. However, quinidine can also cause a variety of side effects, including nausea, diarrhea, headache, dizziness, and even potentially life-threatening arrhythmias. Therefore, it is important to closely monitor patients who are taking quinidine.
In BrS, quinidine is often used to reduce the burden of VT, in patients with ICD. By reducing the incidence and severity of VT, quinidine can help to minimize inappropriate shocks for NSVT, prevent sudden VF arrest, and improve overall outcomes for patients with BrS. However, as with any medication, there are risks and benefits to using quinidine, and it is important for patients and health care providers to carefully weigh these factors when considering treatment options.
Ablation is rarely performed for Brugada storm. Current practice guidelines give ablation a class IIb recommendation for treatment of patients with Brugada storm. Prior studies proposed the use of endocardial ablation of focal premature ventricular complexes (PVCs) within the right ventricular outflow tract (RVOT) or the right-sided His-Purkinje system as a means of preventing VT/VF episodes in BrS patients. Subsequent animal studies suggested that radiofrequency (RF) ablation of the epicardial surface of the RVOT may be more effective in eliminating VT in BrS. Radiofrequency ablation of epicardial ventricular foci may prevent future VT/VF episodes in BrS patients. Newer insights into the pathophysiology of ventricular arrhythmias in BrS has led to use of other ablation targets such as late potentials, as well as low-voltage, fractionated electrograms on voltage mapping. 6
Follow-up
The patient continues to follow up with the cardiology clinic and remains asymptomatic with no reported episodes of syncope, dizziness, or palpitations for 2 years since first contact.
Conclusions
Brugada syndrome is a rare but potentially life-threatening channelopathy, which can present with variable clinical and ECG findings. In elderly patients, acute stressors or drugs can sometimes mask the typical BrP on ECG, and alternative mechanisms such as channelopathies should be considered in cases of polymorphic VT. Repositioning ECG leads to a higher intercostals space can unmask the typical BrP. Most antiarrhythmic medications are contraindicated in BrS, as they can exacerbate ventricular arrhythmias. As seen our patient amiodarone was ineffective. Quinidine therapy was well tolerated and effective in our patient with high NSVT burden and obviated the need for an implantable cardioverter-defibrillator.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient for their anonymized information to be published in this article.
