Abstract
Cardiac disease associated with cancer treatment is a common adverse effect that is well-treated with appropriate monitoring. However, some cardiac adverse effects with cancer treatment are not well-understood, in particular rituximab-associated ventricular tachycardia. We present the fourth case of rituximab-associated ventricular tachycardia in a patient who is rituximab-naive and who does not have known cardiac disease history. This patient developed non-sustained polymorphic ventricular tachycardia 14 hours after rituximab was started and 6 hours after it was stopped, and after extensive monitoring including a 30-day event monitor, did not develop further significant runs of ventricular tachycardia.
Introduction
Cancer therapy with rituximab is a very common treatment especially for hematological and lymphatic malignancies. Rituximab is a monoclonal antibody with anti-CD20 activity often used to treat lymphatic malignancies including non-Hodgkin’s lymphoma and chronic lymphocytic leukemia. Rituximab is also used in some non-malignant conditions including rheumatoid arthritis and granulomatosis with polyangiitis. 1 Rituximab is administered by intravenous infusion, and for the indication of diffuse large B-cell lymphoma (DLCBL), the dose is 375 mg per meter squared body surface area. For the first infusion (ie, rituximab-naive), the infusion rate is started at 50 mg/h. If there are no infusion-related reactions, the infusion rate is increased by 50 mg/h every 30 minutes to a maximum of 400 mg/h. For the second infusion or for patients who have received rituximab previously, the infusion rate is started at 100 mg/h. If there are no infusion-related reactions, the infusion rate is increased by 100 mg/h every 30 minutes to a maximum of 400 mg/h. 2 To help prevent infusion-related reactions, it is recommended to give acetaminophen and antihistamine prior to each rituximab infusion. 1 Additional adjuncts to rituximab plus cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) therapy include methotrexate for patients deemed to be at high risk for relapse of disease in the central nervous system.3,4
The cardiac side effects are not very well-understood, and in particular, its association with arrhythmias is being recently described in the literature. One review has presented evidence that the incidence of supraventricular tachycardia was higher in the group received rituximab in addition to standard cyclophosphamide, doxorubicin, vincristine, and prednisone (CHOP) therapy vs CHOP therapy alone. 5 There are cases of other non–ventricular tachycardia (VT)-related events which appear to affect all parts of the heart (conduction, structural, coronary circulation) including irreversible bradycardia, 6 non-ischemic cardiomyopathy, 7 coronary vasospasm, 8 and coronary artery occlusion and associated ST-elevation myocardial infarction. 9
The occurrence of VT after rituximab is not very well-understood. There have been only 3 cases documented in the literature.10-12 Interestingly, the onset of the ventricular tachycardia in all these cases is quite variable, ranging from within 30 minutes of the start of infusion 11 to as long as several days later on the 8th dose. 10 There is currently no described mechanism of why rituximab may be associated with arrhythmias. We present here the fourth case in the literature of rituximab-induced VT in a patient who is rituximab-naive and whose only known cardiac history is asymptomatic sinus bradycardia.
Case Presentation
A 75-year-old male presented to the hospital for initiation of chemotherapy for DLCBL. The oncologist and patient met within 24 hours prior to this current hospital admission to discuss treatment. The pathway to diagnosis seems to have started since 4 months ago, when the patient actually was diagnosed with a presumed gastric cancer due to the finding of a 27-mm gastric mass on computed tomography (CT) imaging from an outside facility at the time. He underwent upper endoscopy and biopsy for possible gastric cancer but was then lost to follow-up. Computed tomography angiography imaging 3 weeks later revealed a growth of the 27-mm gastric mass to 70 mm (at largest diameter) and revealed a 111-mm upper retroperitoneal mass encasing the celiac artery, superior mesenteric artery, left renal artery and vein, and left adrenal gland; in addition, there were 2 subcentimeter lung nodules in the right lung. The patient was unfortunately lost to follow-up and did not see an oncologist.
Previous records showed DLCBL was diagnosed after a left adrenal mass biopsy with pathology confirming high-grade DLCBL and cytogenetic studies (fluorescence in situ hybridization) showing MYC rearrangement without BCL2 or BCL6 arrangement. The patient presented with significant weight loss (30 pounds within the last few months), night sweats, and dark stools. Given the significant symptoms and aggressive disease, it was agreed to admit the patient to the hospital to start R-CHOP regimen with intrathecal methotrexate administered for central nervous system relapse prophylaxis.
We on the cardiology service were consulted due to episodes of VT on day 3. He has a medical history of prostate cancer post-prostatectomy, type 2 diabetes not on insulin, hypertension, hyperlipidemia, and gastroesophageal reflux disease. He was evaluated by a cardiologist for a pacemaker 9 years prior for having a “heart rate too low.” With the lowest heart rate being 48 beats per minute and the patient’s being asymptomatic, pacemaker was not indicated. He does recall a “fainting episode” 27 years prior in which he lost consciousness for about 2 hours, and the exact cause is unknown; he did not undergo cardiac work-up. He otherwise has no other history of cardiac arrhythmias including atrial fibrillation, and no other history of myocardial infarction/anginal symptoms throughout his life and particularly the last 2 to 3 months. The patient is a former smoker, having smoked 2 packs per day for 30 years but quit 34 years prior. He drinks alcohol socially but does not binge drink. He has never used recreational drugs. There is no family history of premature coronary artery disease, sudden cardiac death, heart failure, or arrhythmia.
The patient presented to the emergency room on the evening of day 1 and had an uneventful evening. On day 2 evening, the patient started cancer therapy—this involved initiation of rituximab and 1 dose of methotrexate; the exact dosing schedule is seen in Table 1 and visualized in Figure 1. Rituximab was prepared as an intravenous infusion at a dose of 375 mg/m2, and using the patient’s weight of 68.9 kg at the time multiplied by the calculated 1.8 m2 body surface area, the final rituximab dose was calculated as 675 mg. Rituximab was ordered as a 10-mg/mL solution and so 675 mg dose was calculated to be 67.5 mL of solution (rounded to 70 mL). 70 mL of rituximab (using seven 10 mg/mL vials) was placed in 630 mL of dextrose 5% solution to make for a rituximab 700 mg in 700 mL in dextrose 5% solution, which was planned for administration. Of note, 23 minutes prior to the start of rituximab, pre-treatment with oral acetaminophen 650 mg and intravenous diphenhydramine 25 mg were given. On day 2, approximately 21 hours after the once-off methotrexate dose, and approximately 14 hours after rituximab was started and 6 hours it was stopped, the telemetry unit called the oncologist notifying of “13 beats of ventricular tachycardia” (Figure 2). The patient did not report symptoms including chest pain, shortness of breath, palpitations, dizziness, or lightheadedness; he was not witnessed to lose consciousness by his wife at the bedside. Vitals taken at the time showed temperature 36.7°C, heart rate 55 beats per minute (sinus bradycardia), blood pressure 103/66, respiratory rate 18 breaths per minute, and SpO2 97% on room air. Further plans for chemotherapy were stopped at this time until cardiology consultation.
Chemotherapy (Rituximab and Methotrexate) Doses Over Time.

Rituximab dose is recorded as infusion rate over time. Heart rate and cardiac events are recorded as reported by nursing or telemetry unit.

Rituximab infusion rate over time with cardiac events (black circles) corresponding to the events seen in Table 1. Cardiac events occurred at time t = 25, 25.5, and 38 hours.
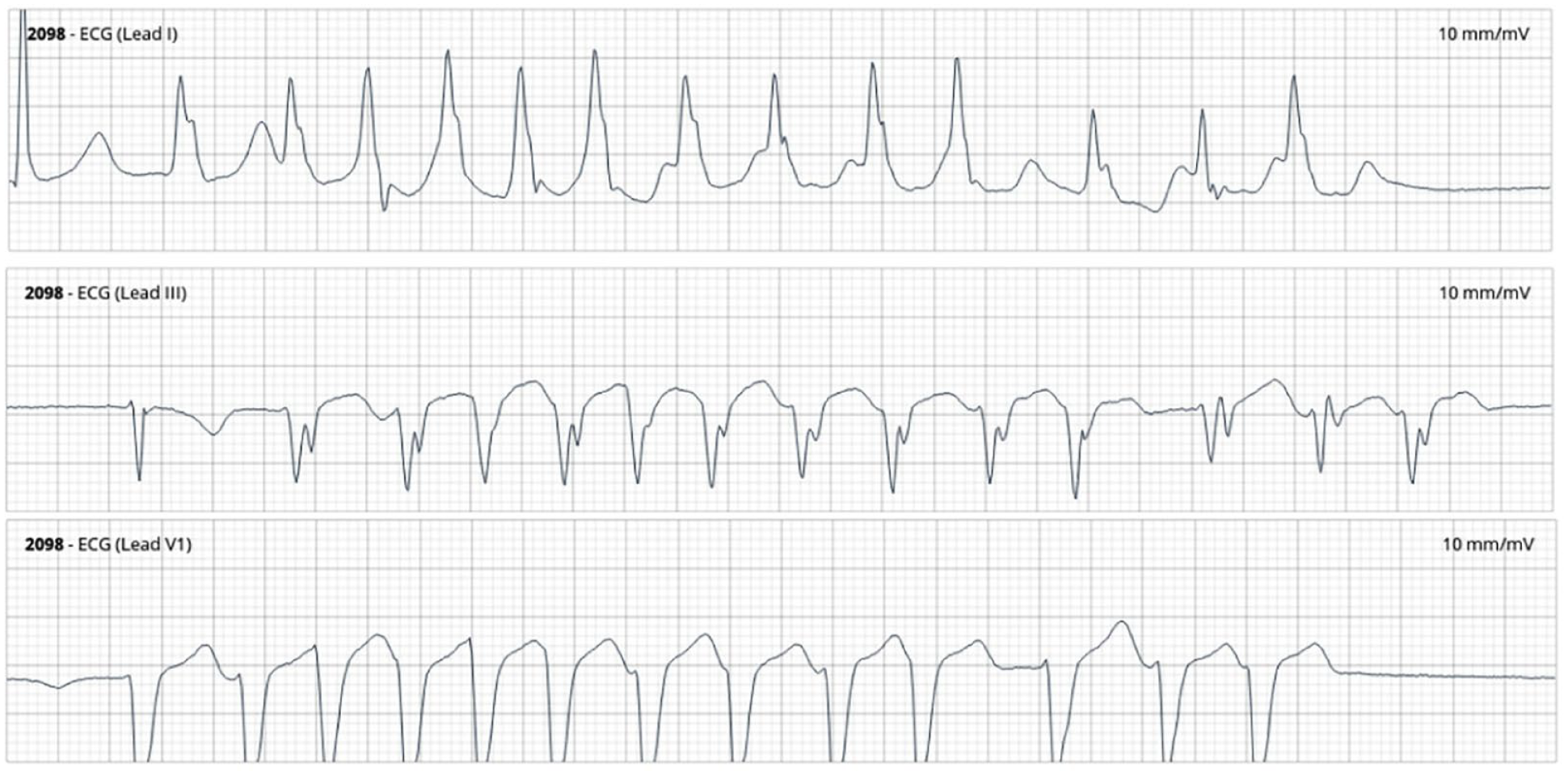
Telemetry strips of ventricular tachycardia event at t = 38 hours. Leads are noted in the top left corner, including lead I, III, and V1. The total strip duration is 10 seconds long. There are 13 beats of approximately 4 seconds of non-sustained polymorphic ventricular tachycardia. No fusion complexes or capture beats are noted.
We evaluated the patient approximately 2 hours after the episode of VT. Vitals were similar as above. On examination, the patient was alert and oriented to time, person, place, and situation. There were no sternotomy scars or pacemaker/defibrillator scars on the chest wall. The jugular venous pressure was not appreciated at 30 degrees with the hepatojugular reflux maneuver. Auscultation revealed normal breath sounds without crackles or wheezes; the S1 and S2 heart sounds were of normal intensity and character, and no added sounds or murmurs were appreciated. There was no lower limb edema. For further history, we spoke to his wife at bedside and sister over the phone. The patient initially came in feeling very well, and per his sister, “he was eating with a good appetite, and felt strong enough to get chemo.” He had no complaints of chest pain, shortness of breath, palpitations, dizziness, or lightheadedness on presentation. When the patient started rituximab on day 1, approximately 60-80 minutes after the rituximab infusion started, the patient’s sister (corroborated by nursing documentation) noted the patient started to look uncomfortable, cold, slightly shaky, and asked for more blankets. The rituximab infusion was stopped immediately and intravenous diphenhydramine was administered within 10 minutes and the patient’s shakes stopped. Approximately 30 minutes later, the rituximab was started at 25 mg/h and continued until completion for 6 more hours, with the final infusion rate being 150 mg/h for a final infusion concentration of approximately 0.21 mg/mL for the last 3 hours of the infusion. Approximately 6 hours post-rituximab completion, telemetry notified the oncologist of “13 beats of ventricular tachycardia” (Figure 2).
The telemetry strip was reviewed and the rhythm was determined to be non-sustained (approximately 4 seconds) polymorphic ventricular tachycardia. Further telemetry strips were reviewed and it was observed that the 13 beats of VT occurred during a 30-second window with another run of 4 betas of non-sustained polymorphic VT, further followed by PVCs (Figure 2). Further events of shorter runs of non-sustained polymorphic VT also occurred (Figure 3). Apparently, even within hours prior to the rituximab infusion starting, another nurse had noted the patient had some infrequent pre-ventricular complexes (PVCs), and that approximately 1-2 hours into the rituximab infusion, there was some ventricular bigeminy. Unfortunately, a thorough search of the telemetry saved events did not reveal this, and prior ECGs did not demonstrate PVCs, so this could not be verified. For comparison, there was a post-VT ECG (Figure 4) and ECG 3 months prior to the current admission (Figure 5) that showed sinus bradycardia without PVCs.

Telemetry strips of ventricular tachycardia (VT) event at t = 38 hours. Leads are noted in the top left corner, including lead I, II, and V1. The total strip duration is 30 seconds long (3 rows of 10 seconds each). In the first row of each lead, there are 13 beats of approximately 4 seconds of non-sustained polymorphic VT. No fusion complexes or capture beats are noted. In the second row of each lead, there are 4 beats of approximately 1 second of non-sustained polymorphic VT, followed by 2 pre-ventricular complexes (PVCs). In the third row of each lead, is a PVC, followed by a sinus beat, followed by ventricular bigeminy, followed by a sinus beat.

ECG recorded 36 minutes after event of non-sustained ventricular tachycardia. ECG shows sinus bradycardia without pre-ventricular complexes.
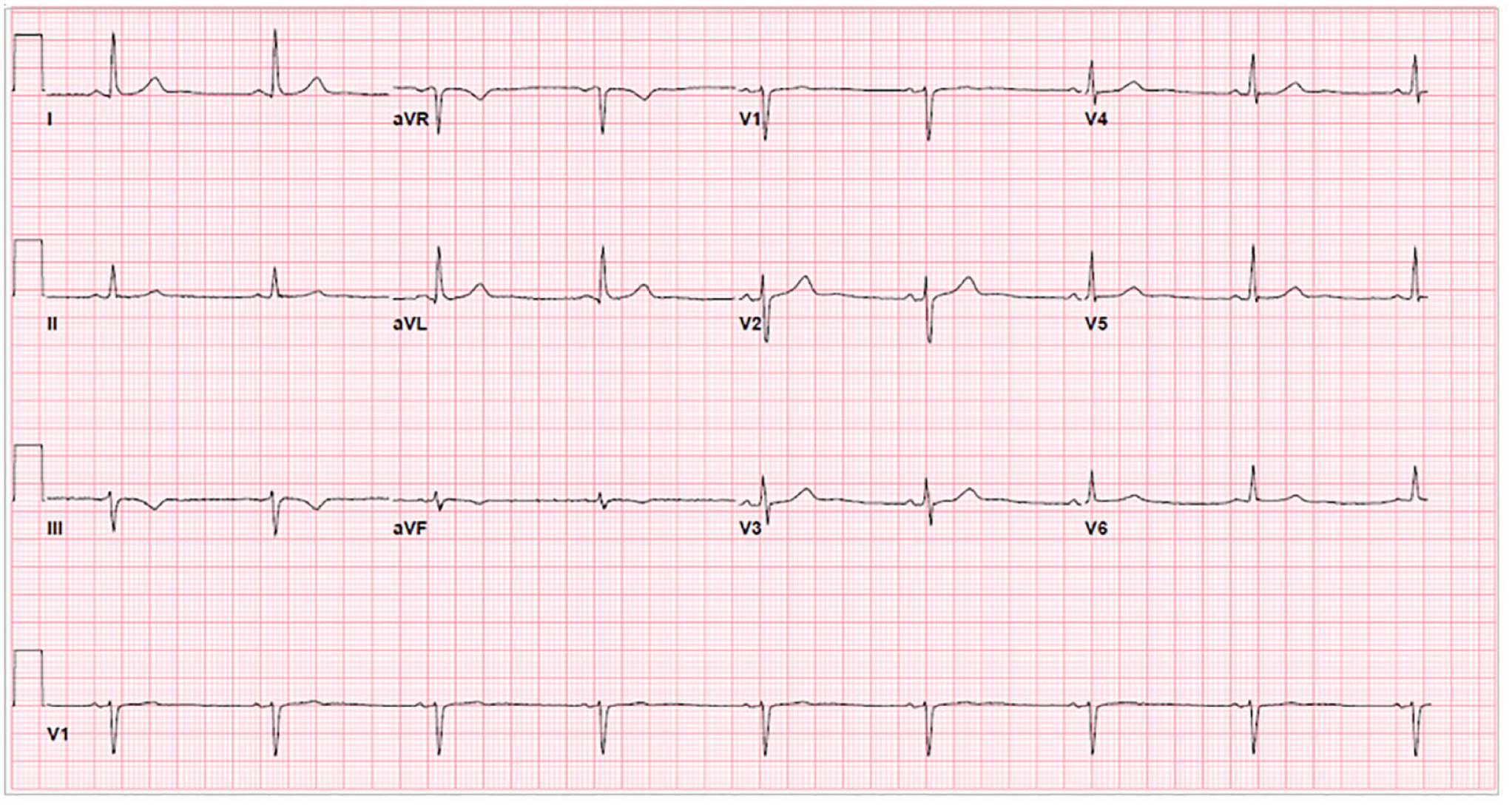
ECG recorded 3 months prior to current admission. ECG shows sinus bradycardia without pre-ventricular complexes.
A transthoracic echocardiogram (TTE) was performed on the day of admission, and there was no evidence of structural heart disease or reduced systolic function or impaired diastolic function. A nuclear myocardial perfusion stress test did not reveal significant fixed or reversible ischemia.
The patient was monitored during the rest of the admission as per usual protocol had no more VT events and had a minimum heart rate of 50 beats per minute. The patient received the rest of the R-CHOP regimen with prednisone 100 mg on days 3 and 4 for a total dose of prednisone 200 mg and cyclophosphamide 1350 mg total (dose of 750 mg/m2). The patient was then discharged.
In an outpatient setting, the patient had a 30-day event monitor. There was no atrial arrhythmia. There were 3 patient-triggered events correlating with normal sinus rhythm and sinus bradycardia. There was 1 automatic trigger demonstrating 4 beats of non-sustained ventricular tachycardia. Ventricular ectopy burden was 1%, supraventricular ectopy burden was less than 1%, tachycardia was 2% of the time, and bradycardia was 49% of the time.
Discussion
We present this patient who demonstrates the fourth known documented case specifically of VT associated with known rituximab infusion. There is clear evidence that the VT is most likely attributable to the rituximab infusion, given the VT occurred after the rituximab, and after remaining off the rituximab, there were no other significant VT runs other than a short 4-beat non-sustained VT that occurred only once on a 30-day cardiac event monitor.
Our case differs from the other 3 known cases of rituximab-associated VT as described further. Arai et al describe the first case of rituximab-associated VT in 2005. This case involved a 79-year-old male being treated with rituximab for mantle cell lymphoma, without known cardiac disease history, with pulmonary emphysema on baseline home oxygen therapy. The patient had an episode of 9 beats of non-sustained monomorphic VT during the eighth infusion of rituximab. Rituximab was immediately discontinued, and no more arrhythmias occurred. 10 Both our patient and the patient in Arai et al did not have known significant cardiac disease history. Interestingly, the patient in Arai et al developed monomorphic VT, whereas our patient developed polymorphic VT. However, the patient in Arai et al did receive 8 doses of rituximab. Whether a dose-dependent relationship between rituximab and VT risk exists or there is simply no relationship at all as seen in our rituximab-naive patient is hard to determine.
Poterucha et al describe the second case of rituximab-associated VT in 2010. This case involved a 79-year-old female with significant cardiac disease history, including atypical left atrial flutter, atrioventricular nodal ablation, ventricular pacing, mild left ventricular systolic dysfunction (left ventricular ejection fraction 48%), and non-obstructive coronary artery disease. The patient was being treated for B-cell CD20 positive malignant lymphoma. This patient was rituximab-naive, and after 30 minutes into the initial rituximab infusion, the patient was witnessed to have a 30-second syncopal episode. The pacemaker was interrogated and revealed 12 seconds of polymorphic VT of heart rate 290 beats per minute, which occurred during the onset of the syncopal episode. Electrolyte levels were normal. Rituximab was immediately discontinued, and no more arrhythmias occurred. 11 Both our case and the patient in Poterucha et al describe the potential for cardiac adverse effects in even rituximab-naive patients, although the patient in Poterucha et al certainly had significant cardiac disease history.
Quinones et al describe the third case of rituximab-associated VT in 2023. This case involved a 21-year-old male being treated for warm autoimmune hemolytic anemia. The patient had no known cardiac disease history and had received rituximab 1 week prior without cardiac adverse effects. However, 30 hours post-completion of the second rituximab infusion, the patient was reported to have multiple episodes of non-sustained polymorphic VT and PVCs on telemetry. Electrolyte levels were normal. Rituximab was immediately discontinued, and no more arrhythmias occurred. 12 Both our case and the patient in Quinones et al did not have known significant cardiac disease history. However, the patient in Quinones et al did receive 1 dose of rituximab 1 week prior.
So far in the 3 cases described above, 2 cases of polymorphic VT were noted, 2 cases describe previous rituximab exposure, and 1 case describes extensive previous cardiac disease history. Our patient developed non-sustained polymorphic VT in the setting of no previous rituximab exposure, and he did not have significant cardiac disease history (other than known sinus bradycardia without underlying structural heart disease or conduction disease).
Certainly, there is no shortage of papers describing the relationship between chemotherapy and cardiotoxicity.13-15 Buza et al 15 provide a systematic overview of the causes of arrhythmias in cancer patients on treatment, which involves anything from electrolyte disturbances from simple causes such as vomiting, to iatrogenic cardiac structural disease and associated myocardial ischemia and systolic dysfunction.
We are still unable to propose a mechanism behind rituximab being associated with the cases of VT described above in our case and the other 3 cases in the literature. Nonetheless, we did find that getting the baseline ECGs and TTE prior to rituximab treatment did allow us to diagnose rituximab-associated VT and rule out underlying structural heart disease or conduction disease. We recommend this as ongoing standard of care in the absence of a known mechanism of rituximab-associated VT, and perhaps, more caution needs to be taken regarding cancer treatment given the increasing prevalence of cardiovascular disease. 16
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
