Abstract
Loperamide is an easily accessible antidiarrheal medication. Unlike other medications in its class, loperamide is unique in that it causes euphoria at supratherapeutic levels due to its effect on opioid receptors. Unfortunately, with its growing abuse potential also comes increasing reports of cardiotoxicity including prolonged QT, torsades de pointes, and sudden cardiac death. We report a case of a 29-year-old female who presented with unstable arrhythmia that further progressed into electrical storm in the setting of loperamide toxicity. Due to its growing popularity and availability, it is important for clinicians to understand loperamide’s mechanisms for causing toxicity as well as how to appropriately treat its complications.
Keywords
Introduction
Loperamide is a common over-the-counter antidiarrheal. It primarily acts on peripheral µ-opioid receptors, but unlike other µ-receptor agonists, loperamide has less central nervous system (CNS) activity. When the medication was developed, it was initially listed as a schedule II medication but later marketed as a nonprescription medication in 1988. This was based on loperamide’s lower abuse potential relative to other medications in the same class, which is attributed to low bioavailability in the CNS. 1
With the emergence of the opioid epidemic and the rising number of opioid-related deaths, there have been increasing reports of loperamide being used as alternatives to prescription opioids. Between 2008 and 2016, there were 179 cases of loperamide abuse reported to the National Poison Data System with more than 50% being reported after 2014. At higher than recommended doses (50-300 mg), loperamide has been shown to cross the blood-brain barrier more readily, providing its consumer with both psychotropic and euphoric effects. 2
In addition to its CNS effects, high doses of loperamide also act on the cardiac myocytes. Although incompletely understood, loperamide is thought to have a dose-dependent antagonistic effect on the calcium, sodium, and inward-rectifier potassium channels, resulting in delayed repolarization. Multiple conduction abnormalities have been reported including prolonged QTc, ventricular tachycardia (VT), ventricular fibrillation (VF), torsades de pointes, wide complex tachycardia, and even sudden cardiac death. Since loperamide’s approval in 1976, 48 cases of serious cardiac complications have been reported to the Federal Drug Administration (FDA). Of these 48, 10 patients died and 31 were hospitalized. 2 This report highlights a patient admitted to Loma Linda University Medical Center for loperamide intoxication and subsequent VT storm.
Case Report
A 29-year-old female with history of heroin abuse and depression presented after being found altered and confused. At the time, the patient was known to be taking an estimated 300 tablets of loperamide daily for “chronic stomach issues” (1 tablet is 2 mg for an approximate total of 600 mg). Her husband reported that within the past year, he would witness her taking at least one entire bottle of loperamide with roughly 96 tablets up to 3 times per day. There were no other medications reported, including use of any antidepressants.
On presentation, she was tachycardic to 156 BPM (beats per minute) and hypotensive to 70/40 mm Hg. Initial electrocardiogram (ECG) showed polymorphic VT with prolonged QTc of 669 ms (Figures 1 and 2). Subsequently, she developed recurrent episodes of VT that degenerated into torsades de pointes, resulting in multiple cardioversions and her admission to the cardiac intensive care unit.
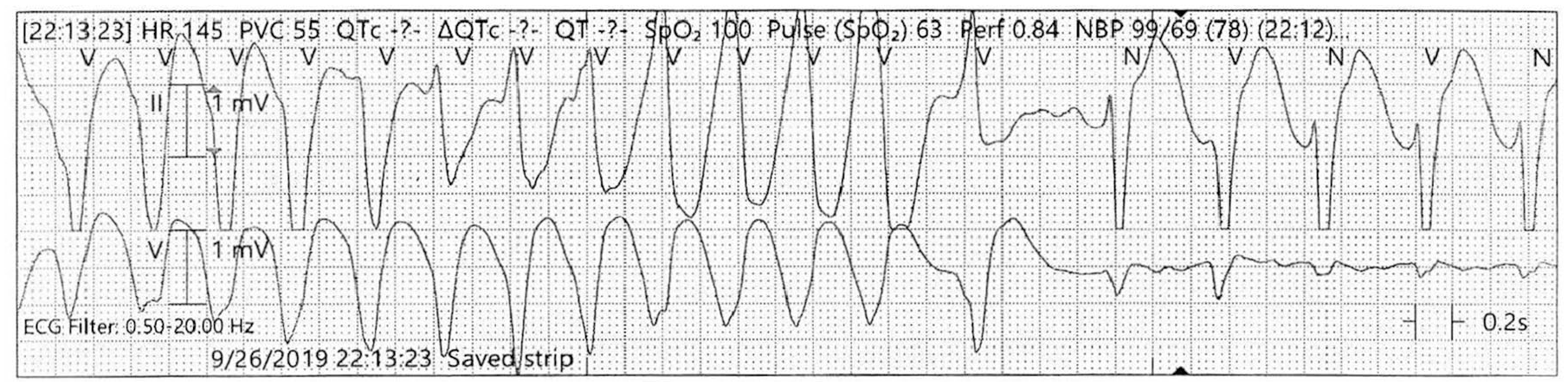
Electrocardiogram rhythm strip done on route showing ventricular tachycardia.

Continuous cardiac monitoring in the emergency room showing polymorphic ventricular tachycardia.
Preliminary laboratory findings were unremarkable except for the following: leukocytosis 18 bil/L, anion gap 16, and lactate 2.2 mmol/L. Chemistries showed a sodium 136 mmol/L, potassium 4.3 mmol/L, calcium 2.2 mmol/L, and magnesium 1.6 mmol/L. Urine drug screen was positive for cannabinoids, and her chest X-ray was normal. A serum loperamide level was ordered but would not result until after her discharge.
After cardioversion in the emergency room, the patient was stabilized. Repeat ECG shortly after demonstrated a QTc of 515 ms. Poison control was contacted immediately and recommended supportive management. Unfortunately that evening, she began having frequent episodes of nonsustained VT triggered by positional change and vomiting (Figure 3). Additional intravenous magnesium sulfate was given but symptoms persisted. A sodium bicarbonate infusion at 150 mEq/h was initiated per the recommendations of the electrophysiologist on call.

Recurrent nonsustained ventricular tachycardia before anti-arrhythmics.
Several hours later, the patient further decompensated into sustained VT. Electrical cardioversion with 120 J was required to maintain hemodynamic stability. Given her tenuous status and recurrent VT storm, she was intubated and sedated to suppress sympathetic overstimulation. Subsequently, an isoproterenol infusion was started at 2 µg/min to reduce the number of subsequent VT episodes.
After 8 hours without VT, isoproterenol was reduced to 1 µg/min and the bicarbonate infusion was discontinued. Her transthoracic echocardiogram was completed and shown to be normal. Isoproterenol was stopped on day 3, and the patient was extubated. By then, she was free of VT for more than 24 hours and her QTc improved to 500 ms. No additional anti-arrhythmics were started, and the patient was downgraded and later discharged from the hospital (Figure 4).

Sinus bradycardia after anti-arrhythmics.
The final results of her serum loperamide and desmethyl loperamide (the primary metabolite of loperamide) were elevated to 26 ng/mL (normal is <10 ng/mL) and 160 ng/dL (normal is <20 ng/dL), respectively.
Discussion
Loperamide is a common antidiarrheal that acts on peripheral µ-opioid receptors. Unlike other µ-receptor agonists, loperamide has less CNS activity. 3 When it was first developed, loperamide was listed as a schedule II medication. By 1988, loperamide was marketed as a nonprescription medication because of its low abuse potential relative to other medications within its class. 1
Despite its low abuse potential, there have been increasing reports of loperamide being substituted for other opioids. Between 2008 and 2016, there were 179 cases of loperamide abuse reported to the National Poison Data System with more than 50% being reported after 2014. 2 Loperamide is a substrate for the P-glycoprotein transporter found in the intestine and CNS. At appropriate doses, the transporter moves loperamide from the cytosol of the vascular endothelial cells into the vascular lumen, resulting in less absorption through the blood-brain barrier. Supratherapeutic levels of loperamide (50-300 mg) cause the P-glycoprotein transporter to become overwhelmed, allowing more loperamide to be absorbed into the CNS and providing its user with a sensation of euphoria.4-6
In addition to its CNS effect, high doses of loperamide also act on the cardiac myocytes. Loperamide has a dose-dependent antagonistic effect on calcium, sodium, and inward-rectifier potassium channels, resulting in delayed repolarization. Multiple conduction abnormalities have been reported, including prolonged QTc, VT/VF, torsades de pointes, and even sudden cardiac death.3,7 Since loperamide’s approval in 1976, 48 cases of serious cardiac complications have been reported to the FDA. Of these 48, 10 patients died and 31 were hospitalized. 2 As a result, the FDA placed a black box warning for torsades de pointes, cardiac arrest, QT prolongation, and death.1,3,8,9
Loperamide’s cardiotoxicity comes from inhibiting sodium and potassium channels. Two channels involved are the hERG voltage-gated potassium channel and the NaV1.5 sodium channel. The hERG voltage-gated potassium channel is responsible for the delayed rectifier current and affects repolarization. The NaV1.5 sodium channel is responsible for fast depolarization during the ventricular action potential. Inhibition of both these channels results in prolonged QT. Loperamide also affects the endothelial release of calcium by modifying calmodulin and decreasing the influx of intracellular calcium that can cause longer QT, hypotension, and bradycardia.3,8,9
Treating loperamide toxicity is mainly supportive and includes the following: advanced cardiopulmonary life support, electrolyte management, sodium bicarbonate, anti-arrhythmic medications, and potentially hemodialysis. Narcan can be used to reverse the opioid effects by competitively antagonizing the µ-receptors. Magnesium cations maintain the gradient between sodium and potassium moving through their respective channels via the magnesium-dependent Na-K-ADPase. By having an appropriate gradient, myocardial stabilization is achieved and the likelihood of arrhythmogenesis is reduced. 10 Intravenous sodium bicarbonate works by decreasing sodium channel blockade. This helps drive sodium through both open and closed sodium channels. Sodium bicarbonate also increases pH levels, which inhibits loperamide’s ability to bind to sodium channels. 7
As for arrhythmia suppression, amiodarone is a class III anti-arrhythmic with following properties: prolongation of the action potential by acting on electrolyte channels reduced AV conduction and inhibition of adrenergic stimulation. Lidocaine is a class Ib anti-arrhythmic that acts by inhibiting sodium channels. Isoproterenol is an inotropic and chronotropic medication that acts on both β-1 and β-2 adrenergic receptors. By increasing heart rate and decreasing repolarization, isoproterenol reduces the QT interval and accelerates atrioventricular nodal conduction.7,10 Mechanical ventilation and sedation help reduce the sympathetic surge, thereby decreasing the trigger for VT storm. In the setting of severe acidosis and other electrolyte derangement, hemodialysis can be used to directly remove loperamide.
Amiodarone and lidocaine were avoided in this scenario to minimize the risk of further QTc prolongation and additional sodium channel blockade. Isoproterenol had no effect on either and thus became the obvious treatment choice. Narcan was not used because patient’s cognition and cardiorespiratory functions were normal. Hemodialysis was never required since her cardiac function improved on isoproterenol.
Conclusion
Loperamide toxicity is a growing concern in health care due to its accessibility and abuse potential. As our case demonstrates, these patients should be closely monitored for cardiac toxicity with telemetry and serial ECGs. In these particular patients who use loperamide for its abuse potential, there is the possibility for other drugs to be in their system that could also induce cardiac arrhythmias. Although unlikely in this situation given the significant dose of loperamide ingested and her urine drug screen being positive for cannabinoid only. Initial management should include contacting poison control, correcting electrolytes, and cardiopulmonary support. Sodium bicarbonate and an appropriate anti-arrhythmic should be started immediately. Mechanical ventilation and sedation reduce sympathetic surge during the VT storm. Hemodialysis should be considered if the patient’s clinical status does not improve.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Ethical approval to report this case was obtained from the institutional review board (IRB# 5200351).
Informed Consent
Informed consent for patient information to be published in this article was not obtained because the institutional review board determined that this activity did not meet the definition of human subject research and no personal identifiers were used.
