Abstract
Calcium channel blocker ingestions remain one of the leading causes of death related to cardiovascular medication ingestion in both adults and pediatric patients. We report a case of a 17-year-old, 103 kg female presenting after an intentional polypharmacy ingestion, including 500 to 550 mg of amlodipine. She presented with profound vasoplegia and cardiovascular collapse requiring high-dose inotropes and eventual life support with extracorporeal membrane oxygenation (ECMO). Current available treatments, designed for adults, including lipid emulsion and methylene blue, provided no sustained clinical improvement. This resulted in the initiation of single-pass albumin dialysis (SPAD). We aim to describe the clinical implications, amlodipine toxic dose effects, and clinical challenges associated with large pediatric patients and high-dose medications. We also discuss several challenges encountered related to dosing and concentration of medications, which led to fluid overload. Given the ongoing obesity epidemic, we routinely see pediatric patients of adult size. This will continue to challenge pediatric use of adult dosing and concentrations to avoid excessive fluid administration for high-dose medications, such as insulin and vasoactive agents. To our knowledge, this is the first successful case of using SPAD in conjunction with ECMO for salvage therapy after refractory life-threatening calcium channel blocker toxicity.
Introduction
Calcium channel blockers (CCBs) belong to a commonly prescribed cardiovascular class of drugs with a broad list of indications, such as hypertension, arrhythmia, heart failure, angina, and migraine. Calcium channel blocker poisoning has the highest degree of mortality of all cardiovascular medications. 1 The mechanism of action includes decreased calcium entry into the cell through blockade of voltage-gated calcium channels. 2 There are multiple types of calcium channel receptors in the body. The 2 types associated with the cardiovascular system are the L-type (long-lasting), found in skeletal and smooth muscle cells, and the T-type (transient), which are found in any cell that has pacemaker activity. 3 Amlodipine targets the L-type receptor leading to reduced electromechanical conduction resulting in bradycardia, reduced contractility, vasodilation, and hemodynamic collapse. 4
First-line management of CCB poisoning includes fluid administration, intravenous calcium, vasoactive medications, and high-dose insulin. Rescue therapies include lipid emulsion, glucagon, and extracorporeal membrane oxygenation (ECMO) support.5-9 In the pediatric population, even small doses can have fatal consequences,10-12 but management still uses adult strategies.7-9,13 Routine methods of dialysis for removal of CCB have not been successful due to the protein-bound nature of CCBs.4,5,8-10 Current EXTRIP recommendations are against extracorporeal treatments (ECTR); however, their data are limited evaluating molecular adsorbent recirculating system (MARS) or albumin dialysis. MARS or single-pass albumin dialysis (SPAD) may provide a therapeutic option as a salvage therapy following CCB ingestion.6,14-16 We present a case of pediatric amlodipine toxicity with refractory cardiovascular collapse and successful treatment of SPAD. We aim to describe the clinical implications of amlodipine toxicity, clinical and pharmacologic challenges with adult-sized pediatric patients, and successful treatment strategies.
Case
A 17-year-old, 103 kg female was presented to an outside hospital roughly 3 hours after intentional overdose of multiple substances, including unknown amounts of acetaminophen, ibuprofen, amoxicillin, along with an estimated 500 to 550 mg of amlodipine. Initially oriented, she became unresponsive during a seizure episode and was subsequently intubated. Fluid resuscitation, calcium infusion, and an insulin infusion at 0.1 U/kg/h were initiated for hypotension, with improvement in her hemodynamics. She was transferred to Children’s Hospital & Medical Center (CHMC) for ongoing care. Doses of epinephrine and norepinephrine were initiated and escalated during transport for ongoing hypotension. On arrival, patient vitals included heart rate of 133 bpm, cuff blood pressure of 86/44 mmHg, oxygen saturation of 95% on an inspired fractional oxygen of 1.0, and a peak end expiratory pressure of 10 on the ventilator. The respiratory rate was 24 with end-tidal carbon dioxide of 60. Glucagon and sodium bicarbonate infusions were initiated on arrival to CHMC, along with escalation of insulin and maximizing the calcium infusion. An arterial line was placed, and the blood pressure was 70/32 mmHg with heart rate in the 130s. Initial arterial blood gas showed a pH of 7.04, PaCO2 of 59, PaO2 of 68, and base deficit of 15.8 mEq/L. The patient’s hemodynamics continued to worsen, and venoarterial ECMO was initiated 9 hours after arrival.
Hypotension with mean arterial blood pressures between 42 and 55 mm Hg persisted after ECMO initiation and despite high-dose epinephrine, norepinephrine, and vasopressin. A lipid emulsion provided only 12 hours of hemodynamic stability. Methylene blue was trialed with no improvement in hemodynamics. The patient developed severe fluid overload with skin tears following ongoing resuscitation and high volume rates due to the high doses of inotropes and insulin (roughly 10 L/d), hemodynamic intolerability of diuretics, and acute kidney injury. Renal replacement therapy was initiated on hospital day 3 as SPAD to facilitate clearance of amlodipine. Single-pass albumin dialysis was performed in series with ECMO using a continuous renal replacement therapy (CRRT) machine (NxStage System One machine; NxStage Medical, Lawrence, Massachusetts). Continuous veno-venous hemodialysis (CVVHD) was performed using a CAR-505 filter (polyethersulfone membrane with a 1.6-m2 surface area). The blood flow rate was 200 mL/min and dialysate flow rate was 2500 mL/h (2000 mL/1.73 m2/h). The albumin dialysate was created to a final albumin concentration of 2.5%. No changes were made to systemic heparin anticoagulation.
The patient’s hemodynamic status improved with 2.5% SPAD; however, the hospital’s supply was quickly consumed, and we were forced to reduce to 1.25% SPAD to avoid stopping altogether. The patient continued to be unstable on lower albumin concentration, despite escalation of insulin to 7 U/kg/h and maintaining other support, with mean arterial pressures in the low 40s. After supply from other institutions arrived, the albumin dialysate concentration was increased back to 2.5% at approximately 40 hours of SPAD, and the patient’s clinical status improved. Between hospital days 5 and 9, all her vasoactive agents were able to be stopped, along with the glucagon, insulin, and calcium infusions. The patient received a total of 117 hours of SPAD, and the patient was followed by an additional 23 hours of conventional CVVHD for fluid removal. Table 1 shows relevant laboratory testing results.
List of laboratory values.
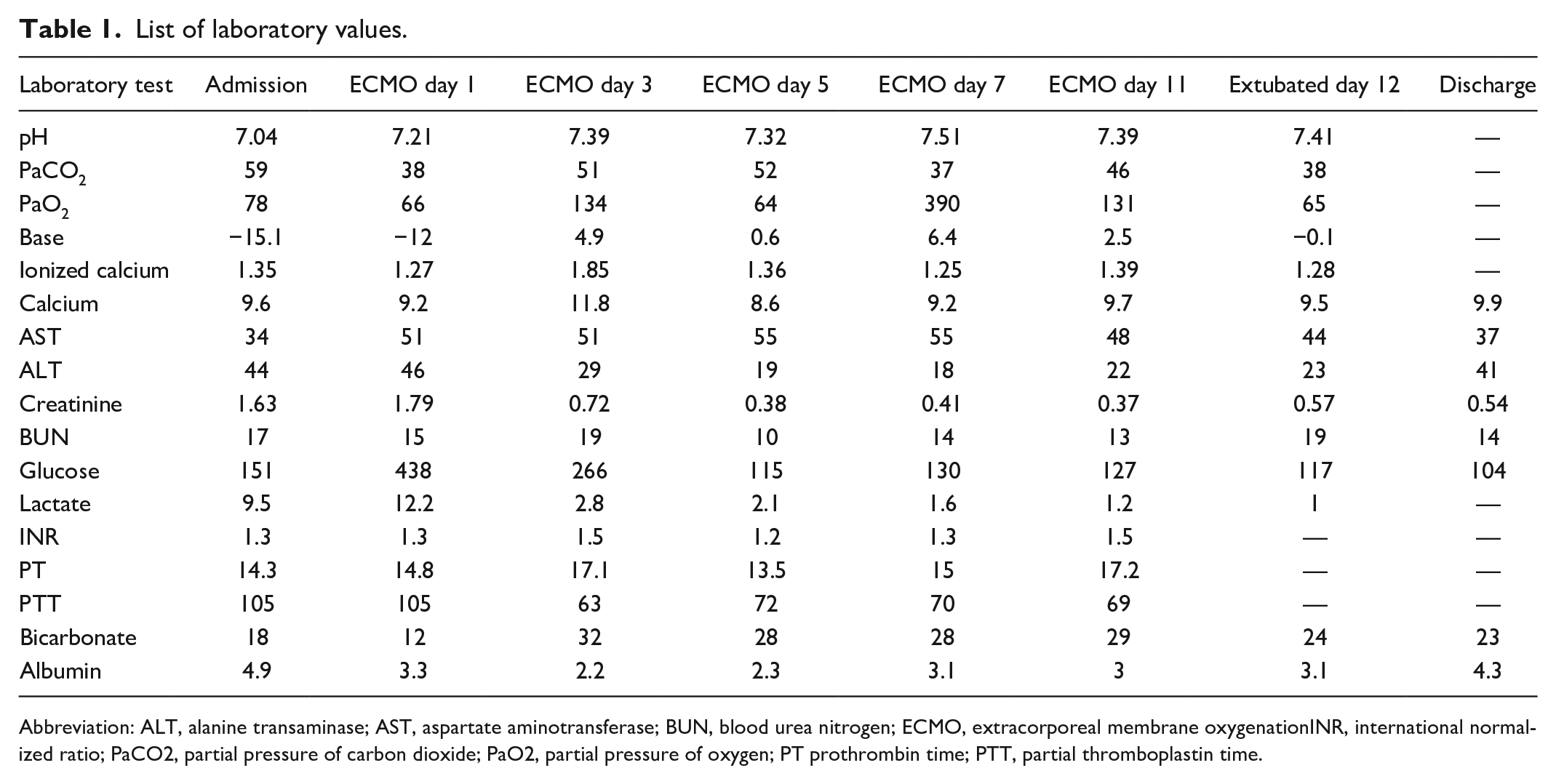
Abbreviation: ALT, alanine transaminase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; ECMO, extracorporeal membrane oxygenationINR, international normalized ratio; PaCO2, partial pressure of carbon dioxide; PaO2, partial pressure of oxygen; PT prothrombin time; PTT, partial thromboplastin time.
She remained on ECMO to facilitate lung recruitment secondary to severe pulmonary edema and fluid overload. On day 11, she was decannulated from ECMO, weaned off all inotropes, and extubated on day 12. After extubation, she was evaluated by psychiatry for intentional overdose along with her previous diagnosis of schizoaffective disorder. She was transferred out of the intensive care unit on day 16 and discharged on day 26 to an acute rehabilitation center for ongoing management or her physical and psychological rehabilitation. The only known medical complication is a stroke with multiple small infarcts resulting in right-sided visual defects. Figure 1 shows her vasoactive agent dosing and blood pressure related to therapeutic interventions.

Timeline from admission through ECMO decannulation and inotropic support with epinephrine (blue), norepinephrine (orange), and vasopressin (green) with interventions (red bars). Until the dialysate was increased to 2.5%, there was no clinical improvement. Other therapies failed to provide sustained clinical improvement. Delayed decannulation due to fluid overload and pulmonary edema.
Discussion
Use of MARS and SPAD on CCB Ingestion
The choice of therapy for removing a toxic substance depends on its molecular size, charge, binding characteristics, volume of distribution, patient size, and availability of extracorporeal techniques. As CCBs are highly protein bound and have a large volume of distribution, they are poorly removed by conventional hemodialysis. Molecular adsorbent recirculating system has been shown beneficial in cases of highly protein-bound CCB toxicity, with reduced vasopressor requirements noted shortly after initiation.4,14,16-19 Pinto et al reported the case of a pediatric patient treated with MARS in tandem with ECMO. 15 They demonstrated reduction in CCB levels and improvements in hemodynamic instability after each procedure. Calcium channel blocker levels were noted to rise between MARS treatments due to rebound effect as blood-tissue drug equilibrium occurs. Unlike MARS which utilizes specialized circuit and filters to recycle albumin, SPAD is a modification of the standard CRRT circuit where albumin is added to the dialysate. Free drug crosses the dialyzer membrane, binding to albumin causing a diffusion gradient allowing more drug to be eliminated. 20 This method has been used for protein-bound drugs in cases of pediatric overdoses of phenobarbital, carbamazepine, valproic acid, acetaminophen, and methotrexate.20-24 Yeşilbaş et al described a case where SPAD was used efficaciously in a pediatric patient with severe verapamil intoxication. 14 Single-pass albumin dialysis offers multiple benefits over other extracorporeal therapies, such as the need for less technical and infrastructure support. 25 As it does not require the use of specialized circuits or filters, it can be performed at any pediatric center with CRRT capabilities. As MARS and charcoal hemoperfusion are not available in many pediatric institutions, SPAD may be the only therapy available for patients critically unstable to transfer to another center. We have shown that SPAD can safely be performed in adjunct with extracorporeal life support.
Pharmacologic Issues
Several challenges were encountered from a pharmacy perspective, including dosing and concentration challenges and managing the albumin dialysis workload. The maximum norepinephrine concentration at our facility is 16 µg/mL, whereas norepinephrine concentrations up to 64 µg/mL have been shown safe in adults. In addition, use of nonstandard concentrations of medications allows for a variety of errors at the bedside. Due to these potential errors, it was determined not to use a higher concentration of vasoactive agents and insulin which contributed to her significant fluid overload of more than 50 L. Given the rising obesity epidemic in the United States, we routinely see pediatric patients that are adult size. This will continue to be a challenge as adult dosing and concentrations may be necessary to avoid life-threatening fluid overload from administration of high-dose medications, such as insulin and vasoactive agents that are high volumes if not maximally concentrated.
Finally, the preparation of albumin dialysate is labor-intensive and costly.23,24 The optimal concentration of albumin necessary to achieve benefit while limiting unnecessary excess albumin is currently unknown. Churchwell et al showed a relationship between the albumin concentration and the clearance of valproic acid and carbamazepine, but not phenytoin. 23 Although drug levels were not directly measured in our study, we saw a strong correlation in hemodynamic status and albumin concentration between 2.5% and 1.25%. The supply of albumin became a barrier as we were only able to prepare 24 hours’ worth of the 2.5% albumin dialysis solution. To temporarily extend the inventory, the albumin dialysis solution was changed to 1.25% resulting in further hemodynamic instability. Once adequate supply was obtained, the albumin dialysis was increased to 2.5%, with subsequent improvement in hemodynamics, weaning of vasoactive infusions, and eventual decannulation from ECMO.
Conclusion
To our knowledge, this is the first description of the use of SPAD in tandem with ECMO. For our patient, SPAD improved hemodynamic parameters and did not add any additional burden. The use of albumin dialysis in the management of CCB is controversial and is not currently recommended by expert groups due to the lack of clinical pharmacokinetic data.8,25 Despite the EXTRIP recommendation, 25 multiple studies have shown clinical improvements in CCB following albumin dialysis.4,14-17,19-25 In addition, the EXTRIP guidelines promote a strong recommendation but with a very low quality of evidence. Although plasma and dialysate drug concentrations were not measured in our study, we saw a correlation between albumin concentration of SPAD and clinical improvement. Although albumin dialysis is not the current standard practice in the treatment of CCB toxicity, we have demonstrated SPAD use as a salvage therapy in cases of life-threatening hemodynamic compromise and that SPAD may constitute an adjunctive treatment to extracorporeal life support. While most ECTR would not work for highly protein-bound substances, SPAD and MARS are specifically designed for protein-bound substance removal. Given this patient’s survival, despite all other therapies, including ECMO, further studies to evaluate the role of SPAD and gather pharmacokinetic data in severe CCB ingestion are warranted.
Footnotes
Authors’ Note
Prior Presentation of Abstract: Case Abstract presented at Midwestern Clinical & Translational Research Meeting in Chicago, IL on March 3, 2022 and March 4, 2022.
Author Contributions
Drs Essink, Sankey, Montange, and Ms Berg conceptualized the case report, drafted the initial case report, and reviewed and revised the case report.
Drs Taylor and Salomon conceptualized the case report, drafted the initial case report, coordinated and supervised data collection, reviewed and revised the case report, and critically reviewed the case report for important intellectual content.
All authors approve the final case report as submitted and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
