Abstract
Primary pulmonary artery sarcomas (PPAS) are extremely rare tumors that are often mislabeled as pulmonary emboli (PE). PPAS usually involve the pulmonary trunk and are histologically classified as leiomyosarcoma, spindle cells sarcoma, fibrous histiocytoma, or undifferentiated sarcoma. Our case involved a 78-year-old man with an undifferentiated PPAS confined to the left pulmonary artery that was initially misdiagnosed as a PE. After a month-long delay in treatment in which the patient was prescribed warfarin, the correct diagnosis was made. Pulmonary artery endarterectomy and left lung pneumonectomy were performed, and he survived for 18 months before disease recurrence and death. Our case helps illustrate some of the clinical and radiographic findings that help distinguish PPAS from PE.
Introduction
The estimated incidence of primary pulmonary artery sarcomas (PPAS) is 0.001% to 0.030%, and only about 400 cases have been reported in the literature.1,2 PPAS are classified according to their histological appearance and are characterized primarily as leiomyosarcoma, spindle cells sarcoma, fibrous histiocytoma, or undifferentiated. 2 These rare tumors are often initially misdiagnosed as pulmonary emboli (PE). 3 Treatment delays due to misdiagnoses are associated with worse outcomes but can often be avoided by recognizing radiographic differences that distinguish PAS from PE. 3
Case Description
Our patient was a 78-year-old male with a past medical history significant for hypertension who initially presented with cough, fatigue, and weight loss. Computed tomography angiography (CTA) of the chest was performed and demonstrated a mass occluding the left main pulmonary artery, which was diagnosed as a PE (Figure 1). He was admitted and started on a heparin drip, which was later transitioned to warfarin. The patient continued to have worsening fatigue and weight loss despite anticoagulation. Follow-up CT a month later showed that the mass in the left main pulmonary artery had grown and the patient was referred to our clinic.

Computed tomography angiography demonstrating occlusion of the left pulmonary artery.
Endobronchial ultrasound with biopsy was performed via the left mainstem bronchus and demonstrated vimentin-positive malignant epithelioid cells. Positron emission tomography (PET)-CT on February 14, 2019, showed a metabolically active mass at the left main pulmonary artery with a standardized uptake value (SUV) of 6.0 (Figure 2) distinguishing the lesion from a pulmonary embolism. Transthoracic echocardiogram was unremarkable, with no systolic or diastolic dysfunction in the right or left heart and no regional wall abnormalities.
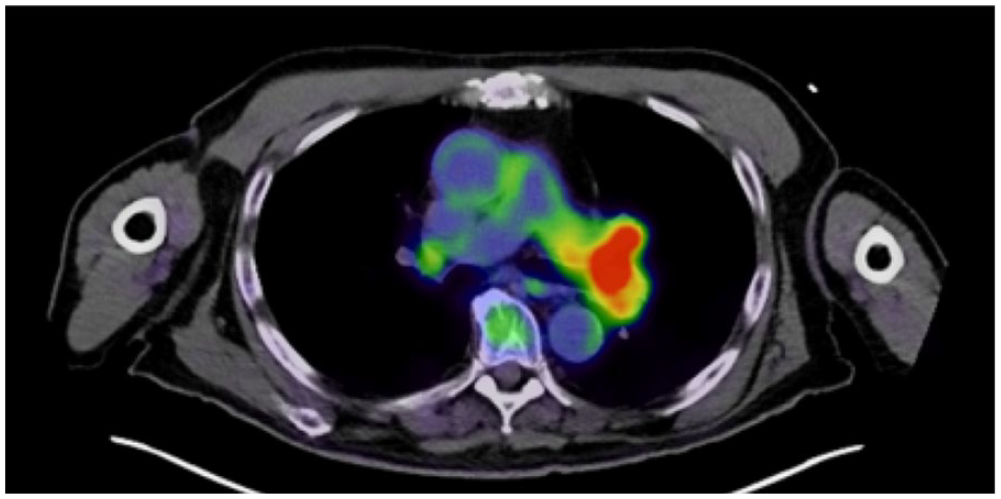
Positron emission tomography–computed tomography scan demonstrating fluorodeoxyglucose avidity of the left pulmonary artery mass.
Intrapericardial left lung pneumonectomy and left pulmonary artery endarterectomy were performed. An 8.2-cm tumor was removed from the intima of the left pulmonary artery. Pathology report revealed high-grade undifferentiated sarcoma within the pulmonary artery that extended to the bronchial wall and hilar soft tissue (Figure 3). All margins and sampled lymph nodes were negative. Tumor cells stained positive for vimentin and CD68, while fluorescence in situ hybridization (FISH) study showed MDM2 positivity. Adjuvant chemotherapy was not given due to the patient’s advanced age.

High-grade undifferentiated sarcoma within the pulmonary artery, 100× (left) and 200× (right). On the left, the pulmonary artery is identified with yellow arrows and the tumor cells are identified by red arrows. On the right, the magnified tumor cells (again identified with red arrows) display significant polymorphism. Necrotic debris is also visible and is identified with orange arrows.
Eighteen months later, the patient’s symptoms of cough, shortness of breath, and fatigue returned. CT scan demonstrated a large filling defect within left mid-pole extending into the right main pulmonary artery concerning for tumor thrombus with right heart strain (Figure 4). Transthoracic echocardiogram showed severe pulmonary hypertension with right ventricular systolic pressure of 82 mm Hg and right ventricular failure. Given these findings, a presumptive diagnosis of malignancy recurrence was made. Right pulmonary artery stent placement with aspiration of the mass was attempted. Unfortunately, the patient went into a ventricular tachycardia during the operation and died despite advanced cardiovascular life support.

Computed tomography angiography demonstrating filling defect within the pulmonary artery and disease recurrence.
Discussion
PPAS are uncommon tumors that are often initially mistaken for PE due to the clinical and radiological similarities of the conditions. 4 Unfortunately, laboratory findings are not useful in distinguishing PPAS from PE: D-dimer, brain natriuretic peptide, C-reactive protein, and erythrocyte sedimentation rate can be elevated in both cases. 5 Clinically, presentations may also be similar, though patients with PPAS are more likely to have gradual onset of symptoms, presence of systemic malaise, and absence of deep vein thrombosis. 6
Radiological findings—usually seen on CT scan—are most useful in distinguishing PPAS from PE. Unlike PE, PPAS more commonly occupy the entire vascular lumen, expand the pulmonary artery, and extend into the extraluminal space.7,8 PE are more likely to have multiple filling defects in the pulmonary arteries with peripheral emboli. 8 PE rarely occupy the entire vascular lumen and do not expand the pulmonary artery or extend into the extraluminal space. 8 On echocardiography, PPAS are more likely to show a mobile mass with bulging morphology, while PE more frequently appear as linear masses. 5 Cardiac-gated magnetic resonance imaging may help make the diagnosis of PPAS by demonstrating an intermediate-intensity mass with branches extending into the pulmonary vasculature. 9 More recently, combined PET/CT imaging has been successfully used to distinguish PPAS from PE and simultaneously stage the malignancy. 9
Regardless of the imaging modality initially used, definitive diagnosis requires biopsy. 9 PPAS are histologically classified as leiomyosarcoma, spindle cells sarcoma, fibrous histiocytoma, or undifferentiated sarcoma. 2 Histologically, our tumor was described as undifferentiated; roughly 10% of all PPAS are thought to be undifferentiated. 2
PE more frequently occludes of the distal branches of the pulmonary arterial system, while PPAS are almost always confined to the pulmonary trunk with or without involvement of the bases of the left and right pulmonary artery. In fact, 80% of PPAS involve the pulmonary trunk, 58% the left pulmonary artery, and 57% the right. 10 Both the left and right artery are involved in 37% of cases of PPAS, while 29% include the pulmonary valve and another 8% include the right ventricle. 10 Our patient’s tumor was unusual, as it involved left pulmonary artery but not the pulmonary trunk. This case demonstrates that although tumors usually involves the pulmonary trunk, PPAS should remain on the differential when a mass is confined to a single pulmonary artery.
Unfortunately, misdiagnosis of PPAS as PE is associated with worse outcomes. 2 In a 20-year observational analysis of PPAS, 47% were initially misdiagnosed as PE and 39% received thrombolytic/anticoagulation therapy; the odds of death increased by 46% for every doubling time from symptom onset to diagnosis. 2 Overall survival for PAS is poor. Survival for patients who undergo surgical resection is 2 to 3 years; for those who do not undergo surgery, median survival is just 6 to 12 months. 11 The use of adjuvant chemotherapy may improve survival: a retrospective review of 77 patients with PPAS over 17 years identified 6 patients who underwent adjuvant chemotherapy and 2 who received neoadjuvant chemotherapy. 11 Among these 8 patients, median survival time was 71 months. 10 Doxorubicin-based chemotherapy is usually the regimen of choice, though there are no trials to back its use. 12 Platinum-vinorelbine regimens have been used effectively at least twice. 13
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
