Abstract
Syncope is common, affecting approximately 1 million Americans every year. Although multiple pathophysiological mechanisms regarding its etiology have been documented, neurocardiogenic or vasovagal syncope is the most common cause of these episodes. Other less appreciated etiologies include various cardiac abnormalities in which a structural, electrical, or obstructive disturbance leads to a temporary reduction in blood flow to the brain, resulting in transient loss of consciousness. Cardiac malignancies, while rare, can present with syncope by either disrupting the cardiac conduction apparatus or simply obstructing blood flow through the cardiac chambers. Electrocardiograms and echocardiography are often very helpful in identifying these abnormalities. Here, we report a rare case of late-stage invasive squamous cell carcinoma of the lung presenting with recurrent syncopal events. The cancer invaded the cardiac right atrium causing various dysrhythmias, leading to a very rare cause of cardiogenic syncope. We also discuss how lung cancer can present in a subclinical manner and at times without obvious respiratory symptoms, dramatic physical examination abnormalities, and/or thoracic imaging abnormalities on chest radiograph.
Introduction
Syncope affects 1 million Americans every year, 1 although this figure may be on the rise due to the increase in the elderly population as well as comorbidities. Syncope is defined as a sudden, transient loss of consciousness associated with an inability to obtain postural tone, followed by spontaneous recovery. In the Framingham study conducted in 1985 to examine the prevalence, morbidity, and mortality of isolated syncope, 3% of men and 3.5% of women reported at least 1 syncopal episode during their lifetime, with the annual incidence of syncope of those 75 years or older at higher than 6%. The study authors concluded that in a completely isolated syncopal episode, that is, the absence of neurological, coronary, or other cardiovascular stigmata, syncope was not associated with increased morbidity or mortality. 2
As there is a wide array of etiological factors that can lead to syncope, patients often need to undergo an extensive workup to rule out any underlying, life-threatening illnesses. Recent data suggest that the most common cause of syncope is neurocardiogenic syncope, also known as vasovagal syncope. Neurocardiogenic syncope is a neurally mediated (or reflex) event that can be associated with carotid sinus hypersensitivity, vagus or glossopharyngeal nerve hyperactivity, defecation or micturition syncope, post-tussive syncope, deglutition syncope, malignancy-associated syncope, and many other conditions. Almost all of the previously reported cases of malignancy-associated syncope were caused by a neurocardiogenic mechanism, where the tumor had activated or hypersensitized the vagus nerve, thus resulting in bradycardia and hypotension and consequent syncope.
Based on our literature search, no prior case has ever been reported where the tumor-induced syncopal event was caused by a purely cardiogenic mechanism, in which the lung tumor invaded cardiac structures, thus impacting the myocardial conduction system resulting in various dysrhythmias leading to syncope. Here, we report a very rare case of non–small cell lung cancer (NSCLC) presenting initially as a purely cardiogenic syncopal event. The available clinical evidence indicates this was due to a direct tumor effect and invasion of the right atrium with subsequent disruption of the cardiac electrical conduction system.
Case Report
A 65-year-old Caucasian man with a history of tobacco dependence presented with a brief episode of dizziness while standing in line at the bank, which was immediately followed by witnessed loss of consciousness for 30 seconds. The dizziness was only “seconds-long” and immediately preceded “passing out.” He reported one previous episode of syncope last summer while “walking in the heat,” which also was sudden without any prodromal or other associated symptoms. His review of systems was negative for seizures, angina, orthostatic hypotension, stroke, or trauma. Over the past 3 months, he experienced an 18.1 kg weight loss with anorexia and constipation. The patient was a retired construction worker, had smoked 50 pack-years, and drank alcohol, but denied intravenous drug use.
On physical examination in the emergency department (ED), the patient was afebrile, with a presenting heart rate of 63 beats per minute (bpm), blood pressure range of 134/73, respiratory rate of 20 breaths per minute, and normal oxygen saturation of 98% on room air. There was no evidence of orthostatic hypotension based on blood pressure and heart rate measurements in the supine, sitting, and standing positions. The patient appeared thin and chronically ill with temporal-wasting, barrel chest, mild egophony of the right lung base, and a scaphoid abdomen. There was no evidence of dry mucus membranes, delayed capillary refill, heart murmur, jugular venous distension, lymphadenopathy, clubbing, cyanosis, edema, or neurological deficits. In addition, there was no evidence of gastrointestinal bleeding, scleral icterus, or jaundice.
Laboratory studies were notable for white blood cell count of 16.9 K/mm3, hematocrit of 28% with normal mean corpuscular volume (MCV), platelets of 650 K/mm3, albumin of 2.6 g/dL, normal glucose and corrected calcium, and negative cardiac enzymes. There were no other electrolyte or biochemical laboratory abnormalities with respect to liver and renal function. A chest radiograph showed a right peri-hilar mass-like dense opacity concerning for malignancy (Figure 1). Electrocardiogram (ECG) revealed bradycardia, variable junctional and ectopic atrial rhythms, ventricular bigeminy, QT prolongation, low voltage, T-wave abnormalities, and short PR interval (Figure 2). Additional rhythm strips in the ED showed ventricular bigeminy followed by a witnessed episode of worsening bradycardia (heart rate in the range of 30-35 bpm) with associated near syncope.
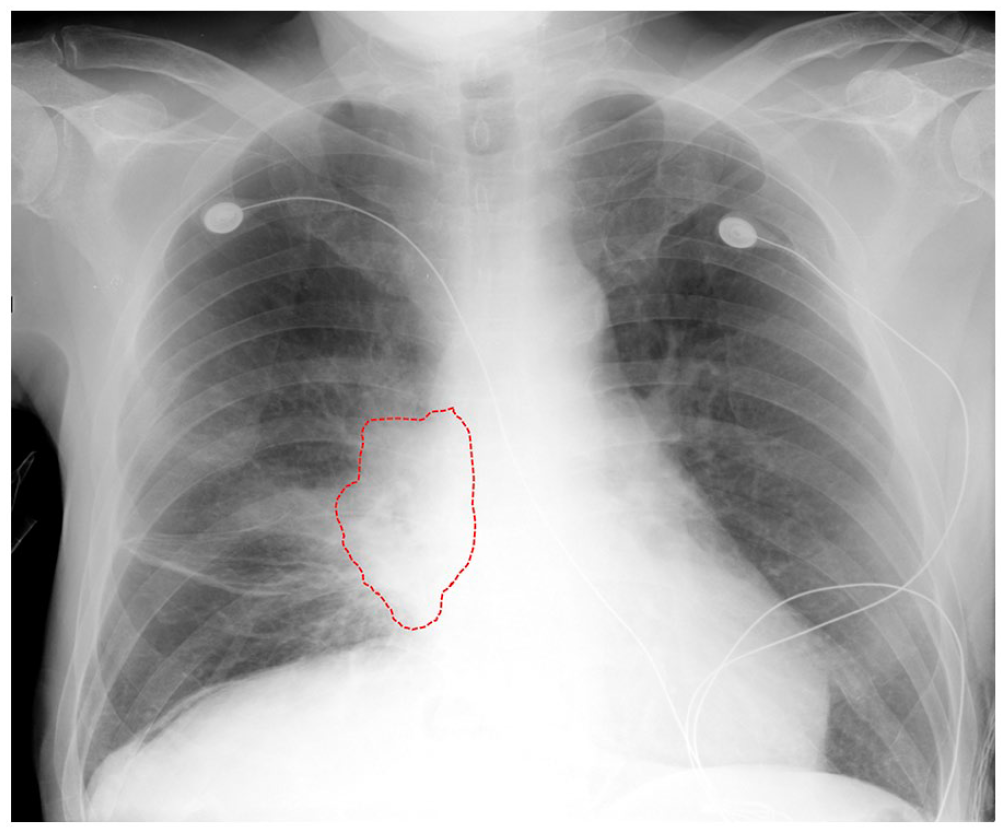
Admission chest radiograph showing a right peri-hilar dense opacity suspicious for tumor mass (outlined by the red dotted line) with surrounding subtle areas of lung atelectasis.

Admission electrocardiogram showing bradycardia, variable junctional and ectopic atrial rhythms, ventricular bigeminy, QT prolongation, low voltage, and short PR interval.
Chest computed tomography (CT) revealed a large cavitating necrotic mass in the right middle lobe contiguous with the mediastinum, invading the superior vena cava and right atrium with surrounding patchy consolidation (Figure 3). Echocardiogram showed an extrinsic pericardial mass at the right atrial roof (Figure 4A) with an atrial mass measuring 5.0 × 4.4 cm (Figure 4B). Bronchoscopy showed erythema and compression within the right bronchus intermedius and external compression of the right upper lobe bronchus (Figure 5). Endobronchial biopsy revealed invasive well-differentiated squamous cell carcinoma and a clinical picture consistent with stage IIIB disease (Figure 6).

Admission chest computed tomography (CT) showing a large cavitating necrotic mass in the right middle lobe contiguous with the mediastinum, invading the superior vena cava and right atrium with surrounding patchy lung consolidation. The tumor mass also encases the bronchus intermedius and anterior segment bronchus of the right upper lobe.

Echocardiogram showing (A) an extrinsic pericardial mass at the right atrial (RA) roof and (B) right atrial mass measuring 5.0 × 4.4 cm.

Bronchoscopy showing mucosal erythema and extrinsic compression in the right bronchus intermedius, with external compression of the right upper lobe bronchus.
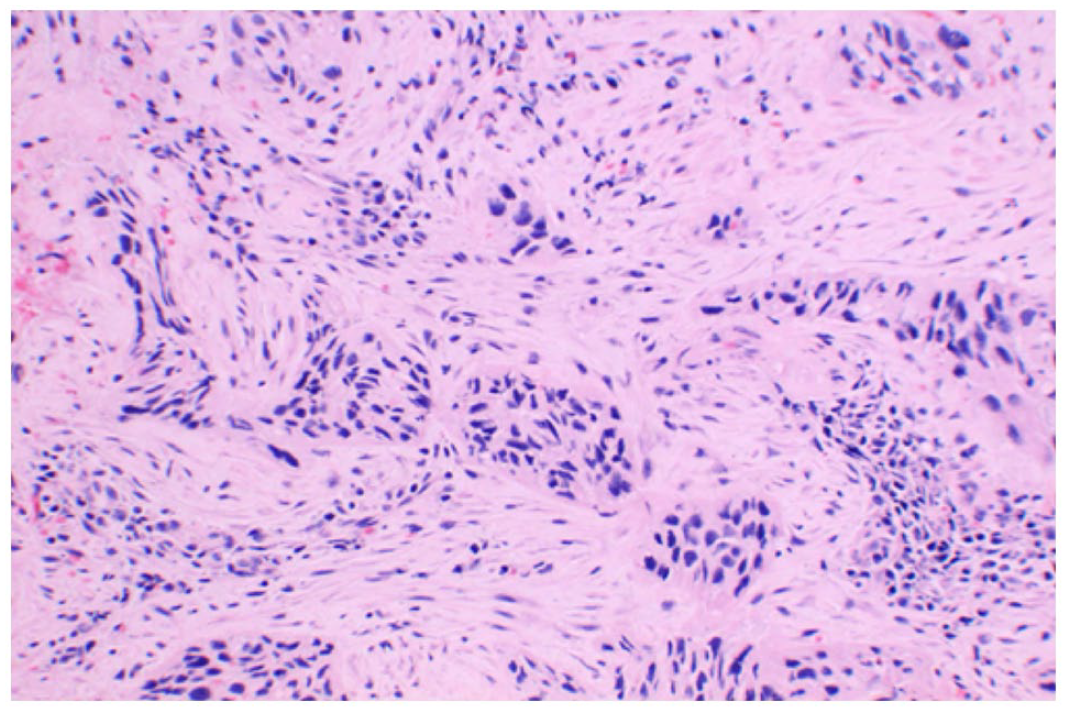
Endobronchial biopsy revealing invasive well-differentiated squamous cell carcinoma.
Unfortunately, the patient missed several chemotherapy appointments to treat his lung cancer. He did not maintain regular follow-up and we do not know whether he had recurrent episodes of syncope. On hospital readmission 6 months later, the patient developed atrial fibrillation and flutter (with frequent ventricular ectopy) and electrocardiographic evidence of right atrial enlargement (Figure 7A, 7B), a right pleural effusion, and hypoxemic respiratory failure. The patient eventually expired from progressive lung cancer opting for comfort care rather than continued medical treatment.
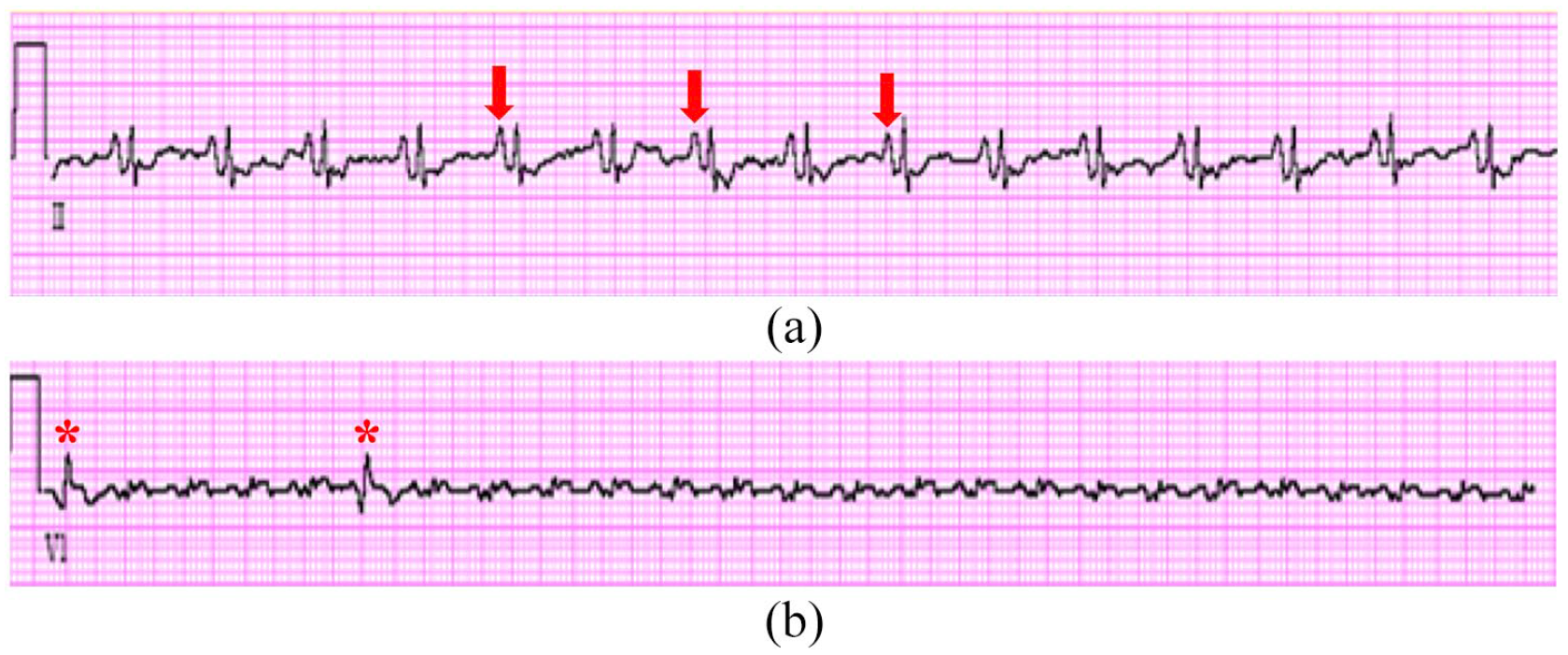
Electrocardiographic rhythm strips showing (A) right atrial enlargement (red arrows) and (B) atrial flutter with variable (2:1) block with 2 premature ventricular complexes (PVC) denoted by red asterisks. (Note: precordial lead with low voltages.)
Discussion
Drs Weiss and Baker were the first to describe the mechanism of malignancy-associated syncope. In 1933, they coined the term carotid sinus syncope syndrome (CSS) in association with head and neck malignancy. 3 In this phenomenon, tumor-induced stimuli (whether mechanical or chemical) activate and hypersensitize the carotid sinus and the glossopharyngeal nerve. This impulse then passes afferently to the dorsal nucleus of the vagus nerve which transmits stimuli to the heart, vasodepressor center, hypothalamus, and blood vessels, ultimately resulting in bradycardia and hypotension. Many cases of malignancy-induced neurocardiogenic syncope have been reported.
An example is a case report of neurally mediated syncope induced by lung carcinoma, where the cancer had invaded the left vagus nerve. 4 In another report, syncope was the presenting symptom of nasopharyngeal carcinoma, where the authors postulated that the tumor had likely involved the glossopharyngeal and vagus nerves, thus resulting in the syncopal events. 5 Neurocardiogenic syncope occurring as an intrathoracic tumor was reported in a case of NSCLC, which encased the left common carotid, left subclavian, and brachiocephalic arteries without compromising blood flow. The left common carotid artery was invaded at the level of branching from the aorta where the cardiac branch of the vagus nerve traversed. Subsequent to ruling out etiologies such as epilepsy, orthostatic hypotension, cerebrovascular disease, structural heart disease, and metabolic disturbances, the authors proposed that syncope in the patient might have been related to the direct activation of the vagus nerve by the tumor. After treatment with systemic chemotherapy, the patient did not have any other syncopal events. 6 A similar case was reported where the patient suffered from neurocardiogenic syncope as a result of bronchogenic carcinoma of the lung. After initiation of systemic chemotherapy, the patient reported no recurrent syncopal episodes. 7
Recurrent syncope can be associated with lung cancer and can present in unusual and cryptic ways including with normal chest radiograph and no respiratory symptoms. 8 Recurrent syncope without a clear cause should also alert the clinician to consider a subclinical presentation of lung cancer. In one case, chest radiography did not reveal any obvious mass until a subsequent chest CT prompted by abnormal labs showed a left mediastinal tumor compressing a branch of the vagal nerve leading to neutrally mediated syncope. 9 However, upon closer examination, the chest radiograph did reveal a different clue—that of an elevated left hemidiaphragm which was later connected to the effects of the left mediastinal mass.
Our case is a rare presentation of invasive squamous cell carcinoma of the lung that directly invaded the right hilar structures and right atrium with apparent effects on the cardiac conduction apparatus, resulting in various dysrhythmias and cardiogenic syncope in the absence of chest pain. Given the sudden onset of syncope without any antecedent or prolonged warning signs, the presence of structural heart pathology due to direct tumor invasion, the patient’s abnormal ECG disturbances with witnessed symptom correlation in the ED, and the absence of any other plausible alternative explanation, we believe that the most probable etiology for syncope in our patient was cardiac arrhythmia(s) leading to reduced cardiac output. Although disturbances in the cardiac rhythm have long been known as a common cause of syncope, it is the invasive lung cancer tumor mass leading to dysrhythmia in this case that makes it rather unique.
Invasion of the tumor into the heart is rare, but the most common ones are those arising from carcinoma of lung, breast, lymphoma, and malignant melanoma. Goldenberg and Ludwig described a case of a right atrial metastasis from a bronchogenic carcinoma with PR segment changes and atrial arrhythmias. 10 On autopsy, they found extensive infiltration of mediastinal structures with the tumor invading the right atrium and surrounding both the superior vena cava and right pulmonary artery. Microscopically, they found extensive infiltration and replacement of the sinoatrial node by moderately well-differentiated squamous cell carcinoma.
Unfortunately, the patient’s family declined our request for autopsy and it remains unknown to what extent the tumor had infiltrated his cardiac conduction system. However, based on the patient’s ECGs, echocardiogram, and thoracic imaging findings, as well as clinical correlates, we believe that his lung cancer infiltrated the atrial conduction system. Furthermore, given the observed patterns of arrhythmias and involvement of the right atrium in particular, the tumor effect also likely involved the sinoatrial node.
Conclusion
It is important to recognize these very rare causes of cardiogenic syncope to make the correct diagnosis and administer appropriate and timely therapy, especially as an oncologic work-up is not typically pursued with such a presentation. This case underpins the importance of performing appropriate and focused diagnostic imaging studies to link the lung mass with the patient’s cardiac abnormalities, and ultimately, clinical presentation of syncope, even if the pulmonary physical examination is not immediately revealing. As we have seen in some reported cases, lung cancer can present with mild or subtle abnormalities or can even appear normal on chest radiograph. Recognition of this rare pulmonary oncologic cause of syncope will alert the thoughtful clinician to consider lung cancer even in cases where it is not obvious.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethics review or approval for reporting individual case reports.
Informed Consent
Verbal informed consent was obtained from the patient regarding the use of his anonymized information to be used in a publication.
