Abstract
We present an adolescent male with a single intracardiac mass and pulmonary emboli, complicated by peripheral venous thrombosis and subsequent development of pulmonary pseudoaneurysms, leading to diagnosis of Hughes-Stovin syndrome. Remission was achieved with cyclophosphamide, corticosteroids, and pseudoaneurysm resection and maintained with infliximab and methotrexate.
Case Report
In May 2021, a 15-year-old male presented with 18 days of rigors, fatigue, night sweats, and 1 week of fever. Additional symptoms included abdominal pain, vomiting, and 18-pound weight loss over 1 month. Preceding his symptoms, he had mild COVID-19 presenting with fever, fatigue, and diarrhea in February 2021 and his first COVID-19 mRNA inoculation in April 2021.
Physical examination was notable for fever, with otherwise non-focal findings. Laboratories revealed leukocytosis (16.63 thousand/μL; normal: 4.5-13.5 thousand/μL), anemia (hemoglobin 11.1 g/dL; normal: 13-16 g/dL), hypoalbuminemia (3.3 g/dL; normal: 3.5-4.9 g/dL), and elevated C-reactive protein (CRP) (18.7 mg/dL; normal: <1.0 mg/dL), sedimentation rate (53 mm/h; normal: 0-15 mm/h), and ferritin level (375.23 ng/mL; normal: 11.1-171.9 ng/mL).
Inflammatory bowel disease was suspected. A magnetic resonance (MR) enterography did not demonstrate gastrointestinal tract abnormalities; it did show, however, 3 basilar lung nodules, with restricted diffusion. A chest computed tomography (CT) with contrast redemonstrated these nodules and revealed a nodular right ventricular (RV) mass.
Bacterial endocarditis, benign/malignant cardiac tumors, intracardiac thrombosis, and cardiac pseudotumor were considered as potential causes for intracardiac mass with pulmonary emboli. Several diagnostic procedures were conducted in the following months to identify the cause of his symptoms.
First admission (May-June 2021): Ceftriaxone and vancomycin were empirically initiated for presumptive culture-negative endocarditis. Staphylococcus capitis and Cutibacterium acnes were identified from a single blood culture; other microbiologic studies were unrevealing (Table 1). Pursuing a tissue diagnosis, a right-sided pulmonary nodule biopsy was performed by interventional radiology using ultrasound and computed tomography guidance; intravenous (IV) dexamethasone 8 mg was administered intraoperatively. Nodal tissue pathological evaluation showed diffuse parenchymal necrosis. Post-surgically, fevers transiently improved. Subsequently, a left cephalic vein thrombosis was identified. Thrombophilia workup was negative (including analysis of protein C and S activity, antithrombin activity, factor V Leiden mutation, prothrombin gene mutation, factor VIII activity, lupus anticoagulant, anticardiolipin, and anti-beta-2 glycoprotein antibodies).
Additional Pertinent Laboratory Studies.
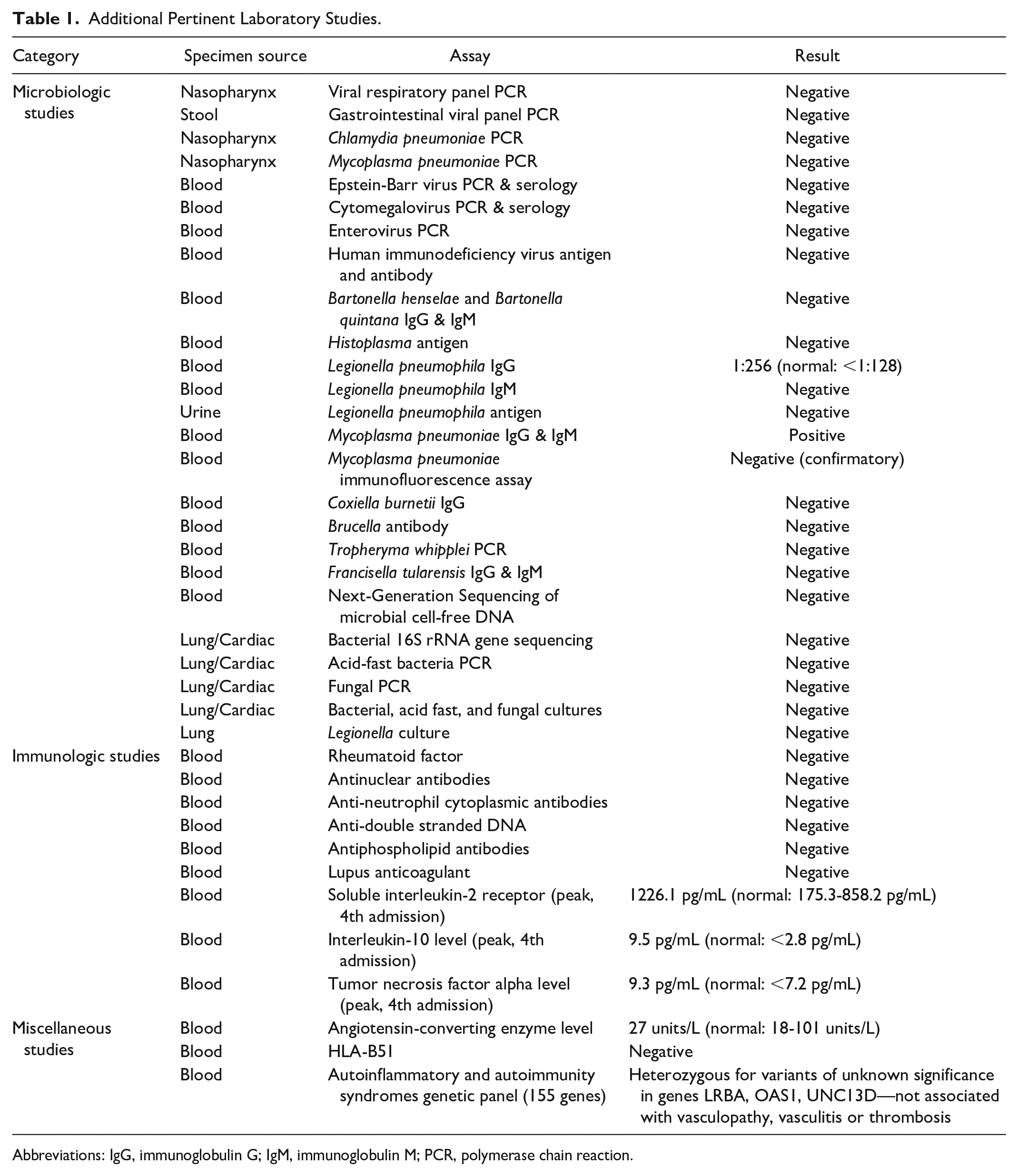
Abbreviations: IgG, immunoglobulin G; IgM, immunoglobulin M; PCR, polymerase chain reaction.
RV mass removal was surgically attempted via median sternotomy. The right atrium was opened with an atriotomy parallel to the atrioventricular groove, and examination of the tricuspid valve demonstrated a large abscess cavity involving the subvalvar apparatus. This cavity was filled with yellow-white exudative material adherent to the myocardium, and semi-solid gelatinous material, which were removed using forceps. IV methylprednisolone 1000 mg was administered intraoperatively per anesthesia protocol. Tissue samples were insufficient for pathological evaluation; those cultured yielded no growth. He defervesced post-procedure and was discharged on IV ceftriaxone plus vancomycin, and rivaroxaban.
Second admission (June–July 2021): Two weeks post-discharge, fever recurrence prompted readmission. A pericardial effusion was noted on echocardiogram, suggesting post-pericardiotomy syndrome; colchicine and naproxen were started. Right subclavian and axillary vein thromboses were also identified; anticoagulation was changed to enoxaparin. Fever initially improved. Additional studies were without findings (Table 1). Chest CT angiography and cardiac MR showed new pulmonary emboli and a stable RV mass. The patient was discharged on colchicine, enoxaparin, ceftriaxone, and vancomycin.
Third admission (July–August 2021): Fever persisted prompting readmission 3 weeks later. Through a re-do sternotomy, the cardiac mass was surgically biopsied. During the procedure, 2 right atrium empty-appearing sacs were removed, and 11 further specimens were removed from the RV including normal and abnormal myocardium. IV methylprednisolone 1000 mg was administered intraoperatively per anesthesia protocol. He defervesced post-procedure. Pathological examination of the cardiac tissue revealed fibrinopurulent exudate, granulation tissue (Figure 1), and rare gram positive coccoidal forms worrisome for infectious endocarditis. Additional microbiologic studies were negative (Table 1). Daptomycin, levofloxacin, and penicillin were initiated, and the patient was discharged.

Cardiac mass histology: Hematoxylin-eosin stains of endomyocardial biopsy showing fibrinopurulent exudate on the endocardial surface (left). The photograph on the right shows a higher magnification of the exudate, including fibrin and inflammatory cells with necrotic debris.
Fourth admission (August–September 2021): Fever recurred, leading to readmission 2 weeks later. Echocardiogram showed a new small cystic RV mass, with otherwise stable findings. Chest CT angiography showed 2 large right pulmonary artery pseudoaneurysms (PAPs) and new pulmonary emboli (Figure 2A, C). Brain/neck CT angiography were unremarkable. An ophthalmologic examination was normal. No association was established between his illness and clinical COVID-19 or receipt of first COVID-19 vaccine.
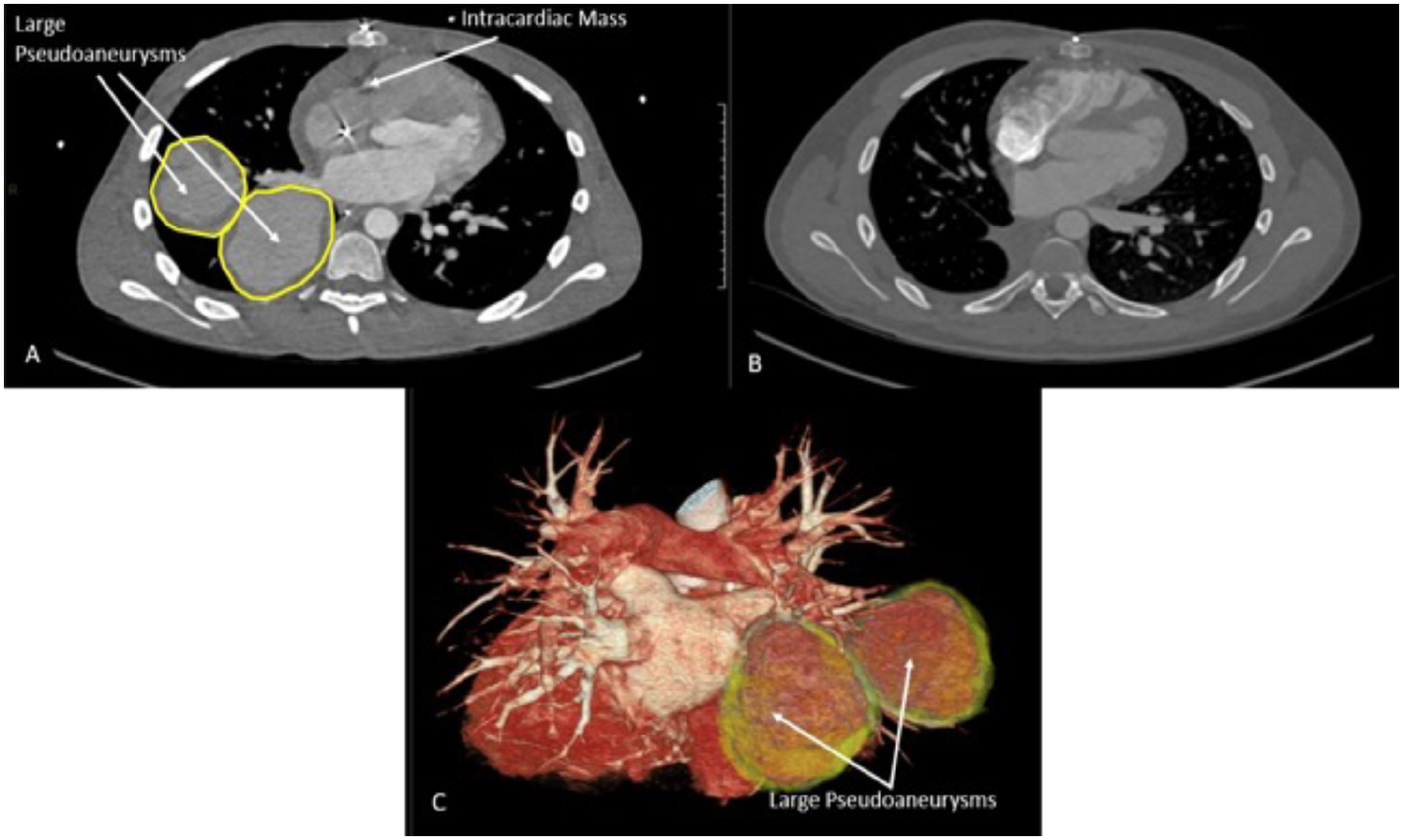
Chest computed tomography angiography pre- (A) and post-treatment (B). (C) Three-dimensional reconstruction of 2 right pulmonary artery pseudoaneurysms.
After excluding malignancy and receipt of adequate courses of broad-spectrum antimicrobials for fitting infectious etiologies, his RV mass was deemed to be thrombotic. Intracardiac thrombosis, recurrent pulmonary and peripheral thrombosis, and PAPs led to the diagnosis of Hughes-Stovin syndrome (HSS), following multidisciplinary discussion with international experts. IV high-dose methylprednisolone was initiated and later transitioned to prednisone, with sustained fever resolution and CRP improvement. Considering the size of the pseudoaneurysms and high likelihood of rupture leading to catastrophic pulmonary hemorrhage, the patient underwent pseudoaneurysmal plugging via right heart catheterization, immediately followed by right lower lobectomy with pseudoaneurysm resection via right posterolateral thoracotomy at the fifth intercostal space. Post-surgically, heparin anticoagulation was started, followed by apixaban. Pulmonary artery histology was supportive of HSS (Figure 3).

Pulmonary artery histology: Hematoxylin-eosin stains of the pulmonary artery wall showing a lymphocytic inflammatory infiltrate in the arterial wall, marked dilatation of the lumen, intraluminal thrombosis, multifocal intimal fibrinoid necrosis, and elastic layer fragmentation (left). The photograph on the right shows a higher magnification of the lymphocytic inflammatory infiltrate in the pulmonary arterial wall and elastic fragmentation.
After 3 weeks of post-surgical wound healing, cyclophosphamide 500 mg IV every 2 weeks (6 doses) was started. Four weeks post-cyclophosphamide completion, infliximab 500 mg IV every 4 weeks and subcutaneous methotrexate 15 mg weekly were initiated.
Two weeks after cyclophosphamide initiation, an echocardiogram showed minimal evidence of RV mass, and chest CT angiography revealed no PAPs or emboli (Figure 2B). Eleven months post-diagnosis, the patient is clinically well and off corticosteroids. He continues to receive infliximab, methotrexate, and apixaban.
Discussion
We present an adolescent male with constitutional symptoms and systemic inflammation, in the setting of intracardiac, pulmonary, and venous thromboses, with later development of PAPs. His clinical course was atypical after receipt of a prolonged duration of broad-spectrum antimicrobials for culture-negative endocarditis. An inflammatory etiology was suspected due to recurrent thrombosis despite anticoagulation and symptomatic improvement following high-dose intraprocedural corticosteroids. The unusual combination of widespread thrombosis and pulmonary artery aneurysms (PAAs) or PAPs is unique to two vasculitides: Behcet’s disease (BD) and HSS.1-3 Although some classify HSS under the BD spectrum, these are still considered as separate entities.1,3 BD is a variable vessel vasculitis, infrequent in children (<5% of cases), and unlikely in our patient due to lack of mucocutaneous and other characteristic features.2,4,5 Our patient’s presentation was most consistent with HSS and further supported by classic pulmonary artery histology findings. 1 HSS is exceedingly rare (<60 cases previously reported), most common in young males, and characterized by extensive thrombosis and aneurysm formation, with predilection for the pulmonary circulation.1,3 Its etiology is unknown, and there are no formal diagnostic criteria or confirmatory serologies; thus, the diagnosis relies on identifying the typical features, including widespread vaso-occlusive disease (with normal coagulation profiles) and visualization of PAAs/PAPs on CT angiography. 3
In HSS, vaso-occlusive disease may manifest as deep venous, cerebral venous, intracardiac or arterial thrombosis, or recurrent superficial thrombophlebitis, and the presenting symptoms vary accordingly.1,3 HSS typically evolves in 3 stages, starting with peripheral thrombophlebitis, followed by formation of large pulmonary and/or bronchial aneurysms, ultimately leading to massive hemoptysis and death from aneurysmal rupture (21.2% fatality rate).1,3 Our patient followed a similar course; fortunately, adequate treatment prevented progression to fatal hemoptysis.
The treatment of HSS mirrors BD management. Immunosuppression with corticosteroids and cytotoxic agents is used to induce remission. Aggressive immunosuppression with cyclophosphamide and high-dose corticosteroids was pursued in our patient, as recommended for PAAs/PAPs in BD and HSS.3,6 Anti-tumor necrosis factor alpha inhibitors (TNFi) have been used for refractory BD and in 4 HSS cases.3,6 We favored the use of TNFi (infliximab) plus methotrexate over cyclophosphamide for remission maintenance due to a safer side effect profile.
Unlike most previously reported people with HSS who had bilateral PAAs/PAPs, 3 our patient’s unusual presentation with unilateral PAPs allowed for a combined medical-surgical approach. Surgical options are considered for PAAs/PAPs to prevent massive hemorrhage, with embolization being preferred over open resection. 6 Proposed indications for surgical intervention in HSS include PAA/PAP size >30 mm, rapid PAA/PAP growth, or leaking unstable PAAs/PAPs 3 ; our patient met 2 of these indications. According to a recently developed HSS radiographic atlas, he had stage 5 pulmonary vasculitis, which portrays the highest mortality risk from pseudoaneurysmal rupture. 7 The surgical approach to this problem can be treacherous. Therefore, the decision for surgical care centered on preoperative vessel embolization followed immediately by thoracotomy and right lower lobectomy/pseudoaneurysmal resection. Our successful experience could serve as a reference for immunosuppressive and surgical management of future patients with this rare condition.
Conclusions
Our case calls for consideration of HSS and other inflammatory etiologies in the differential diagnosis of an intracardiac mass. In addition, it underscores the diagnostic and management challenges of HSS, a complex and likely underrecognized entity. Last, it highlights the importance of multispecialty collaboration to develop a combined medical and surgical approach for people with HSS and similar conditions.
Footnotes
Acknowledgements
We sincerely thank Dr Nicole Hames for reviewing the preliminary case summary.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Dr Prahalad is supported, in part, by the Marcus Foundation Atlanta, Inc.
Ethics approval
The Institutional Review Board approved the preparation of this article by granting a Non-Human Subject determination to this study.
Informed consent
Written informed consent was obtained from a legally authorized representative(s) for anonymized patient information to be published in this article.
