Abstract
Three young males with Hugh-Stovin’s syndrome presented with cough, haemoptysis, fever, raised inflammatory markers, and pulmonary artery aneurysm. Only one had recurrent oral ulcers suggestive of Behcet’s disease, and none were HLA B51 positive. All responded well to immunosuppression but eventually needed either an endovascular procedure or surgery.
Keywords
Introduction
Hughes-Stovin syndrome (HSS) is a rare primary variable vessel vasculitis of unknown cause. It is characterized by segmental pulmonary artery aneurysms (PAAs), and venous thrombosis (superficial and deep). 1 Hughes-Stovin syndrome shares clinical and pathological similarities with Behcet’s Disease (BD) and consequently is regarded by some authors as incomplete BD. 2 With only 90 cases described in PubMed, 3 no specific diagnostic and management guidelines are available in the literature.
Rupture of a PAA resulting in massive hemoptysis is a dreaded complication, and the overall outcome is very poor in the absence of definitive treatment guidelines. Treatment, which may include immunosuppression, anticoagulation, and surgical intervention, must all be judiciously used. Here we present our experience with 3 cases of HSS with a specific focus on different management strategies.
Case 1
In August 2015, a 24-year-old man presented with persistent high-grade fever, cough, and breathlessness with streaks of hemoptysis all of 2 months duration. Physical examination was normal, except for minimal basal lung crepitations. Complete blood count was normal, and erythrocyte sedimentation rate (ESR) was 55 mm/hr. Computed tomography pulmonary angiogram (CT-PA) was suggestive of bilateral pulmonary embolism with no evidence of deep vein thrombosis (DVT) on the lower limb doppler. He was treated with anticoagulation, and an inferior vena cava (IVC) filter was inserted. There was a transient improvement, but over the next few weeks, there was a recurrence of high-grade fever, cough, hemoptysis, left-sided chest pain, right flank pain, and lower limb claudication. Investigations revealed an IVC filter clot extending up to the right renal vein, which was managed with heparin and anticoagulants.
Since the symptoms were recurrent, he was referred to our centre for further management. Detailed history revealed that he had 2 episodes of oral ulcers recently. Clinically he appeared pale. Laboratory reports were: Hb: 7.3 gm %, WBC: 6000/mm3, platelets: 6.44 lakh/mm3, ESR: 139 mm/hr, and C-reactive protein (CRP) 192 IU/L. Renal and liver function tests were normal. Blood culture and sputum culture were sterile; urine culture grew Escherichia coli; echocardiogram was normal, and serum procalcitonin was negative. Procoagulant factors and serological tests were negative. Computed tomography pulmonary angiogram showed a large pseudoaneurysm arising from the segmental branch of descending interlobar pulmonary artery, almost completely occupying the whole of the left lower lobe, measuring 9.3 × 8.1 × 10 cm with associated thrombus adherent to the superior and inferior wall of the pseudoaneurysm (Figure 1A). There was adjacent collapse consolidation of the lung suggestive of hemothorax. Based on the consistent clinical features, pulmonary artery aneurysm, and DVT, HSS was diagnosed and pulsed with intravenous steroids. Given the rapid fall in haemoglobin, hemothorax, and large pseudoaneurysm, feeding artery endovascular coil embolization was done after the steroid pulse (Figure 1C and D). Subsequent treatment included oral steroids, intravenous cyclophosphamide, and oral anticoagulation. The haemoglobin stabilized, and there was a gradual improvement in systemic and respiratory symptoms along with laboratory parameters.

All images are of case 1 (A) computed tomography of the pulmonary artery (CTPA) showing a large pseudoaneurysm arising from the segmental branch of descending interlobar pulmonary artery, almost completely occupying the whole of the left lower lobe, measuring 9.3 × 8.1 × 10 cm. It is associated with hyperintense nonenhancing thrombosis adherent to superior and inferior walls of the pseudoaneurysm. There was adjacent collapse consolidation of the lung suggestive of hemothorax. (B) CT aortogram showing saccular aneurysm of descending abdominal aorta just above the origin of renal arteries 1. (C) DSA of pulmonary vessels showing aneurysm of the pulmonary artery. (D) DSA showing coils inside the aneurysm used for embolization.1. (E) Chest X-ray showing post-left lobectomy status of the patient.
Three months later, he developed painless diminution of vision secondary to bilateral infiltrative optic neuropathy with vitritis, which was treated successfully with intravenous methylprednisolone, tapering oral prednisolone and mycophenolate mofetil. Over the next 6 years, he had an intermittent relapsing course of optic neuropathy requiring tapering doses of prednisolone without any recurrence of hemoptysis or systemic features. Repeat CT angiography showed a saccular abdominal aneurysm without any bleeding complication (Figure 1C).
In 2022, he again developed intractable hemoptysis. Repeat CTPA showed left lower lobe cavity with clot. Since the hemoptysis was unabated, a left lower lobectomy was performed (Figure 1E). The patient eventually improved and was discharged in a stable state. He is doing well at 6 months of follow-up.
Case 2
A 24-year-old man complained of recurrent cough, hemoptysis, dyspnea on exertion, and a few episodes of oral ulcers. Chest X-ray done elsewhere showed bilaterally enlarged hilum, and 2D echo showed pulmonary artery hypertension (PAH) (70 mm hg). Computed tomography chest showed bilateral PAAs with thickening of the vessel wall and internal thrombus. But the patient did not have any other systemic feature and was managed with anticoagulants (rivaroxaban) and started on ambrisentan and tadalafil for PAH.
Two years later, he consulted us. Physical examination revealed a loud second heart sound. Laboratory tests showed: Haemoglobin 16 gm %, Total WBC count: 5150/mm3, platelet: 1.71 lakh/mm3, ESR: 50 mm/hr, CRP: normal, total protein: 7.5 mg/dL with albumin of 4.7 mg/dL, and creatinine: 0.8 mg %. Viral markers were negative. Chest radiograph showed bilateral diffuse patchy opacities. Computed tomography of the pulmonary artery showed aneurysmal dilatation and peripheral wall thickening of the distal main pulmonary artery, left and right pulmonary artery, and bilateral descending pulmonary arteries (Figure 2A to C). There were peripheral hypodensities with filling defects in distal bilateral descending pulmonary arteries, suggesting long-term thrombi-embolism. Also, distal segmental arteries bilaterally showed irregularity with tortuosity with an occasional aneurysm. Positron emission tomography (PET) CT scan showed no uptake in the vessel wall but an internal thrombus adherent to the wall of the descending pulmonary arteries. Venous doppler of both lower limbs was normal. A 2D echo showed dilated right heart, main pulmonary artery and its branches with severe PAH. Procoagulants factors, other autoimmune serology, and HLA B51 were all negative (Table 1). Hughes-Stovin Syndrome was diagnosed, and the patient was treated with prednisolone, mycophenolate mofetil, tadalafil, ambrisentan, and anticoagulation.
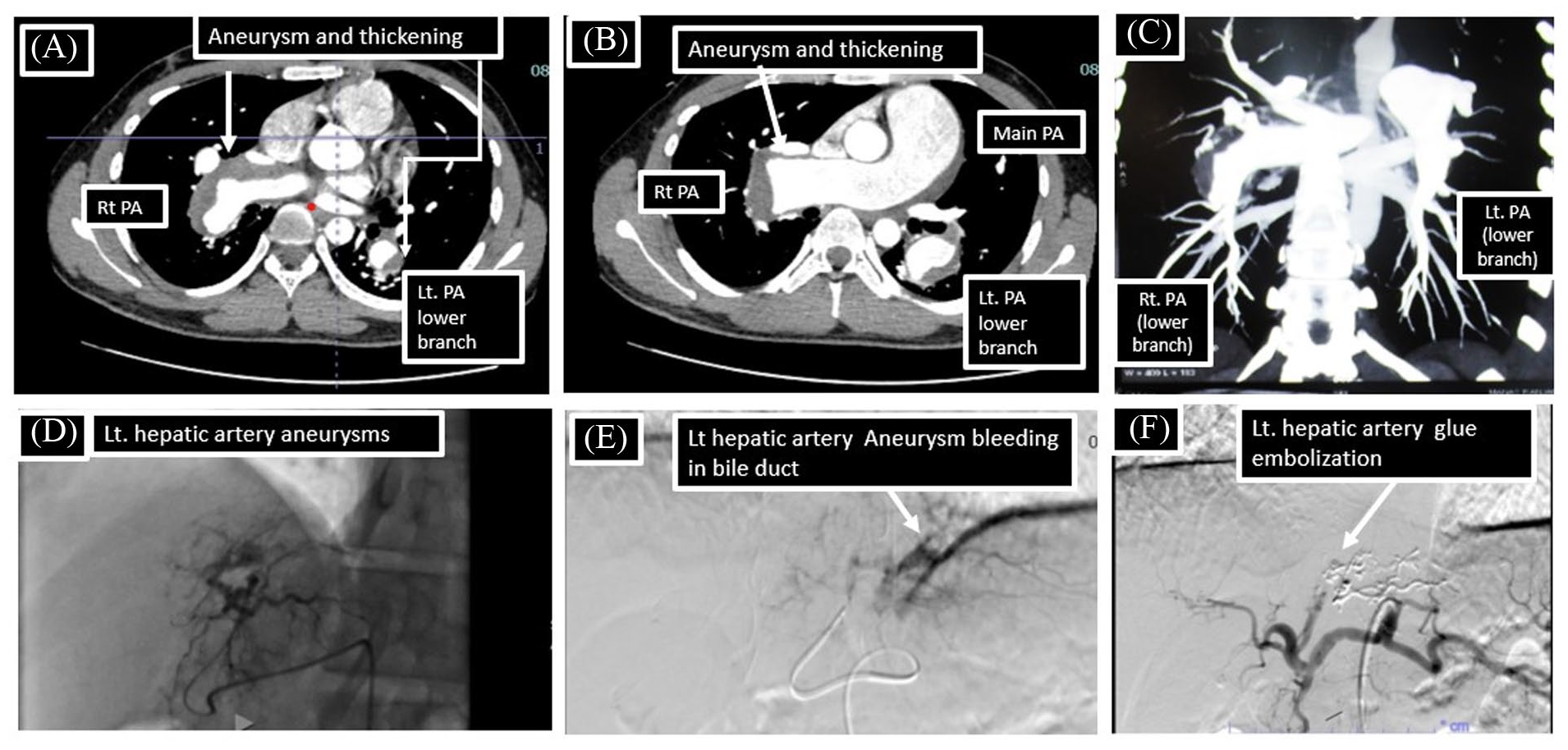
All images are of case 2. (A) CTPA showing aneurysmal dilatation and peripheral wall thickening of the right pulmonary artery and lower branch of the left pulmonary artery. 2. (B) CTPA showing aneurysmal dilatation and peripheral wall thickening of the distal main pulmonary artery and right pulmonary artery 2. (C) CTPA showing aneurysmal dilatation of bilateral lower branches of the pulmonary artery 2. (D) DSA of hepatic vessels showing aneurysm of the left hepatic artery 1. (E) DSA of hepatic vessels showing communication between the ruptures left hepatic artery aneurysm in the common bile duct 1. (F) DSA-hepatic vessels showing diminished left hepatic artery flow secondary to glue embolization.
Showing clinical features, imaging findings, and treatment of all 3 patients with Hughes-Stovin syndrome.

Abbreviations: ANA, antinuclear antibody; ANCA, anti-neutrophilic cytoplasmic antibody; CRP, C reactive protein; CT, computed tomography; ESR, erythrocyte sedimentation rate; HLA B51, human leucocyte antigen B51; MMF, mycophenolate mofetil; PAH, pulmonary artery hypertension.
Procoagulant factors -homocysteine, protein C, protein S, factor V Leiden mutation, antithrombin III, antiphospholipid antibodies.
A year later, he developed left eye mild optic neuritis, which improved spontaneously. For the next 4 years, he did well on treatment. In 2022, he was admitted with malena, exertional dyspnea, fatigue, and severe anaemia (Hb:3 gm/dL). Upper gastrointestinal (GI) endoscopy showed prepyloric telangiectasia, gastric ulcers, and hiatus hernia. It was managed conservatively, but the malena recurred within a few months. Computed tomography angiogram of the abdomen and chest showed an enhancing outpouching from segment II left hepatic artery (10 × 8.8 mm), suggestive of pseudoaneurysm with long-term pulmonary thrombosis and severe PAH (PASP: 70 mm hg). After a blood transfusion, DSA (Figure 2D) showed left hepatic artery pseudoaneurysm with arterio-portal fistula leading to haemobilia. Bleeding was controlled with glue embolization (Figure 3E and F). After the procedure, the patient improved and was discharged on prednisolone, mycophenolate, tadalafil, and ambrisentan.

All images are of case 3: (A) Computed tomography of the pulmonary artery (CTPA) showing 2 large aneurysms of the lower branch of the left pulmonary artery with internal thrombus 2. (B) CTPA showing aneurysmal dilatation with internal thrombus in the lower branch of the right pulmonary artery 2. (C) Chest X-Ray posterior anterior (CXR PA) view showing aneurysmal dilatation of right pulmonary artery 2. (D) DSA of the right pulmonary artery with large aneurysm 1. (E) DSA of the right pulmonary artery with partial diminution of the aneurysm with embolic plug 2. (F) DSA showing of the right pulmonary artery with complete regression of aneurysm secondary to embolic plugs 1. (E) CXR-PA view of the patient at follow-up showing complete regression of pulmonary aneurysm secondary to embolic plugs.
Case 3
A 32-year-old man presented to us with cough, hemoptysis, and intermittent fever. The patient was vitally stable, and the clinical exam was unremarkable. He gave a history of streaks of intermittent hemoptysis in the past 6 months, but at a presentation, he had an episode of hemoptysis (300 mL) for which he was admitted and further evaluated. Routine blood tests showed haemoglobin was 9.1 gm %, normal WBC and platelet count, ESR: 78 mm/hr. and CRP: 43 IU/L. Viral markers, serum homocysteine, anti-nuclear antibody (ANA), anti-neutrophilic cytoplasmic antibody(ANCA), antiphospholipid antibodies, and HLA B 51 were all negative. Chest X-ray showed a few fibrotic shadows in the right upper zone and a large nodular shadow in the right infra-hilar region in both the lower lobes. Computed tomography chest with pulmonary angiography showed 2 large aneurysms in the inferior branch of the right pulmonary artery with partial thrombosis (Figure 3A to C). Venous Doppler of the lower limb and 2D echo were normal. Pulmonary artery embolization with vascular plugs was done as shown in Figure 3D and E. Patient was treated with oral prednisolone and oral cyclophosphamide, which was later switched to azathioprine. He responded well to treatment with the resolution of symptoms. At 5 years follow-up, there is no recurrence of symptoms, and a repeat X-ray shows resolution of previous aneurysms as shown in Figure 3F.
Discussion
All 3 patients were young men, consistent with literature being more common in males. 4 Two patients had a history of fever with raised inflammatory markers at presentation. All had a history of the streak of hemoptysis before 2 of them had major hemoptysis requiring emergency endovascular intervention. Oral ulcers were seen in 2 patients, while none had genital ulcers. Two patients developed optic neuropathies during the disease course. All patients had normal procoagulant factors, and autoimmune serology was negative with normal viral markers. None were HLA B51 positive. Two patients had bilateral PAAs, while 1 had unilateral PA aneurysms.
Hughes-Stovin syndrome is a rare inflammatory vasculitis similar to BD. Even though oro-genital ulcers and uveitis are uncommon in HSS, some consider it an incomplete form of BD due to the lack of other features of complete BD. 5 These individual PAAs may be recognized as complete PAAs or pulmonary artery pseudoaneurysms (PAP). 3 Pseudoaneurysms are histologically characterized by the involvement of only the external layers of the arterial wall (the media and adventitia) and are at greater risk of ruptures and bleeding. 6 Inflammation of the vessel wall, angiodysplasia of the bronchial vessel and thrombus inflammation have been implicated as causes of aneurysm. 3 Thrombosis is often seen in these aneurysms, and this can lead to embolism and pulmonary hypertension.
As proposed by HSS International Study Group (HSSISG) 7 and also summarized by Sanduleanu and Jansen 3 and Kechida et al, 8 the radiological features can be described as (1) early vasculitis—characterized by aneurysmal wall thickening and dilatation as seen in our case 2. (2) Stable aneurysmal disease—seen as a contrast-filled dilation either as complete aneurysm or pseudoaneurysm in the pulmonary, lobar, segmental, or bronchial artery as seen in our case 3 or (3) unstable aneurysmal disease—seen as signs of extra-luminal leakages of the aneurysm in the form of peri-aneurysmal ground-glass opacities or consolidation with or without loss of aneurysmal wall definition as seen in our case 1. All 3 of our cases had intra-luminal ‘in-situ’ thrombus adherent to the aneurysmal wall, a finding similarly noted by the HSSISG group in 77% of cases. 7 In our patients, pulmonary artery involvement was bilateral in 2 patients while it was unilateral in 1 case. Thus, our cases document the varied involvement of the pulmonary artery and differing imaging patterns seen in HSS.
Management of HSS is complicated because an aneurysm has a bleeding risk, and there is concomitant thrombosis with a risk of thromboembolism that may need an anticoagulant. Furthermore, HSS may be complicated by massive hemoptysis secondary to ruptured aneurysms and a high mortality rate, often needing various emergency endovascular or surgical procedures. Our first case had a bleeding aneurysm leading to a sudden drop in haemoglobin and needed coil embolization with immunosuppression to control hemoptysis, followed by anticoagulation. Later, on recurrence of massive hemoptysis, a left lobectomy was needed. In our third patient, embolic plugs were used to treat the aneurysm. Our second patient with a stable disease with an aneurysm dilatation and thickening had low bleeding risk and was thus started on immunosuppression and anticoagulation upfront. We propose a treatment strategy as given in Figure 4. All patients of HSS should be started on immunosuppression from the start, and risk stratification concerning bleeding risk should be done before the start of anticoagulation. There is currently limited literature on the effectiveness of anti-TNF in HSS, with only 2 reported cases, 1 of which demonstrated efficacy.9,10 However, due to the shared pathological mechanisms between HSS and Behcet’s in which TNF has been proven effective, 11 it may be worthwhile to consider using anti-TNF therapy in cases that are recurrent or resistant to other treatments.
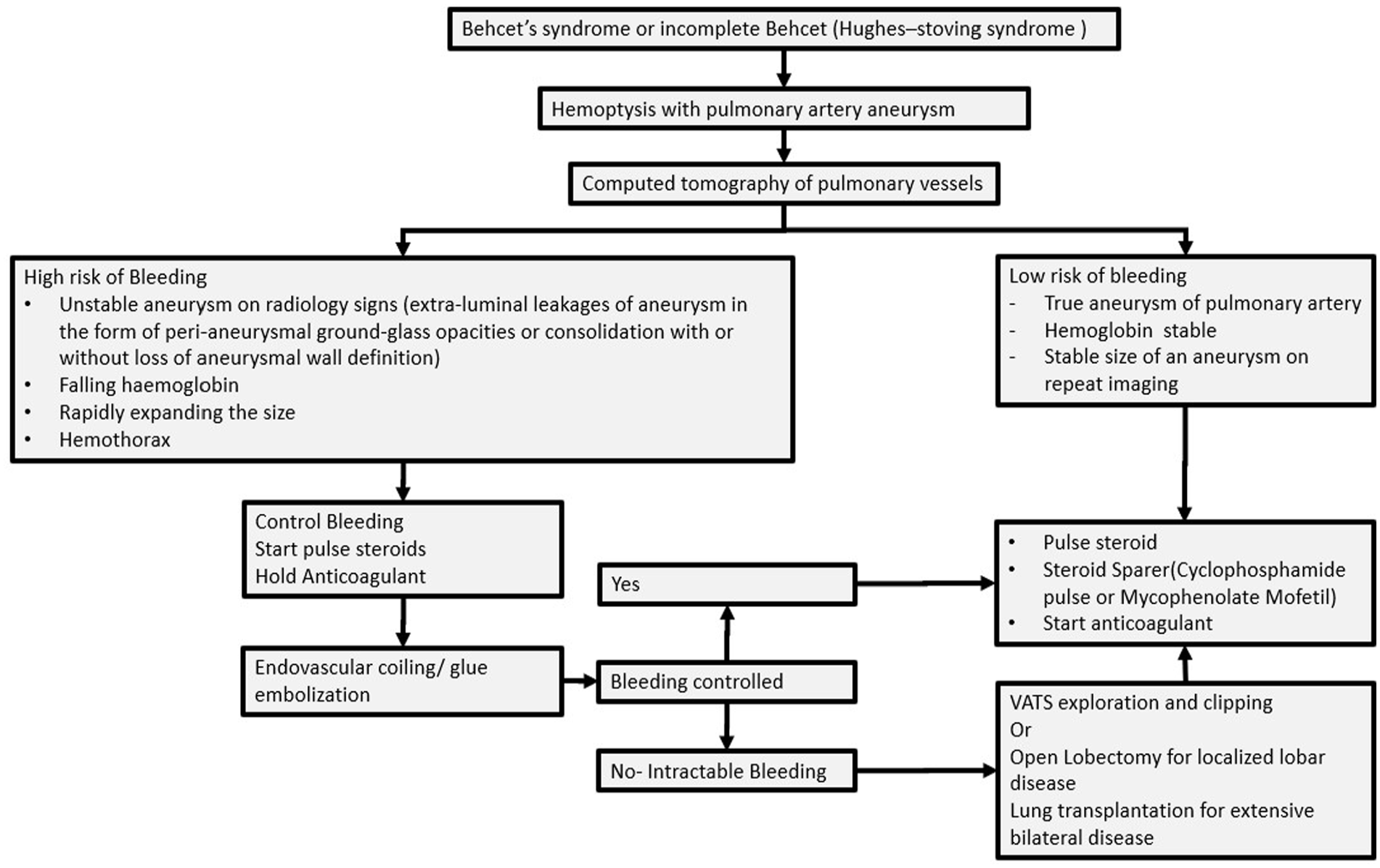
Flow chart showing proposed management of Hughes-Stovin syndrome.
Patient 1 had a concomitant saccular aneurysm in the abdominal aorta (Figure 1C). Only one previously reported case documents the concurrent occurrence of the abdominal aorta in HSS. 12 Patient 2 had concomitant bleeding of the hepatic artery aneurysm communicating with the bile duct. It was successfully treated with glue embolization. Involvement of left hepatic artery aneurysm in HSS patients is rare and reported only by Herb et al. 13 Behcet's disease is rarely associated with hepatic artery aneurysm.14-16 Also, extrapulmonary arterial involvement in HSS has been previously reported in the splenic artery, 17 internal iliac artery aneurysm,18,19 and femoral arteries branches. 12 Optic neuritis, 20 as seen, 2 of our patients, and pan-uveitis, 21 have been previously reported in HSS. Overlapping features between HSS and BD have been previously reported in the literature. 12 Involvement of extrapulmonary vessels, the patchy occurrence of oro-genital ulcers and eye involvement during the natural history of HSS may indicate that it may be a form of evolving BD. However, as the mortality of HSS is high due to massive hemoptysis, and hence all cases may not have the chance to evolve into complete BD.
The literature on long-term follow-up of HSS is limited due to its rarity and fatal outcomes from aneurysm rupture. The first patient experienced a relapsing course over 7 years, ultimately requiring a left lower lobe lobectomy. Similarly, the second patient experienced a recurrence of aneurysm rupture in the hepatic artery after a 4-year interval of remission. The third patient who was followed for 5 years had a monophasic course. It is likely that HSS exhibits a relapsing course in the same vascular territory or an aneurysm in a different vascular territory. These findings emphasize the importance of long-term monitoring and early intervention in patients with HSS to prevent catastrophic events and improve patient outcomes.
In conclusion, our 3 patients with HSS add to the described clinical characteristics and varied radiological findings of HSS. Prevalence of DVT and HLA 51 positivity were uncommon. Our cases also illustrate several endovascular techniques that could prevent mortality in HSS patients with life-threatening hemoptysis secondary to pulmonary aneurysms. Anticoagulant use is still controversial, but we propose a risk-guided approach that needs large-scale study. The concomitant involvement of other systemic vessels affirms its pathological similarity to BD. All our patients responded to immunosuppression, anticoagulant, and endovascular procedures and did well on prolonged follow-ups.
Footnotes
Acknowledgements
None
