Abstract
Tricuspid atresia is a congenital malformation of the tricuspid valve resulting in a lack of blood flow between the right atrium and the right ventricle. Management generally involves staged surgical intervention enabling affected individuals to survive into adulthood. Although surgical intervention greatly improves morbidity and mortality in this patient population, there are many long-term complications associated with the creation of a surgical shunt. We report a case of a 33-year-old male with tricuspid atresia who underwent Fontan surgery as a child and presented to our hospital with acute liver failure.
Keywords
Introduction
Tricuspid atresia (TA) is a congenital anomaly resulting in aberrant connection between the right atrium and the right ventricle. Less than 10% of affected individuals survive past the age of 1 without surgical intervention. 1 In 1969, Fontan performed the first successful right-heart bypass on a patient with TA, and over the years there have been numerous modifications to the procedure improving shunt hemodynamics and circulation. 2 This historic event led to a shift in focus from palliation to long-term survival and quality of life in patients with TA. 3 With this new paradigm, numerous studies have shed light on the various long-term complications associated with the Fontan procedure.3,4 We describe a case of a long-term TA survivor who underwent Fontan surgery and presented to our hospital with shunt-related complications.
Case Presentation
A 33-year-old male with a medical history significant for TA status post-modified Fontan procedure approximately 30 years ago and atrial flutter presented with 1 week of progressive fatigue and malaise. He also endorsed body aches, anorexia, subjective fevers, chills, nausea, and 2 episodes of nonbilious, nonbloody emesis. Of note, he received his second dose of the Pfizer COVID-19 vaccine and had dental work performed without prophylactic antibiotics 2 weeks prior to presentation. He reported no chest pain, palpitations, shortness of breath, cough, diarrhea, leg swelling, night sweats, or unintentional weight loss. He was reportedly adherent to home medications including sotalol 80 mg daily and aspirin 81 mg daily. Additionally, he denied the use of tobacco, alcohol, or illicit drugs.
Upon presentation, the patient had a blood pressure of 105/85 mmHg, heart rate of 193 beats per minute (bpm), temperature of 98.2°F, and saturation of 98% on room air oxygen. His examination was notable for dry mucous membranes, scleral icterus, and tachycardia with an irregular rhythm. His pertinent laboratory studies are shown in Table 1.
Initial Laboratory Results.

Electrocardiogram showed narrow QRS tachycardia with a rate of 190 bpm and nonspecific ST changes (Figure 1). Computed tomography scan of the abdomen and pelvis revealed micronodular changes within the liver with perihepatic and perisplenic ascites (Figure 2).

Electrocardiogram demonstrating narrow QRS tachycardia with a rate of 190 bpm and nonspecific ST changes.
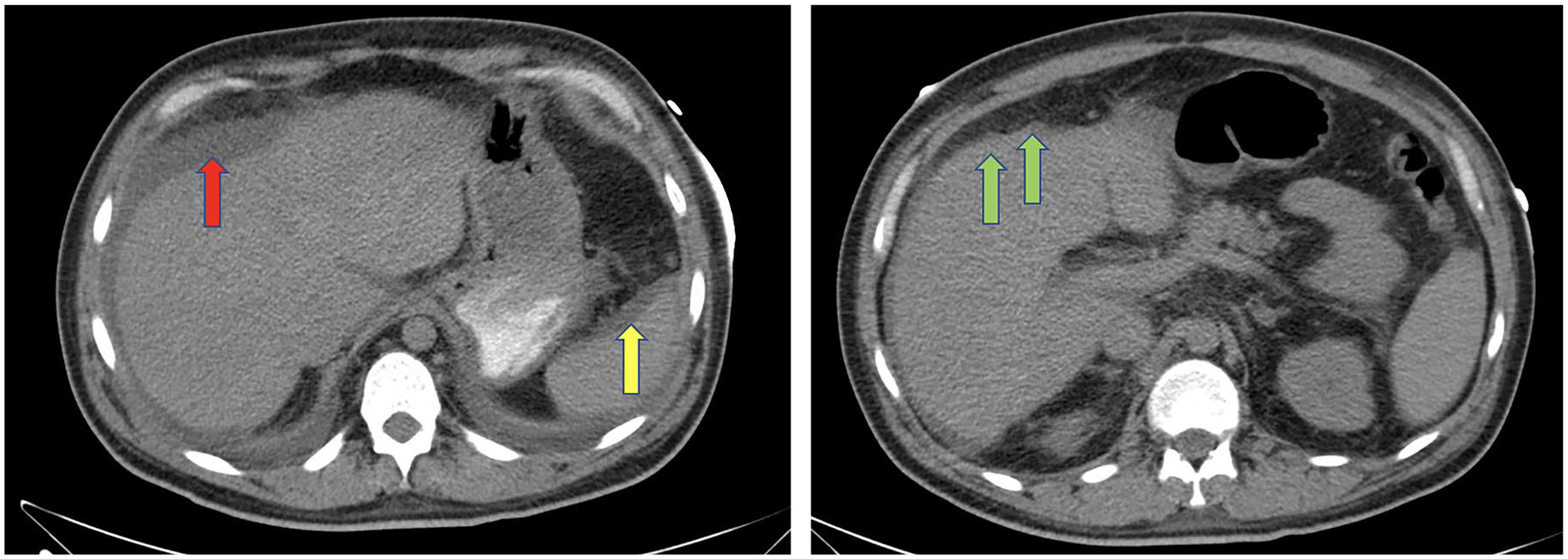
CT scan of the abdomen and pelvis demonstrating perihepatic (red arrow) and perisplenic (yellow) ascites along with micronodular changes (green arrow) in hepatic parenchyma.
The patient was started on intravenous (IV) diltiazem with initial bolus of 0.25 mg/kg followed by a rate of 10 mg/h, IV normal saline at a rate of 150 mL/h, 3 doses of IV N-acetyl cysteine at rates of 200, 125, and 62.5 mL/h, and prophylactic broad spectrum antibiotics including IV vancomycin 15 mg/kg every 24 hours and piperacillin-tazobactam 2.25 g every 6 hours. After resolution of his tachyarrhythmia, the patient was admitted for acute liver failure, complicated by atrial flutter with rapid ventricular response, and type 2 non-ST segment elevation myocardial infarction.
At this point, some of the leading differential diagnoses included medication-induced acute liver failure, viral hepatitis, infective endocarditis, hepatorenal syndrome, and congestive hepatopathy. Metabolic, infectious, and autoimmune work-up was conducted including and not limited to acetaminophen level, blood cultures, viral hepatitis panel, antinuclear antibody, smooth muscle antibody, and liver kidney microsomal antibody, resulting negative.
A transthoracic echocardiogram (TTE) demonstrated findings concerning for a right ventricular thrombus and a left ventricular ejection fraction of 41% to 45%. High-intensity heparin infusion was initiated and over the following 4 days, the patient’s clinical status improved to baseline. The patient was eventually transferred to a nearby institution with an adult congenital heart disease service and remained stable on outpatient follow-up. At a 3-month follow-up visit with his congenital cardiac disease specialist, the patient was in his usual state of health and TTE showed no evidence of thrombus or shunt malfunction.
Discussion
Tricuspid atresia is a congenital agenesis of the tricuspid valve. In TA, blood from the venous system is forced across the patent foramen ovale into the left atrium in prenatal circulation. As a consequence, 80% of the patients present with symptoms postnatally early on in life, in which 50% present on the first day of life and 30% present on the first month. 1 The presenting symptoms are usually cyanosis, dyspnea, and difficulty feeding. Tricuspid atresia is the third most common cyanotic congenital heart defect in the newborn, with a prevalence of 0.3% to 3.7% in patients with congenital heart defects, and 1 per 10 000 of all live births. 5 While some studies in mice with TA have shown mutations in FOG2 gene play a role in cardiac development, the pathogenesis is still unknown. 6 Tricuspid atresia can be divided into 3 types: normal anatomy of the great vessels, dextro-transposition of the great vessels, and malposition of the great vessels other than dextro-transposition. 4 Based on the imaging and cardiac magnetic resonance image from a previous admission, we believe that our patient had type 1, normal anatomy of great vessels. The current management for TA thus far is the Fontan procedure, while the definitive treatment for failing Fontan circulation remains cardiac transplantation. 7
The main principle of the Fontan procedure is rerouting the systemic venous blood to the pulmonary arteries, bypassing the right ventricle. The original Fontan procedure consisted of direct anastomosis of the inferior vena cava to the left pulmonary artery and anastomosis of the superior vena cava to the right pulmonary artery superiorly. A recent meta-analysis evaluating 5859 patients with Fontan circulation found survival estimates at 5, 10, and 15 years to be 90.7%, 87.2%, and 87.5%, respectively. 3 Originally, it was thought that the right atrium will improve pulmonary blood flow but was found later through experiments by de Leval et al that the right atrium lost its contractility. 8 The right atrial failure not only failed to maintain the pulmonary flow but created turbulent flow which later resulted in thrombosis and risk of arrhythmia. 9 These complications have led to the discovery of a modern Fontan procedure that involves anastomosis of the SVC to the right pulmonary artery superiorly and IVC to the right pulmonary artery inferiorly via either an intra-atrial lateral tunnel conduit or an extra-atrial cardiac conduit. 9 In the intra-atrial conduit, part of the atrial wall is used to form the conduit tunnel, whereas in extra-atrial method only prosthetic material is used. 9 Intra-atrial tunnel remains more advantageous than extra-atrial as the conduit enlarges as the child grows, thus it can be used as an option for newborns. 9 The use of intra-atrial tunnel, however, has the risk of arrhythmia. 9 Moreover, the use of any intravascular prosthetic material augments the risk of thrombosis and occlusion. In the intra-atrial method, a small opening between the conduit and the right atrium is created. This functions as a valve, creating a right to left shunt, to prevent pulmonary congestion and to maintain caval pressure. 10
Although Fontan circulation has proven to be an effective therapeutic intervention, the procedure itself is associated with many long-term complications, including and not limited to myocardial dysfunction, arrhythmias, aortic root insufficiency, and thrombosis. 11 Furthermore, myocardial dysfunction coupled with shunt failure and a pumpless pulmonary circulation can lead to increased caval pressure and the subsequent development of portal hypertension, cirrhosis, liver failure, and increased risk for hepatic carcinoma.11,12 A retrospective study by Kiesewetter et al suggested that cirrhosis is more common in Fontan shunt associated with elevated venous pressure and that hepatic dysfunction is correlated with the duration of Fontan circulation. 12
Evidently, our patient was discovered to have occlusion in intra-atrial conduit and dilation of the right atrium on a TTE. Furthermore, the patient’s acute liver failure, dilation of the right atrium on echocardiogram and atrial flutter, suggests a failure of the Fontan circulation as a possible mechanism of his symptomatology.
Conclusion
Tricuspid atresia is a congenital anomaly that can be associated with significant morbidity and mortality. Our patient’s clinical course can be attributed to altered Fontan shunt hemodynamics and subsequent reopening of intracardiac shunt resulting in reestablishment of normal physiological blood flow. Further studies evaluating the long-term outcomes of the various types of Fontan shunts in TA are warranted to expand our understanding of congenital cardiac pathologies.
Footnotes
Acknowledgements
The authors have no additional acknowledgements to make.
Authors’ Note
Prior Presentation of Abstract Statement: This article has not been presented at a meeting.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
