Abstract
The differential diagnosis of a patient presenting with constitutional symptoms and pulmonary cavitary lesions on chest X-ray is broad, including pulmonary tuberculosis (TB) and vasculitic lung disease. Clinicians may consider sociodemographic factors in sequencing their differential, which should be verified with diagnostic tests according to appropriate pretest probability. Here, we present a case of misdiagnosing granulomatosis with polyangiitis as pulmonary TB. Although native to an endemic country, this patient had multiple negative TB tests and did not respond to rifampin, isoniazid, pyrazinamide, and ethambutol therapy. We consider anchoring implicit bias as a contributing factor for this patient’s misdiagnosis and make recommendations for clinicians.
Background
Granulomatosis with polyangiitis (GPA, formerly known as Wegner’s granulomatosis) is an uncommon small-medium vessel necrotizing vasculitis. 1 It’s estimated that the annual global incidence of GPA is 10 to 20 cases/1 million. 2 GPA is an anti-neutrophil-cytoplasmic-antibody (ANCA)-associated vasculitis syndrome associated with c-ANCA (proteinase 3). GPA typically presents with nonspecific symptoms including fever, malaise, weight loss, and myalgias. Ninety percent of patients also have upper and lower respiratory tract involvement, and fifty percent of patients will have pulmonary infiltrates on chest imaging. 1 Other important symptoms include vasculitic skin findings and glomerulonephritis. Management includes empiric antibiotics, oxygen support, and immunosuppressants to trigger remission of the disease, followed by either corticosteroids and/or immunosuppressants for maintenance. 3
Given their significant overlap, there are documented case reports of GPA misdiagnosed as tuberculosis (TB), and vice versa.4-6 TB is likely at the top of a clinician’s differential diagnosis for a patient from an endemic country presenting with cavitary lung lesions. However, when extra-pulmonary symptoms are involved, the differential should be broadened to include less common diagnoses.
When considering competing diagnoses in a differential, a physician must consider and apply appropriate pretest probabilities in selecting diagnostic and confirmatory tests. Bayes’ Theorem posits that the likelihood of disease depends on the patient’s context and diagnostic evidence. 7 If a physician focuses too much on context, this may lead to anchoring bias: a premature focus on initial information without appropriately adjusting to later evidence. 8 Often, this initial information is history, sociodemographic, or epidemiologic data pertaining to the patient. Occasionally, these biases fail, and physicians may order unnecessary tests, or fail to order the appropriate tests, delaying the workup and correct diagnosis, all of which leads to inappropriate therapy. 9 Moreover, previous systematic reviews have shown that implicit bias, influenced by race and disability, has been strongly associated with treatment decisions. 10 This can potentially lead to patient harm, greater healthcare costs, and substandard care.
Case Presentation
A 45-year-old Hindi-speaking male with prior BCG vaccine, who immigrated from India 1 year ago, was admitted with a 6-week history of shortness of breath, significant weight loss, nonproductive cough, and a back abscess.
The patient subsequently developed hemoptysis, leading to clinical suspicion of TB. Empiric therapy of rifampin, isoniazid, pyrazinamide, and ethambutol (RIPE) was initiated. Although PCR tests and sputum cultures for TB returned negative, RIPE therapy was continued based on the clinical suspicion of TB, with malignancy and fungal infection considered less likely.
The patient experienced ongoing nausea and vomiting, attributed to the RIPE regimen, and an episode of supraventricular tachycardia. Still on RIPE, he was discharged after 12 days inpatient with a plan for health department follow-up.
Ten days after discharge, the patient was readmitted due to worsening systemic symptoms, including fever, generalized weakness, hypotension, and tachycardia, all thought to be due to RIPE therapy toxicity. Given his deteriorating condition, further evaluation was conducted at a tertiary care center.
The differential diagnosis was then expanded to include GPA due to additional findings of epistaxis, hearing loss, scleral injection, and worsening cavitary lung lesions. Dermatology and rheumatology services were consulted at this time. Laboratory testing, including positive c-ANCA and specific complement levels, supported the diagnosis of GPA. A skin biopsy confirmed small vessel vasculitis.
The patient was discharged on prednisone with a confirmed diagnosis of GPA and continues to follow-up with rheumatology for rituximab infusions. Two weeks after the second hospital admission, the patient reported feeling better and complained only of open wounds on his feet bilaterally and pretibial edema. He reports no epistaxis, fever, chest pain, or hemoptysis.
Discussion
This case highlights the critical role of differential diagnosis in patients presenting with complex and nonspecific symptoms, particularly when the initial diagnostic approach is heavily influenced by epidemiological factors. This patient, a recent immigrant from India, presented with a constellation of symptoms, including weight loss, nonproductive cough, hemoptysis, and cavitary lung lesions, which are classic features of TB. Given his origin from a TB-endemic region, it was reasonable to prioritize TB in the initial differential diagnosis. However, this focus led to “anchoring,” where subsequent evidence was discarded due to incompatibility with the initial diagnosis. 11
Anchoring bias in medicine can result in the premature closure of the diagnostic process, leading to delayed or missed diagnoses of other conditions. In this case, despite the lack of clinical improvement with RIPE therapy and multiple negative TB test results, the differential remained overly focused on TB. The possibility of GPA, a rare but serious vasculitic disorder, was not initially considered. GPA is a critical diagnosis to consider when the clinical response to TB treatment is suboptimal. 2 The delay in considering GPA in this patient’s diagnostic workup may have contributed to his prolonged morbidity.
This case illustrates the importance of re-evaluating the working diagnosis when new clinical signs emerge, or the patient does not improve as expected. It further illustrates the unconscious cognitive biases clinicians may hold (in this case, that TB is the most likely culprit given the patient’s country of origin) that are sometimes employed in workups and testing; these biases modulate what “appears” to be the most likely scenario. Taking stock of one’s unconscious biases is essential when treating an increasingly diverse patient population. Although epidemiological factors should be considered in developing a differential, it should not be the primary driver of initiating empiric therapy.
Had GPA been considered earlier, targeted immunosuppressive therapy could have been initiated sooner, potentially improving the patient’s outcome. While a multidisciplinary approach was eventually taken, coalescing evidence from a bottom-up approach rather than a top-down approach with a single culprit in mind may improve the accuracy of the differential.
While TB remains a critical diagnosis to consider in patients from endemic areas presenting with cavitary lung lesions, clinicians should remain vigilant of alternative diagnoses, such as GPA. Early recognition and treatment of GPA are essential to preventing irreversible organ damage and improving outcomes.
Conclusion
This patient presented with complex nonspecific symptoms and pulmonary lesions on chest X-ray. Despite negative TB testing, he was initiated on RIPE therapy; he subsequently had a poor clinical response to the regimen and later re-presented with additional “typical” vasculitis symptoms. During the second admission, an interdisciplinary approach was taken to broaden the differential, and a correct diagnosis of GPA was made. This case report considers the consequences of anchoring bias in workups and demonstrates the importance of re-evaluating the differential when clinical signs suggest alternative diagnoses (Figures 1-3).

Chest X-ray during first hospitalization demonstrating 5 cm right suprahilar mass, 2.6 cm cavitating mass in left lower lobe, and 12.6 mm nodule in the medial left lower lobe.
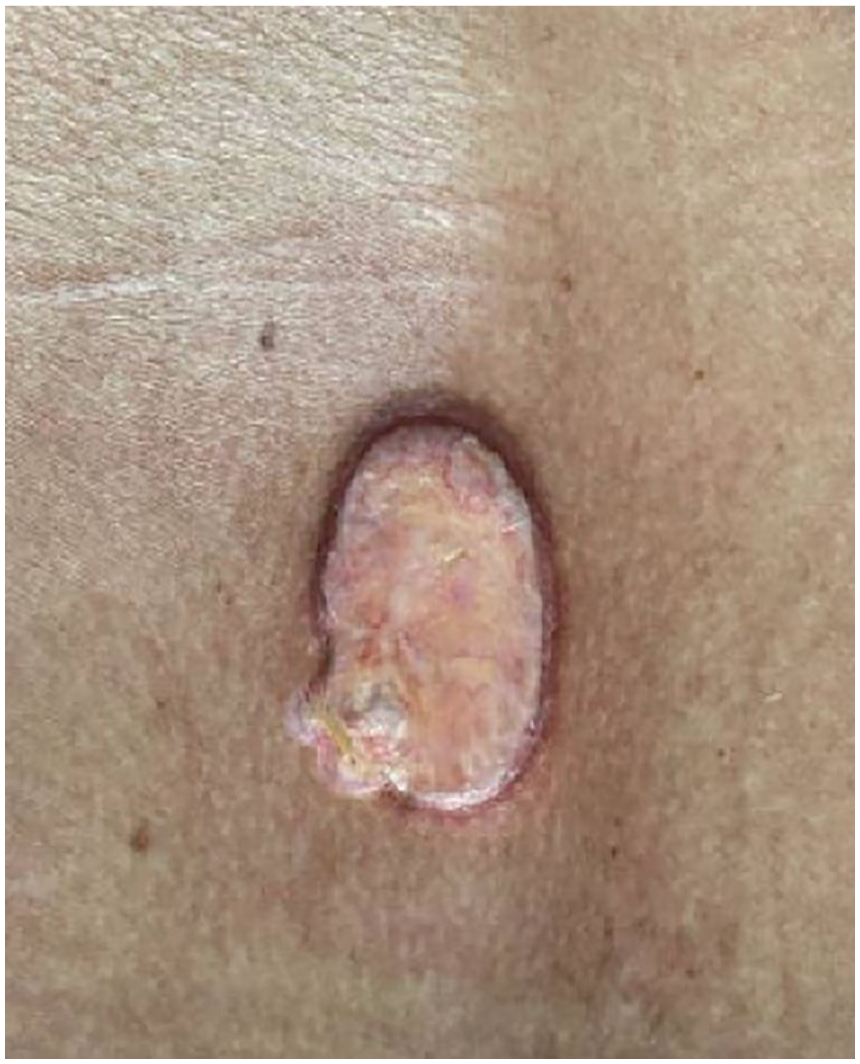
Back lesion with purulent discharge.
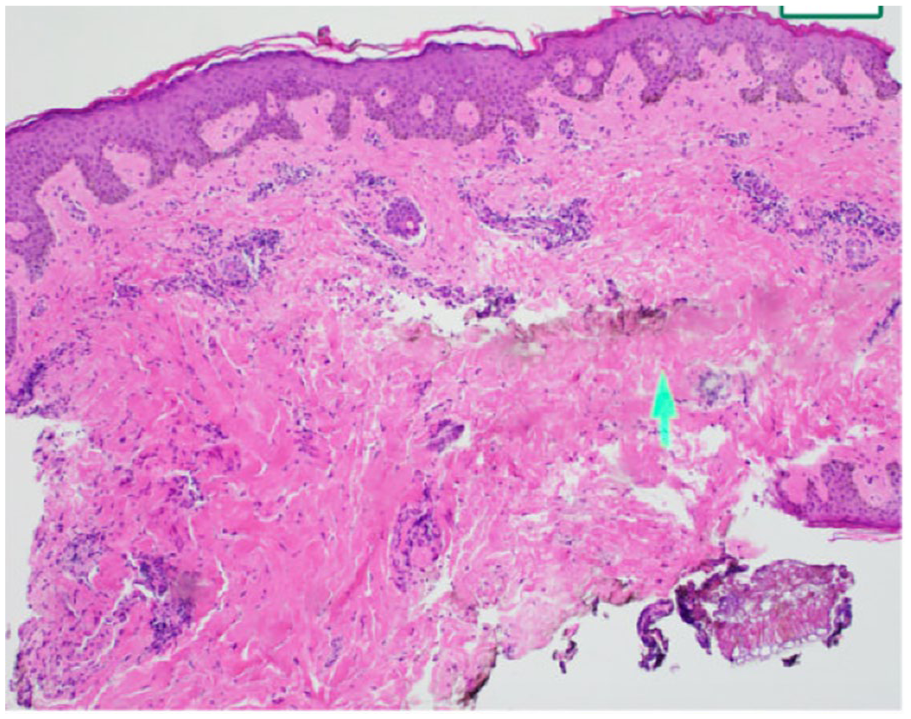
Nasal biopsy shows punctate areas of necrotizing granulomatous inflammation within sinonasal submucosa, eroding bone with dense acute and chronic lymphoplasmacytic inflammation and granulation type tissue.
Footnotes
Acknowledgements
N/A.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
N/A.
