Abstract
Although well documented, constrictive pericarditis is a rare entity and an uncommon cause of heart failure. A stiff and noncompliant pericardium creates the disease’s unique hemodynamics and leads to elevated venous pressures, hepatic sinusoidal congestion, and draining of protein-rich fluid into the peritoneal cavity presenting as ascites. The low incidence in addition to its varied and subtle clinical presentations can often lead to a delay in diagnosis. Here, we present 2 clinical cases of constrictive pericarditis in which ascitic fluid analysis was important—one patient who presented with new-onset ascites with concern for cirrhosis and another patient who presented with symptoms concerning for heart failure with ascites. Through their hospital course and workup, we highlight the importance of diagnostic sampling of ascitic fluid to prompt the consideration of constrictive pericarditis followed by utilizing advanced diagnostics, such as echocardiogram and cardiac catheterization to reach the correct diagnosis in an otherwise often overlooked pathology.
Keywords
Introduction
Constrictive pericarditis is an uncommon cause of heart failure with preserved ejection fraction. While most cases in North America are idiopathic, risk factors include prior cardiac surgeries, chest radiation therapy, and prior acute pericarditis. 1 The pathophysiology is complex and can lead to manifestations within several organ systems. An inelastic pericardium limits cardiac filling leading to decreased preload, stroke volume, and cardiac output. 1 As the right ventricle is constricted in space, it is unable to fill properly, leading to elevated venous pressures and back flow into the valveless hepatic venous system. 2 This causes hepatic sinusoidal congestion and increased permeability of the liver resulting in protein-rich fluid draining into the peritoneal cavity presenting as ascites. Patients typically present with heart failure symptoms including lower extremity edema, dyspnea on exertion, palpitations, fatigue, and chest discomfort. 1 Less often patients present with advanced symptoms from hepatic congestion such as jaundice and ascites. One study showed that only 8% of patients with constrictive pericarditis present with abdominal pain and distension as their initial complaints and only 13% had an elevated bilirubin. 2
Here, we present 2 cases of constrictive pericarditis where ascitic fluid analysis was key to the diagnosis. The first case describes a patient with a recent clinical diagnosis of cryptogenic cirrhosis with ascites. The second case involves a patient presenting with typical heart failure symptoms along with ascites. Despite the differing clinical presentations, ascitic fluid analysis followed by advanced diagnostic testing helped elucidate their shared underlying diagnosis.
Case 1 Description
A 68-year-old man with a medical history of hypertension (HTN), hyperlipidemia (HLD), alcohol abuse, obesity, and recent diagnosis of cryptogenic cirrhosis presented with volume overload with ascites.
Five months prior, the patient received a clinical diagnosis of cryptogenic cirrhosis after presenting with acute encephalopathy, elevated ammonia, and imaging consistent with cirrhotic morphology. The etiology of his cirrhosis was considered multifactorial given his persistent alcohol use, HTN, HLD, obesity, and family history of nonalcoholic fatty liver disease (NAFLD). He did not undergo liver biopsy and previously never had ascites requiring diagnostic paracentesis.
On admission, physical examination revealed tense ascites and profound scrotal and lower extremity edema. His serum studies were normal outside of a mildly elevated bilirubin of 2.8 mg/dL, international normalized ratio (INR) of 1.45, and brain natriuretic peptide (BNP) of 301 pg/mL. Diagnostic paracentesis revealed an elevated serum ascites albumin gradient (SAAG) of 1.2 g/dL and elevated ascitic protein of 3.4 g/dL. The SAAG was consistent with portal HTN; however, cirrhosis was unlikely to cause the ascitic protein level to be >2.5 g/dL. These results led to a deeper investigation into the patient’s history and revealed he had an episode of pericarditis in his twenties with calcification years following. Transthoracic echocardiogram (TTE) was pursued and revealed a normal ejection fraction with evidence of thickened, calcified pericardium. In addition, it revealed several factors concerning for constrictive physiology including septal bounce, annulus reversus (mitral septal annular velocity = 0.16 m/s > mitral lateral annular velocity = 0.12 m/s), dilated, non-collapsible inferior vena cava, and biatrial enlargement with signs of ventricular interdependence and hepatic vein reversal (Figure 1).

Transthoracic echocardiogram doppler imaging that shows the pathophysiology during expiration that causes decreased right ventricular filling and hepatic vein reversal during late diastole.
The patient underwent left and right heart catheterization which revealed normal cardiac output and elevated filling pressures (Table 1). Dedicated computed tomography (CT) heart imaging confirmed the diagnosis of constrictive pericarditis (Figure 2) and demonstrated a large loculated pericardial collection exerting mass effect on the right heart chambers. His clinical diagnosis of cryptogenic cirrhosis was believed to be the result of congestive hepatopathy from longstanding constrictive pericarditis.
Right Heart Catheterization Results Confirmed Elevated Right-Sided Heart Pressures With Normal Cardiac Output.

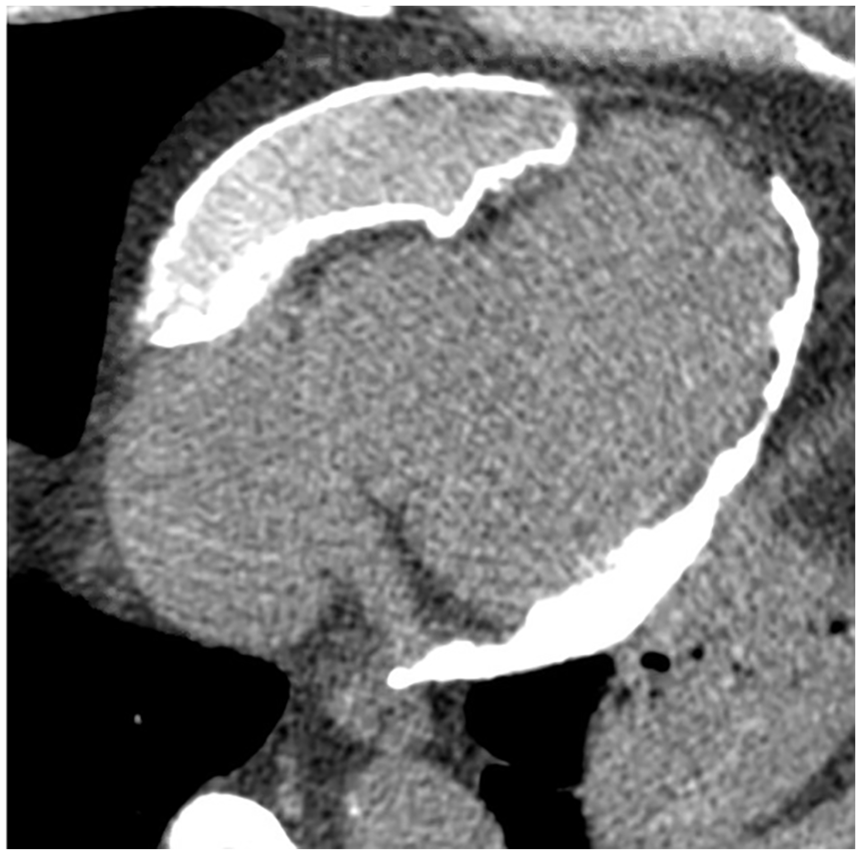
Computed tomography (CT) of the heart revealed constrictive pericarditis with thick circumferential pericardial calcifications and a loculated anterior pericardial collection overlying and exerting mass effect on the right heart chambers.
The patient underwent successful complete pericardiectomy with a reduction in right atrial pressure from 33 to 10 mmHg intraoperatively. He was discharged to rehab with outpatient follow-up with cardiothoracic surgery and hepatology. He did very well as an outpatient reporting it was the “best he had felt in years” and was back to doing usual activities such as golfing, which he was previously unable to do. A few months later, he underwent liver biopsy which revealed cardiac hepatopathy with sinusoidal dilation and liver outflow obstruction. There was no pathologic evidence of cirrhosis.
Case 2 Description
A 60-year-old man with a medical history of HTN, alcohol use disorder, and obesity presented with a chief complaint of lower extremity swelling.
This patient reported that he had a long history of bilateral lower extremity swelling for which he had been prescribed increasing doses of furosemide over the last year. In the last 3 weeks, his edema had gotten much worse and was associated with abdominal bloating and right-sided discomfort in addition to increased shortness of breath and chest tightness with activity. His symptoms had gotten to the point where he was so short of breath at his job that his supervisor encouraged him to go to the emergency department (ED) to be evaluated.
On arrival, the patient had signs of hemodynamic instability with blood pressure of 92/58 and pulse of 122. He was otherwise afebrile with an oxygen saturation of 96% on room air. On examination, he was tachycardic with regular rhythm. He had decreased heart sounds without murmurs and no jugular venous distension. His abdomen was distended with an appreciable fluid wave. He also had mild bibasilar crackles and 3+ pitting edema. Laboratory workup was most notable for normal electrolytes, decreased bicarbonate of 19 meq/L, increased anion gap 20, and increased lactic acid of 5.3 mmol/L. He also had elevated creatinine of 1.54 mg/dL, increased total and direct bilirubin of 3.1 and 1.5 mg/dL respectively, normal albumin of 3.6 g/dL, and elevated BNP of 482 pg/mL. A bedside cardiac ultrasound was performed in the ED that was notable for a small to moderate pericardial effusion, significant pericardial calcifications, septal bounce, and appearance of ventricular interdependence. Subcostal images revealed a very enlarged inferior vena cava and ascites. Diagnostic paracentesis was performed, and ascitic fluid analysis was notable for albumin of 1.8 g/dL, total protein of 3.3 g/dL, and absolute neutrophil count of 40/mm3. His SAAG of 1.8 g/dL and elevated total ascitic protein raised further suspicion of a cardiac etiology for his ascites. The patient underwent cardiac catheterization, which demonstrated elevated left and right heart filling pressures, normal cardiac output and index, and simultaneous right and left ventricular pressures with interdependence, pressure equalization, and sharp y descents all consistent with constriction (Figure 3).
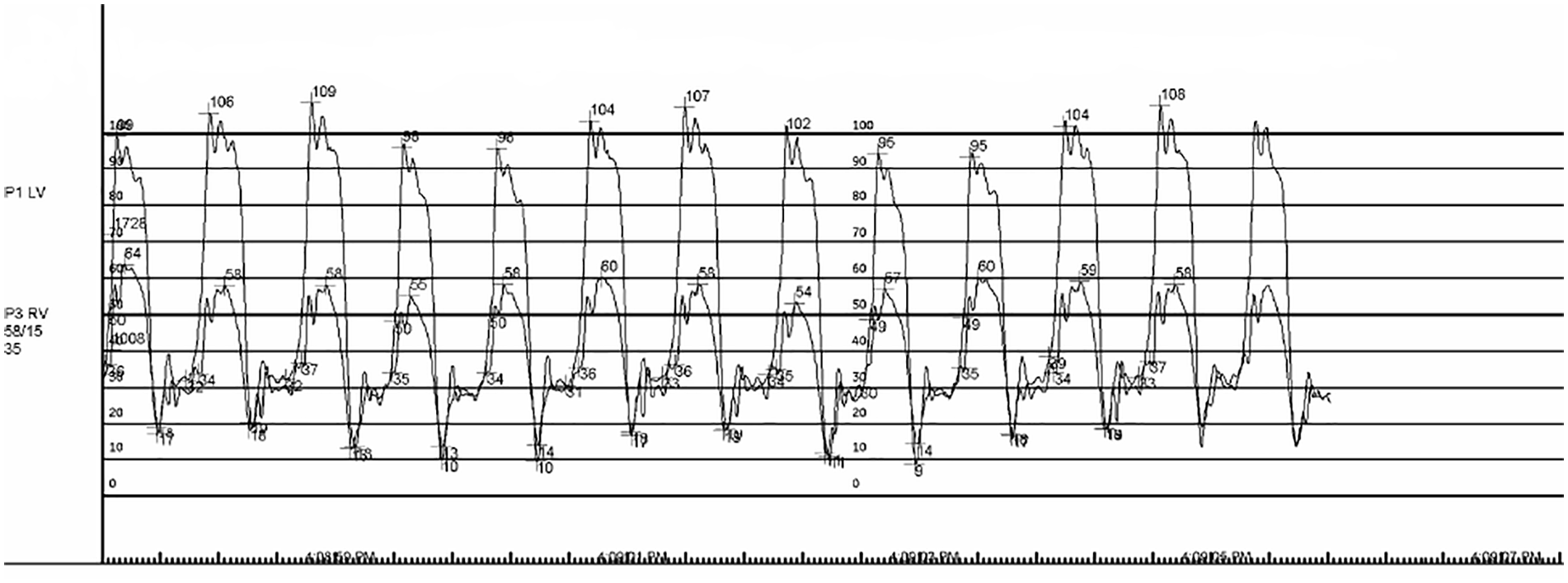
Simultaneous right and left heart catheterization showing ventricular interdependence.
His laboratory workup and catheterization findings were consistent with congestive hepatopathy secondary to constrictive pericarditis. The patient underwent aggressive diuresis in addition to a large volume paracentesis before undergoing a pericardiectomy. Intraoperative cultures grew Cutibacterium acnes, a rare cause of constrictive pericarditis, and the patient was treated with a prolonged course of ceftriaxone.
Discussion
We present 2 cases of constrictive pericarditis where ascitic fluid analysis was key to the diagnosis. While cirrhosis is the underlying etiology of ascites in a large majority of people (85%), one must consider other causes of peritoneal fluid accumulation. 3 Seven percent of ascites is due to peritoneal malignancy, 3% is due to heart failure, while the remaining 5% is due to multiple causes. 4
According to the American Association for the Study of Liver Disease, diagnostic paracentesis is first line to assist with diagnosis of new-onset ascites. 4 A serum albumin ascites gradient (SAAG) of greater than or equal to 1.1 is consistent with ascites secondary to portal HTN. 3 When a SAAG is ≥1.1, a total protein level of greater than or equal to 2.5 g/dL is consistent with portal HTN due to congestive hepatopathy from heart failure. One study found that a SAAG of ≥1.1 and ascites fluid total protein of ≥2.5 g/dL was 87% specific and 53% sensitive to diagnosing ascites from heart failure and congestive hepatopathy. 3 As compared to cirrhosis, ascites in heart failure has higher protein levels as the sinusoids are normal and leak protein-rich fluid into the peritoneal cavity. In cirrhosis, sinusoids undergo capillarization and do not leak these large proteins. 3 Both of our patients had ascites protein levels greater than 3 g/dL.
In addition to ascitic fluid analysis, diagnosis of constrictive pericarditis requires imaging and cardiac catheterization. B-type natriuretic peptide is often of little utility as there is only minimal elevation given lack of significant ventricular stretching. 1 In 27% of cases, chest radiograph will show pericardial calcifications. 1 CT and magnetic resonance imaging (MRI) can also demonstrate pericardial thickening and calcification (Figure 4). A respiration-related ventricular septal shift on TTE is 93% sensitive for the diagnosis of constrictive pericarditis. 1 This is shown by the interventricular septum moving toward the left ventricle during inspiration and back to the right with expiration. As the patient inspires and the intrathoracic pressure becomes more negative, the right ventricle is unable to expand to allow venous return. Therefore, the interventricular septum shifts toward the left ventricular space leading to decreased left ventricular volume and cardiac output. 1 Cardiac catheterization also captures this ventricular interdependence through simultaneous left and right heart pressure tracings. The right and left ventricular pressures are discordant and vary with respiration. As the right ventricular pressure increases during inspiration, the left ventricular pressure decreases and vice versa. This is highly specific for constrictive pericarditis, making cardiac catheterization the gold standard for diagnosis. 5 Along with ascitic fluid studies, MRI, TTE, and cardiac catheterization were vital to assist in establishing the diagnoses for our 2 patients.

Computed tomography (CT) of the chest showing a large hyperdense pericardial effusion and calcifications.
Another important finding on TTE is the reversal of hepatic flow during diastole. As the right ventricle is constricted in space, it is unable to fill properly leading to elevated venous pressure and back flow into the valveless hepatic venous system. 2 This leads to sinusoidal congestion and increased permeability of the liver causing protein-rich fluid to drain into the peritoneal cavity.
Symptom management includes diuresis followed by pericardiectomy as the definitive treatment. 6 Survival after pericardiectomy is dependent on the etiology of constrictive disease. One case series showed that patients with idiopathic etiology had the best chance of 7-year survival with 88% alive 7 years after surgery, compared to 27% and 66% in the post-radiation group and post-surgical group, respectively. 7 Model for end-stage liver disease (MELD) score has been shown to be an independent predictor of mortality for patients who undergo pericardiectomy. 8
In summary, we present 2 clinical presentations of constrictive pericarditis where ascitic fluid analysis demonstrated cardiac ascites prompting further diagnostic workup for constrictive pericarditis. Although rare, it is crucial to keep cardiac etiologies on the differential for patients with new-onset ascites.
Footnotes
Acknowledgements
All authors had a role in writing this case report manuscript. No external funding for this work has been provided.
Authors’ Note
The first case was presented as a poster presentation at the 2021 ACG Annual Scientific Meeting in Las Vegas, NV, in October by one of our authors. The conference also published this work as an abstract in The American Journal of Gastroenterology, Volume 116, October 2021.
Author Contributions
Sarah Barbina: drafting of the manuscript; critical revision of the manuscript for important intellectual content
Umakanthan Kavin: drafting of the manuscript; critical revision of the manuscript for important intellectual content
Michael Sutton: drafting of the manuscript; critical revision of the manuscript for important intellectual content
Marc Heincelman: drafting of the manuscript; critical revision of the manuscript for important intellectual content
Meghan Thomas: drafting of the manuscript; critical revision of the manuscript for important intellectual content, supervisory
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal informed consent was obtained from the patients for their anonymized information to be published in this article.
