Abstract
Constrictive pericarditis (CP) presents as a pathophysiological state where the pericardium becomes inelastic due to fibrotic changes, most commonly secondary to a protracted inflammatory process. The disease is characterized by compromised diastolic cardiac function due to loss of pericardial compliance. Immunoglobulin G4 (IgG4)-related disease, an entity marked by the insidious proliferation of IgG4-positive plasma cells and subsequent fibrosis within various organs, is an infrequent but recognized cause of CP. A case of a 55-year-old male patient with clinical manifestations of dyspnea and edema in the lower extremities elucidates the diagnostic complexity inherent to CP. Echocardiography revealed a constellation of signs, including annulus reversus, septal bounce, and a congested inferior vena cava; cardiac magnetic resonance imaging (MRI) demonstrated diffuse pericardial thickening with delayed gadolinium enhancement, suggestive of a long-term inflammatory state; and right heart catheterization confirmed the hemodynamic hallmark of CP—equalization of diastolic pressures across the cardiac chambers. The serological analysis elicited elevated serum levels of IgG4 and IgE, pointing to the differential diagnosis of IgG4-related disease. Given the nonspecific clinical presentation of IgG4-related CP, a heightened index of suspicion combined with a systematic approach to imaging and serological evaluation is paramount.
Keywords
Introduction
Constrictive pericarditis (CP) is characterized by impaired diastolic filling due to a thickened and fibrotic pericardium that follows inflammation. Long-term inflammation may result from idiopathic causes, viral pericarditis, cardiac surgery, radiation therapy, or infections, including tuberculosis, human immunodeficiency virus, and uremia. 1 The most common cause in the Western part of the world is idiopathic and tuberculosis in Africa and Asia. 1 The incidence of CP is estimated to be less than one person among 100 000 people annually. 1 Patients with CP may present with symptoms of left or right heart failure, such as peripheral edema, shortness of breath, abdominal discomfort due to hepatic congestion, anasarca, and cachexia. 2 Patients can be misdiagnosed with restrictive cardiomyopathy or hepatic cirrhosis, which could not only delay diagnosis and management but also lead to unnecessary interventions. 2
The diagnosis of CP often starts with a high clinical suspicion based on the patient’s symptoms and history. Transthoracic echocardiography (TTE) is the initial diagnostic tool, identifying signs of ventricular interdependence indicative of CP, such as annulus reversus, septal bounce, and plethoric inferior vena cava (IVC). 3 These findings reflect the hemodynamic impact of a rigid pericardium on ventricular filling dynamics. Tissue Doppler and speckle tracking imaging further distinguish CP from restrictive cardiomyopathy, often showing preserved global longitudinal strain (GLS) in CP. 3 The presence of pericardial calcification on chest X-rays can be pathognomonic, but its sensitivity is low. Cardiac magnetic resonance imaging (MRI) is highly accurate in detecting pericardial thickening and can be used to assess the extent of active inflammation, guiding the therapeutic approach. Cardiac MRI excels in delineating myocardial involvement, displaying delayed gadolinium enhancement characteristic of IgG4-related fibrosis.3,4 When MRI is not feasible, computed tomography (CT) scans can be used in detecting pericardial calcifications and can provide information on alternative causes of dyspnea. 3 Utility of positron emission tomography (PET) in CP diagnosis is not well-established. 4 Right heart catheterization remains the definitive diagnostic procedure for CP, revealing a “dip-and-plateau” pressure pattern, equalization of end-diastolic pressures, and ventricular discordance during the respiratory cycle, phenomena resulting from the pericardium’s decreased compliance and the consequent hemodynamic effects.3,4
Treatment usually involves pericardiectomy, which is the definitive management for long-term CP. 2 Some patients with subacute inflammatory CP may respond to anti-inflammatory therapy. 2
Case Presentation
A 55-year-old male with history of type 2 diabetes mellitus presented to the emergency department (ED) with subacute exertional dyspnea, fatigue, worsening lower extremity edema, unintentional weight loss of approximately 20 pounds over the past 6 months, and most recently, daily episodes of scant hemoptysis.
Upon arrival, the patient was afebrile with a heart rate of 77 beats per minute, a blood pressure of 114/76 mm Hg, and an oxygen saturation of 98% on room air. The examination was significant for decreased breath sounds in the right lower lobe, normal S1 and S2 without murmurs, knocks, rubs, or gallops, elevated jugular venous distension (JVD), and 4+ pitting edema bilaterally, extending up to the abdomen. Laboratory studies showed an elevated C-reactive protein of 12.2 mg/L (reference range: <9.9 mg/L), and an elevated brain natriuretic peptide (BNP) of 137 pg/ml (reference range: 0-100 pg/ml), but electrolyte levels and troponin levels were within normal limits. An electrocardiogram (EKG) illustrated normal sinus rhythm with a normal axis and no stigmata of recent or past ischemia. A chest radiograph (XR) revealed a large right-sided pleural effusion.
Given the elevated BNP and signs and symptoms of congestive heart failure, the patient underwent a TTE. The initial TTE poorly visualized left ventricular ejection fraction and global right ventricular systolic function. The IVC was dilated and collapsed more than 50% with respiration. Hepatic vein inspiratory reversal was mild and appeared similar to expiratory flow reversal, with minimal flow reversal augmentation on expiration. Given the concern for CP, a cardiac MRI was obtained to better visualize the structures and detect possible active inflammation. The MRI demonstrated diffuse thickening of the pericardium with increased enhancement on delayed myocardial enhancement and an adequate ventricular ejection fraction of 45% (Figure 1). A repeat echocardiogram revealed a septal bounce with annulus reversus and a plethoric IVC; the GLS was −19 (Figure 2), findings suggestive of CP. A left heart catheterization was planned to evaluate the presence of coronary artery disease, given the patient’s signs and symptoms of new-onset heart failure, and to assess the left heart’s hemodynamics prior to possible pericardiectomy. The patient was treated with intravenous diuresis and systemic steroids and subsequently underwent a left heart catheterization, which showed nonobstructive coronary artery disease. A right heart catheterization provided evidence of equalization of end-diastolic pressures, the “dip and plateau” sign, and ventricular discordance consistent with CP (Figure 3).

Cardiac magnetic resonance imaging showing diffused thickening of the pericardium with delayed gadolinium enhancement.

Panel A. 2D echocardiography, parasternal short axis view showing respiratory related septal shift. Panel B. Shows a dilated inferior vena cavae of 2.33 cm with less than 50% respiratory variation. Panel C and D. Tissue Doppler imaging demonstrated higher medial than lateral early diastolic annulus velocities (17.1 cm/s and 14.6 cm/s, respectively). Evidence of annulus reversus.

Panel A. Right heart catheterization showing “dip and plateau” or “square root sign” Panel B. Right heart catheterization shows equalization of diastolic pressures pulmonary capillary wedge pressure of 26, left ventricle pressure of 24, right ventricle pressure of 23, and right atrium pressure of 26. Left ventricle and right ventricle tracing with ventricular discordance.
The differential diagnosis included CP secondary to infection (such as tuberculosis or viral), neoplasm (such as myeloproliferative disorders), autoimmune processes, or IgG4-related disease (IgG4-RD). Constrictive pericarditis secondary to tuberculosis or a malignant process was initially suspected, given the incidental findings on chest XR and the presence of constitutional symptoms. However, the QuantiFERON Gold test for tuberculosis was negative, and thoracentesis yielded 1600 mL of serous fluid, which was transudative based on Light’s criteria. Autoimmune markers were also within normal limits. Elevated total IgG and IgE levels raised clinical suspicion for IgG4-RD, with serum IgG4 levels of 1216 mg/dL (reference range: 2-96 mg/dL) and IgE levels of 645 IU/mL (reference range: 6-495 IU/mL) being noted.
The patient underwent a pericardiectomy, and pathology revealed a thickened fibrotic pericardium with focal mild long-term inflammation and many hemosiderin-laden macrophages (Figure 4). Cytochemistry with Congo red staining was negative for amyloid. No significant tissue eosinophilia was noted, and immunohistochemical staining for IgG4 was not performed. A bone marrow biopsy with immunohistochemical analysis revealed polyclonal plasma cells without an overt increase in eosinophils. The patient did not exhibit symptoms, serological signs, or imaging findings suggestive of other organ involvement in IgG4-RD.
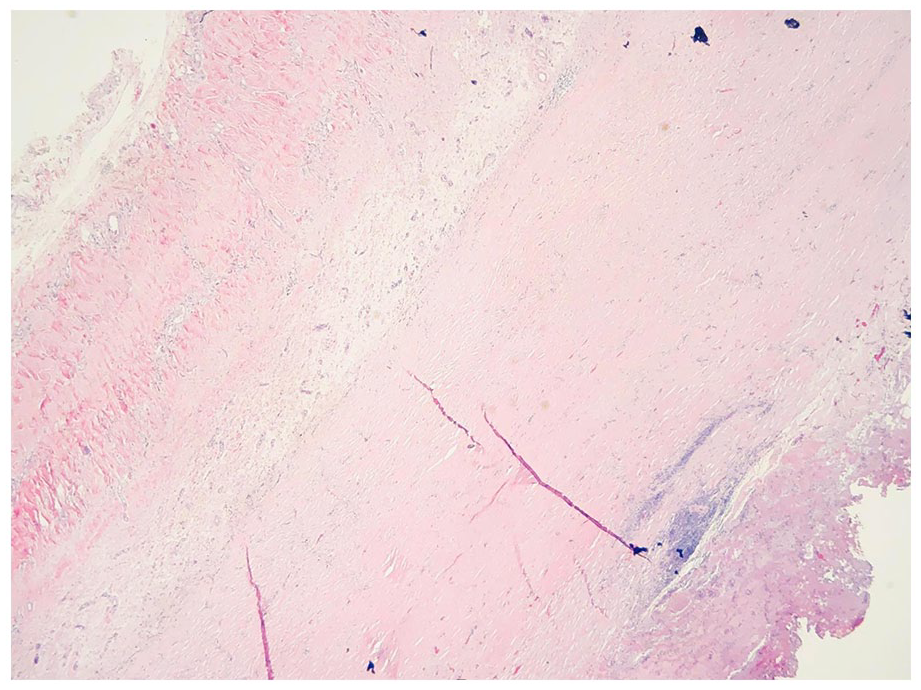
Hematoxylin & Eosin stain with thickened fibrotic pericardium with hemosiderin-laden macrophages and eosinophils.
Upon discharge and at a 2-month follow-up after the pericardiectomy, the patient presented with an uncomplicated postoperative course, improved exercise tolerance, and no signs of congestive heart failure.
Discussion
Immunoglobulin G4-related disease is an immune-mediated condition that can affect multiple organ systems, characterized by dense lymphoplasmacytic organ infiltration and often elevated serum IgG4 concentrations. 5 The incidence of IgG4-RD is still being determined, with estimates from Japan suggesting a range from 0.28 to 1.08 cases per 100,000 people annually. 5 However, the exact incidence globally is unknown.
The pathophysiology of IgG4-related disease involves complex immune mechanisms, including the oligoclonal expansion of B cells and T follicular helper cells, leading to various tissue manifestations such as lymphadenopathy and organ enlargement. This process is characterized by hypergammaglobulinemia, class switching to IgG4, and the production of plasmablasts, resulting in lymphoplasmacytic infiltration, storiform fibrosis, and elevated IgG4 and IgE levels. 5 A recent large retrospective cohort concluded that patients with IgG4-RD often have an elevated IgE level, correlating to the disease activity. 6 Specifically, a serum IgE >125 KU/L indicated relapsing disease. 6 Our patient’s IgE level was also noted to be elevated.
Around 38% of patients with IgG4-RD present with at least 3 organ systems affected, predominantly causing autoimmune pancreatitis or tubulointerstitial nephritis. 7 The pericardial manifestation of IgG4-related disease is rare and may progress to CP due to plasma cell infiltration and storiform fibrosis of the pericardium. Douman et al 8 reported a review of 32 cases of IgG4 RD pericarditis—only 2 of the cases presented with pericardial involvement as a single manifestation. Based on our literature review, 18 cases of IgG4-RD CP are present. Compared to most previously published IgG4-related CP cases, our case will contribute to the literature due to the scarce data on cases with only pericardial involvement, one of the least common manifestations of IgG4-RD. 9
The 2019 American College of Rheumatology/European League Against Rheumatism (ACR/EULAR) classification criteria for IgG4-RD incorporate a 3-step process initially involving the evaluation of at least one organ involvement of the pancreas, salivary glands, bile ducts, orbits, kidney, lung, aorta, retroperitoneum, pachymeninges, or thyroid gland. 10 Exclusion criteria are applied following clinical, radiological, or pathological evidence of at least one organ involvement. Exclusion criteria consist of a lack of the following: fever, leukopenia, peripheral eosinophilia, unresponsiveness to steroids, disease-specific antibodies, radiologic finding suggestive of malignancy, splenomegaly, prominent neutrophilic inflammation, necrotizing vasculitis, prominent necrosis, primarily granulomatous inflammation, known of diagnosis of the multicentric Castleman’s disease, Crohn’s disease or ulcerative colitis, and Hashimoto thyroiditis. 10 If the patient does not meet any of the inclusion criteria, proceed to the next step, weighted inclusion criteria. Inclusion criteria have a point scale including histopathological, immunostaining, IgG4 levels, and type of organ involvement. Patients with a score of 20 or more meet the classification criteria. These criteria have been validated across a large cohort, demonstrating high specificity and sensitivity for facilitating the diagnosis of IgG4-RD. 10 However, it has limitations in diagnosing IgG4-RD presenting with atypical organ involvement, like in our case, since pericardial involvement is not a part of the classification criteria.
Table 1 summarizes the revised 2020 diagnostic criteria for IgG4-RD, which necessitates a combination of clinical, radiological, serological, and histopathological findings for a diagnosis. 11 These include evidence of organ involvement, elevated serum IgG4 levels (>135 mg/dL), and specific histological features such as dense lymphoplasmacytic infiltration, a high ratio of IgG4-positive plasma cells, and storiform fibrosis or obliterative phlebitis. Patients can be classified with definite, probable, or possible IgG4-RD depending on which criteria are met. 11 Diagnosing IgG4-related CP requires a multifaceted approach, emphasizing the role of multimodality cardiovascular imaging, particularly because serum IgG4 levels lack the specificity and sensitivity needed for a definitive diagnosis. 7
Revised 2020 IgG4-RD Diagnostic Criteria.

As per the international consensus on IgG4-RD management, the first line of treatment is glucocorticosteroids. 12 Disease-modifying agents low-dose cyclophosphamide and mycophenolate mofetil (MMF) have been studied as steroid-sparing medications for IgG4-RD. Clinical trials indicated that 50 to 100 mg/day of cyclophosphamide, in addition to steroid therapy, decreases the relapse rate compared to mono treatment with glucocorticosteroids. 13 Another trial of MMF 1000 to 1500 mg/day in addition to glucocorticosteroid therapy increased the induction rate with lower relapse compared to glucocorticosteroid therapy alone. 14 Rituximab, a B cell-depleting agent, has shown promise in treating refractory cases or in those relapsing after steroid treatment. 15 Generally, anti-inflammatory medications and diuresis are the hallmarks of treatment of IgG4-RD CP; however, pericardiectomy may provide significant symptom relief. 16 Immunosuppressive therapy may be used for patients refractory to glucocorticosteroids or pericardiectomy. 7
Conclusion
In patients with IgG4-related disease presenting pericarditis, CP may develop, marked by symptoms of diastolic heart failure due to long-term inflammation of the pericardium. Diagnosing IgG4-RD-induced CP necessitates comprehensive cardiovascular imaging, given the disease’s propensity for causing progressive organ dysfunction and failure. Early and accurate diagnosis through multimodal imaging is therefore critical for managing and potentially mitigating the disease’s impact on cardiac function.
Footnotes
Acknowledgements
The authors report no acknowledgment
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed Consent
Verbal consent was obtained from the patient for their anonymized information to be published in this article.
Prior Presentation of Abstract Statement
Abstract of this case was presented as titled “A Rare Case of Constrictive Pericarditis Diagnosed via a Multimodality Approach” at 72. Annual American College of Cardiology Meeting in New Orleans, LA, March 4-6 2023.
